Abstracts
The córrego Paulicéia is a direct tributary of the rio Mogi-Guaçu, located in Santa Rita do Passa Quatro, State of São Paulo, Southeastern Brazil. This stream runs inside a savannah-like (cerrado) environment and it associated vegetation. Biological and ecomorphological aspects of its fish community were studied in three stretches of the stream, denominated herein as the upper, middle and lower courses. The fish fauna recorded in this study consisted of 15 species, belonging to five orders and nine families, based in a total of 715 collected individuals representing 1,450 kg of biomass. The most important abiotic factors related to the occurrence and distribution of the fish fauna were the substrate, current speed, and presence or absence of riparian vegetation. The analysis of 336 stomach contents of 13 species demonstrated that 65% of food items are autochthonous, 32% allochthonous, and 3% are of unknown origin. Four alimentary guilds could be identified, based on the predominant food items: omnivores with tendency to insectivory - Astyanax paranae, A. fasciatus and Piabina argentea; larvivores - Corydoras difluviatilis, Cetopsorhamdia iheringi, Phenacorhamdia tenebrosa, Characidium gomesi, Eigenmannia virescens, and Rhamdia quelen; periphyton feeders - Hisonotus sp., Hypostomus ancistroides and Synbranchus marmoratus; and piscivore - Hoplias malabaricus. Reproductions data are presented for Astyanax altiparanae and Hisontus sp. Three ecomorphological groups could be established, based on swimming, feeding, and microhabitat strategies: nektonic (A. fasciatus, A. paranae, Bryconamericus stramineus, P. argentea, H. malabaricus and Phallotorynus jucundus), benthic (C. gomesi, P. tenebrosa, C. iheringi, R. quelen, C. difluviatilis, H. ancistroides and Hisonotus sp.), and necktobenthic (E. virescens and S. marmoratus).
Ichthyofauna; Trophic ecology; Reproduction; Upper Paraná basin; Cerrado Biome
O córrego Paulicéia é um afluente direto do rio Mogi-Guaçu, situado no município de Santa Rita do Passa Quatro, Estado de São Paulo, correndo dentro de um gradiente de fisionomias de cerrado e formações associadas. Aspectos da biologia e ecomorfologia de sua comunidade de peixes foram estudados para três trechos do córrego Paulicéia, aqui denominados de superior, médio e inferior. Durante o período de estudo foram coletadas 15 espécies de peixes pertencentes a cinco ordens e nove famílias, num total de 715 indivíduos coletados e 1.450 kg de biomassa. Os fatores ambientais de maior importância na ocorrência e distribuição dos peixes deste córrego foram o tipo de substrato, a velocidade da corrente, e a presença ou ausência de vegetação ripária submersa. A análise de 336 estômagos pertencentes a 13 espécies mostrou que 65% dos itens alimentares consumidos são autóctones, 32% alóctones e 3% são de origem desconhecida. Quatro guildas alimentares puderam ser definidas com base nos itens alimentares predominantes: onívoros com tendência a insetivoria - Astyanax paranae, A. fasciatus e Piabina argentea; larvófagos - Corydoras difluviatilis, Cetopsorhamdia iheringi, Phenacorhamdia tenebrosa, Characidium gomesi, Eigenmannia virescens e Rhamdia quelen; perifitívoros - Hisonotus sp., Hypostomus ancistroides e Synbranchus marmoratus; e piscívoro - Hoplias malabaricus. Dados sobre reprodução são fornecidos para Astyanax altiparanae e Hisonotus sp. Três agrupamentos ecomorfológicos foram estabelecidos de acordo com o tipo de natação, estratégia alimentar e micro-hábitat ocupado: nectônicos (A. fasciatus, A. paranae, Bryconamericus stramineus, P. argentea, H. malabaricus e Phallotorynus jucundus), bentônicos (C. gomesi, P. tenebrosa, C. iheringi, R. quelen, C. difluviatilis, H. ancistroides e Hisonotus sp.), e nectobentônicos (E. virescens e S. marmoratus).
Biology and ecomorphology of stream fishes from the rio Mogi-Guaçu basin, Southeastern Brazil
Katiane M. Ferreira
Laboratório de Ictiologia de Ribeirão Preto (LIRP), Depto. de Biologia da FFCLRP-USP, Av. Bandeirantes, 3900, 14040-901 Ribeirão Preto, SP, Brazil. katiane@usp.br
ABSTRACT
The córrego Paulicéia is a direct tributary of the rio Mogi-Guaçu, located in Santa Rita do Passa Quatro, State of São Paulo, Southeastern Brazil. This stream runs inside a savannah-like (cerrado) environment and it associated vegetation. Biological and ecomorphological aspects of its fish community were studied in three stretches of the stream, denominated herein as the upper, middle and lower courses. The fish fauna recorded in this study consisted of 15 species, belonging to five orders and nine families, based in a total of 715 collected individuals representing 1,450 kg of biomass. The most important abiotic factors related to the occurrence and distribution of the fish fauna were the substrate, current speed, and presence or absence of riparian vegetation. The analysis of 336 stomach contents of 13 species demonstrated that 65% of food items are autochthonous, 32% allochthonous, and 3% are of unknown origin. Four alimentary guilds could be identified, based on the predominant food items: omnivores with tendency to insectivory - Astyanax paranae, A. fasciatus and Piabina argentea; larvivores - Corydoras difluviatilis, Cetopsorhamdia iheringi, Phenacorhamdia tenebrosa, Characidium gomesi, Eigenmannia virescens, and Rhamdia quelen; periphyton feeders - Hisonotus sp., Hypostomus ancistroides and Synbranchus marmoratus; and piscivore - Hoplias malabaricus. Reproductions data are presented for Astyanax altiparanae and Hisontus sp. Three ecomorphological groups could be established, based on swimming, feeding, and microhabitat strategies: nektonic (A. fasciatus, A. paranae, Bryconamericus stramineus, P. argentea, H. malabaricus and Phallotorynus jucundus), benthic (C. gomesi, P. tenebrosa, C. iheringi, R. quelen, C. difluviatilis, H. ancistroides and Hisonotus sp.), and necktobenthic (E. virescens and S. marmoratus).
Keywords: Ichthyofauna, Trophic ecology, Reproduction, Upper Paraná basin, Cerrado Biome.
RESUMO
O córrego Paulicéia é um afluente direto do rio Mogi-Guaçu, situado no município de Santa Rita do Passa Quatro, Estado de São Paulo, correndo dentro de um gradiente de fisionomias de cerrado e formações associadas. Aspectos da biologia e ecomorfologia de sua comunidade de peixes foram estudados para três trechos do córrego Paulicéia, aqui denominados de superior, médio e inferior. Durante o período de estudo foram coletadas 15 espécies de peixes pertencentes a cinco ordens e nove famílias, num total de 715 indivíduos coletados e 1.450 kg de biomassa. Os fatores ambientais de maior importância na ocorrência e distribuição dos peixes deste córrego foram o tipo de substrato, a velocidade da corrente, e a presença ou ausência de vegetação ripária submersa. A análise de 336 estômagos pertencentes a 13 espécies mostrou que 65% dos itens alimentares consumidos são autóctones, 32% alóctones e 3% são de origem desconhecida. Quatro guildas alimentares puderam ser definidas com base nos itens alimentares predominantes: onívoros com tendência a insetivoria - Astyanax paranae, A. fasciatus e Piabina argentea; larvófagos - Corydoras difluviatilis, Cetopsorhamdia iheringi, Phenacorhamdia tenebrosa, Characidium gomesi, Eigenmannia virescens e Rhamdia quelen; perifitívoros - Hisonotus sp., Hypostomus ancistroides e Synbranchus marmoratus; e piscívoro - Hoplias malabaricus. Dados sobre reprodução são fornecidos para Astyanax altiparanae e Hisonotus sp. Três agrupamentos ecomorfológicos foram estabelecidos de acordo com o tipo de natação, estratégia alimentar e micro-hábitat ocupado: nectônicos (A. fasciatus, A. paranae, Bryconamericus stramineus, P. argentea, H. malabaricus e Phallotorynus jucundus), bentônicos (C. gomesi, P. tenebrosa, C. iheringi, R. quelen, C. difluviatilis, H. ancistroides e Hisonotus sp.), e nectobentônicos (E. virescens e S. marmoratus).
Introduction
The diversified neotropical continental ichthyofauna consists of about 4,500 described species (Reis et al., 2003). In the upper rio Paraná basin the ichthyofauna is composed of about 170 species distributed in 22 families. In the upper rio Paraná basin, headwater streams are inhabited primarily by small-sized species usually with restricted distribution, with little or any comercial value, and largely dependent of the riparian vegetation for feeding, reproduction and shelter (Castro & Menezes, 1998).
Unfortunately, in the last decades, the fish fauna associated with headwaters and streams has been drastically affected by anthropogenic activities. Alterations in chemical, physical and biological properties of these lotic systems have resulted in several changes in the distribution and structure of its fish assemblages (Agostinho et al., 1995; Maret et al., 1997). Among Brazilian biomes, the Cerrado (with a savana-like vegetation) is one of the most reduced in terms of original area, being mostly restricted to areas under permanent conservation policy, such as the area in which the Paulicéia stream is located.
In this paper qualitative and quantitative data of the ichthyofauna of the Paulicéia stream were collected and analyzed according to a longitudinal gradient, including data concerning diet feeding, behavior, gonadal analysis and ecomorphology. The goal of this study is contribute to the knowledge of the fish fauna of the heavily impacted ecosystem of the Brazilian Cerrado biome.
Study site. This study was conducted in the Paulicéia stream, a direct tributary of Mogi-Guaçu River in the upper rio Paraná basin. The Paulicéia stream is located in the municipality area Santa Rita do Passa Quatro, State of São Paulo, Brazil (Fig. 1), and has approximately 15 km in length, draining an area of native cerrado vegetation located inside a protected environmental area named "Pé-de-Gigante". The general climate is sub-tropical, with the rainy season occurring during the summer months (September to April) and the dry season occurring during the winter months (May to August) (Nimer, 1989). The mean annual temperature of the region varies between 18 and 24.5 ºC (Nimer, 1989), and the mean annual rainfall is approximately 1,300 mm (Pivello et al., 1999).
Field work was accomplished in three stretches of 70 meters each along the Paulicéia stream (Figs. 1-2), named as upper, middle, and lower stretches. The upper and the middle stretches have more similar physiographical and physicochemical characteristics compared to the lower stretch (see Table 1).
Material and Methods
Field work was undertaken monthly (October 2000 to September 2001) in two consecutive days. Data were taken in a standardized way during all study period. In the first day, data of air and water temperature (ºC), pH, conductivity, dissolved O2, horizontal transparency of the water, current velocity, and discharge were taken. Four trap nets were distributed equidistantly in each studied stretch, baited with bread and bovine liver. These baits were kept within cotton bags so as not to disturb the gut analysis. In the next morning (second day), the trap nets were removed and the entrapped fishes were fixed. In each 70 m of the studied stretches, block nets (5 mm-mesh) were installed to avoid escape. In the upper and middle stretches, fishing was performed by two collectors using metal hoop nets (70 cm in diameter and 2.5 mm-mesh), usually close to riparian vegetation for approximately 40 minutes. In the lower stretch, the hoop nets were substituted for one seine (2 m long, 1.5 m high and 2.5 mm-mesh).
To verify the efficiency of the fishing methods used during this study, a different methodology was employed in the three studied stretches during the last day of field work. This methodology consist of three electrofishing passes, combined with two passes with a seine and one pass with metal hoop nets (see Castro et al., 2003, for a detailed explanation of this methodology).
Species richness was estimated through ICE (Incidence-based Coverage Estimator; Lee & Chao, 1994), which calculates the correction factor using the incidence of rare species (frequency of occurrence). ICE was processed using the EstimateS program (Colwell, 1997). Shannon-Wiener (H'), Evenness (E') and Simpson (S') diversity indexes were also applied to each one of the studied communities, and the Jaccard Similarity Index was used to compare the three communities. These analyses were performed using the BIO~DAP program (Magurran, 1988). Constancy (c) was calculated after the frequency of each species was recorded, being the relationship expressed as percent by the formula c= (p x 100)/P, where "p" was the number of samples containing the species in question, and "P" was the total number of collections (Dajoz, 1983). Species present in more than 50% of the collection were considered residents, species present in 25 to 50% of the collection were considered accessory, and species present in fewer than 25% of the collection were considered accidental.
All the collected fishes were deposited in the fish collection of the Laboratório de Ictiologia de Ribeirão Preto, Universidade de São Paulo (LIRP 3050-3161).
Diet. The stomach content of all species was analyzed, except for Bryconamericus stramineus and Phallotorynus jucundus, because only one specimen of these genera was sampled. The utilized methods were frequency of occurrence (Bowen, 1992) and percent composition (Hynes, 1950). In cases where stomachs were not discernible, only the anterior portion of the gut was analyzed. The food items were grouped in broad taxonomic or ecological categories according to their origins: aquatic insects, fishes (including fish scales), algae, annelids, thecamoebas; and crustaceans were considered autochthonous; whereas terrestrial insects, vascular plants and organic matter were considered allochthonous. The similarity between the diets was calculated using the software PC-ORD 4 version (McCune & Mefford, 1999), using the Bray-Curtis coefficient (Valentin, 1995). The Morisita index was used to calculate the overlap alimentary index. Values e" 0.58 were considered significant (Linton et al., 1981). The test of Incidence Rate was used to compare diets among species that occurred in two stretches or more, employing the software BioEstat 2.0 (Ayres & Ayres Jr., 2000) in which the p value is considered significant when smaller than 0.01.
Reproduction. The gonads were extracted and examined using a stereomicroscope for macroscopic identification. Four gonadal stages were identified: immature, maturing, mature, and empty (see Vazzoler, 1996).
The gonadal analyses were intended to determine the first sexual maturation length (L50), the maturity stages, sexual proportion [being considered significant value for the chi-square (c2) test above of 3.84] and the reproductive period for females and males (see Vazzoler, 1996 for methodology).
Ecomorphology. For the ecomorphological analyses 10 adult specimens of each species were selected whenever possible. Twenty corporal measurements were taken, following Beaumord & Petrere (1994): a) standard length; b) maximum width; c) maximum body height; d) medium line height; e) head length; f) head height; g) height of the medium line of the eyes; h) mouth width; i) mouth height; j) angle of the mouth; k) pectoral fin length; l) pectoral fin width; m) caudal fin height; n) caudal peduncle length; o) caudal peduncle width; p) caudal peduncle height; q) area of the body (Mahon, 1984); r) dorsal-fin area; s) pectoral-fin area; t) caudal-fin area. All of these measurements were done with calipers and the fin areas were calculated using their contours on metric paper.
From these measurements, 16 ecomorphological attributes were calculated, and their interpretation followed Gatz (1979), Mahon (1984), Watson & Balon (1984), Balon et al. (1986), Winemiller (1991) and Beaumord & Petrere (1994): a) compression index (CI); b) relative depth (RD); c) relative caudal peduncle length (RPL); d) caudal peduncle compression Index (IPC); e) index of ventral flattening (IVF); f) relative area of the dorsal fin (RAD); g) relative area of the pectoral fin (RAP); h) pectoral fin aspect ratio (PAR); i) relative area of caudal fin (RAC); j) caudal fin aspect ratio of (CAR); k) relative head length (RHL); l) eyes position (EP); m) relative mouth width (RMW); n) relative mouth height (RMH); o) ratio of the mouth configuration (RMC); and p) mouth Orientation (MO).
Principal Component Analysis (PCA) using variance/covariance matrix containing the arithmetic average of the 16 attributes for the 15 studied species was used to allow the analyses and interpretation of the morphological variation. In the Tukey Test performed in the GraphPad InStat 3.01 version, each attribute was separately calculated and compared for each one of the three groups established with PCA; the attribute was considered significant when p< 0.05. The Ward's method, with the Person coefficient (Valentin, 1995), was used to join the species according to ecological similarity. Both analyses were done in PC-ORD version 4 (McCune & Mefford, 1999).
Species that are active swimmers were defined as nektonic; species that swim close the bottom were defined as benthic, and species that usually live at mid-water, but that feed and shelter in the substrates, were defined as nektobenthic (Lincoln et al., 1995).
Results
Ichthyofauna. Seven hundred and fifteen fish specimens, belonging to five orders, nine families, 14 genera and 15 species were collected (Fig. 3). Among the collected species, 40% were characiforms, 39% siluriforms, 7% cyprinodontiforms, 7% gymnotiforms and 7% synbranchiforms.
Astyanax paranae and Hisonotus sp. were the most abundant species, representing 85% of the collected specimens. These two species were residents in the three studied stretches, being more abundant in the upper stretch. In the upper stretch, most of the species were residents (50%), in the lower stretch most species were accidental (50%), and in the middle stretch the percentages of resident, accessory and accidental species were the same (33.4%) (Table 2). The highest difference between the observed and the estimated richness occurred in the lower stretch where 12 species were collected and 15 species were estimated (Table 2).
The upper and the middle stretches presented a very similar fish fauna, sharing 67% of its species, although the lower stretch shares only 27% of its species with the upper and the middle stretch. In the lower stretch there were seven additional species, and Hisonotus sp. was the more abundant species (Table 2). Lower diversity occurred in the upper and the middle stretch, with dominance of just a few species, but in the lower stretch a higher diversity and lower dominance occurred (Table 2). The largest indexes of Shannon-Wiener and evenness obtained were in the lower stretch (1.81 and 0.73, respectively), and the largest value of the Simpson index obtained was in the upper stretch (0.435). The largest similarity obtained was in the upper and middle stretches (0.500).
With the electrofishing methodology, no new occurrence was registered in the upper stretch. Synbranchus marmoratus and P. tenebrosa was the new occurrence in the middle and lower stretches, respectively. Furthermore, species that were previously considered accessory and accidental in the lower stretch were collected in abundance.
Diet and food overlap. The stomach contents of 336 specimens were examined; 65% of all items found were autochthonous, 32% were allochthonous and 3% were items that could not be identified. Among the autochthonous items, the most representative were Diptera (mainly Chironomidae larvae), Trichoptera larvae, green algae (mainly Mesotaenium, Spirogyra and Oedogonium), blue algae (mainly Oscillatoria, Anabaena and Raphidiopsis), and diatoms (mainly Navicula, Spirogyra, Tabellaria and Gomphoneis). Among the allochthonous ones, the most representative items were terrestrial insects (mainly Coleoptera, adults of Diptera and Hymenoptera). No significant differences were observed in the diets with respect to the seasons, nor among the same species in different stretches.
Figure 4 shows the percent composition of the gut contents of the fish species grouped in broad taxonomic or ecological categories. Based on similarity of the predominant food items, fish species were grouped into the flowing feeding categories (Fig. 5):
a) Omnivores with tendency to insectivory: These species represented 20% of the species sampled in the Paulicéia stream that eat 60% of allochthonous items (Diptera [Chironomidae], Hymenoptera [Formicidae], Coleoptera, Hemiptera, Isoptera, Pseudoptera, arachnids and vascular plants) and 40% of autochthonous items (Dipterans larvae [Chironomidae and Culicidae], Lepidoptera larvae, Odonata larvae, Ephemeroptera nymphs, Plecoptera, Collembola, Coleoptera, Hemiptera, fishes or fish scales, crustaceans, annelids, aschelminthes, and filamentous algae).
b) Larvivores: These species represented 40% of the species sampled in the Paulicéia stream that eat 85% of autochthonous items (Dipterans larvae [Chironomidae and Culicidae], Lepidoptera larvae, Ephemeroptera Trichoptera, Odonata larvae, Collembola, Plecoptera Coleoptera, thecamoebas, nematods and algae) and 15 % of the food of allochthonous items (Coleoptera and Orthoptera).
c) Periphitivores: These species represented 20% of the species sampled in the Paulicéia stream; all of these consumed items were allochthonous: Cyanophyceae (Oscillatoria, Anabaena and Raphidiopsis), Chlorophyceae (Mesotaenium, Oedogonium, Spirogyra and Chlorella), Bacillariophyceae (Tabellaria, Navicula, Synedra, Bacillaria, Gomphonema, Gomphoneis and Amphora), Eugnophyceae (Phacus and Trachelomonas), Zygnemaphyceae (Closterium, Micrasterias and Cosmarium), filamentous algae, zooplancton (Cladocera and Copepoda), sand and periderm of aquatic plants. In the stomach of S. marmoratus aquatic dipterans were also found (Chironomidae) (see Fig. 4).
d) Piscivore: Only one piscivore species was found in the Paulicéia stream. Its stomach contained only fishes and scales of A. paranae.
Significant feeding overlap was found between 31 pairs of species (see Table 3). In the upper stretch there was food overlap between E. virescens and C. difluviatilis, and between E. virescens and A. paranae. In the middle stretch there was food overlap between C. iheringi, C. difluviatilis and E. virescens; between A. paranae and E. virescens; and between Hisonotus sp. and S. marmoratus. In the lower stretch there was food overlap between C. difluviatilis, P. tenebrosa, P. argentea, C. gomesi and R. quelen; between A. paranae, A. fasciatus and P. argentea; and between H. ancitroides and Hisonotus sp.
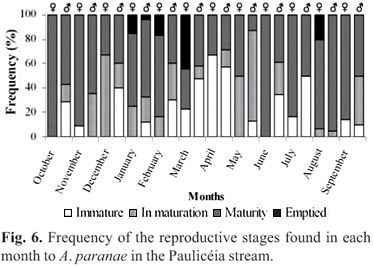
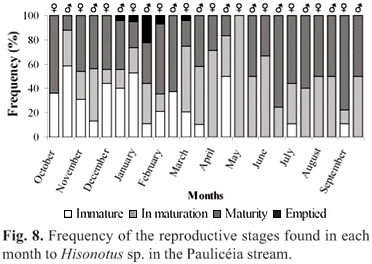
Reproductive behavior. The gonads of 564 specimens, representing the totality of the sampled specimens in the Paulicéia stream were analyzed. During the study period, 67% of the species (A. fasciatus, A. paranae, P. argentea, C. difluviatilis, E. virescens, C. iheringi, Hisonotus sp., H. ancistroides, P. tenebrosa and C. gomesi) presented gonads in maturation or mature, without young individuals. The remaining 33% (A. paranae, A. fasciatus, E. virescens Hisonotus sp. and H. ancistroides) presented both sexually mature and young individuals. Only for A. paranae and Hisonotus sp. was a detailed study on the reproductive behavior possible, because these two species were very abundant during the whole study period.
Two hundred and two specimens of A. paranae were analyzed, being 90 females and 164 males. The sex ratio proportion found in this study was 1.8:1 (male: female) which was significantly different from the expected value 1:1 (c2= 9). Immature individuals, in maturation and maturity were found throughout the year (Fig. 6). Individuals with emptied gonads were sampled in January to March and in October (Fig. 6). The size at first maturation (SL50) differed between males and females, being the size range between 30-35 mm for males and and females, being the size range between 30-35 mm for males and 35-40 mm for females. The smallest mature female had 36.5 mm and was sampled in August, and the smallest mature male had 31.2 mm and was sampled in November. The size in which all of the individuals are capable of actively reproductive process (SL100) is also different for males and females, being the size range between 40-45 mm for males and 45-50 mm for females. Among small sized individuals (15-50 mm) there was a predominance of males, while among the largest individuals (over 50-80 mm) there was a predominance of females (Fig. 7).
Two hundred and eighty-eight specimens of Hisonotus sp. were analyzed, being 148 females and 140 males. The sex ratio presented no significant differences with the expected 1:1 proportion (c2= 0.04). Immature individuals were observed basically throughout the year, except in the months May, June and August (Fig. 8). Individuals in maturation and maturity were found throughout the year (Fig. 8). Individuals with emptied gonads were sampled in January to March (Fig. 8). The size at first maturation (SL50) differed between males and females, with a size range of 15-17.5 mm for males and 20-22.5 mm for females. The smallest mature female had 15.8 mm and was sampled in April, and the smallest mature male had 17.5 mm (sampled in December). The size in which all the individuals were capable of actively participating in the reproductive process (SL100) was also different for males and females, with the size range between 22.5-25 mm for males and 25-27.5 mm for females. Among small sized individuals (12.5-25 mm), there was a predominance of males. On the other hand, between the largest individuals (25-35 mm) there was a predominance of females (Fig. 9). The females of Hisonotus sp. presented oocytes at different sizes during the "in maturation" and "maturation" stages. This is characteristic of species with asynchronic spawning (or multiple spawning), in which oocytes in different stages of development are found inside of the ovary and their elimination occurs when the oocytes are mature (cf. Vazoller, 1996). Usually, there were few mature oocytes close to the urogenital opening and many oocytes in initial stages of development in the anterior portion of the ovary (Fig. 10).
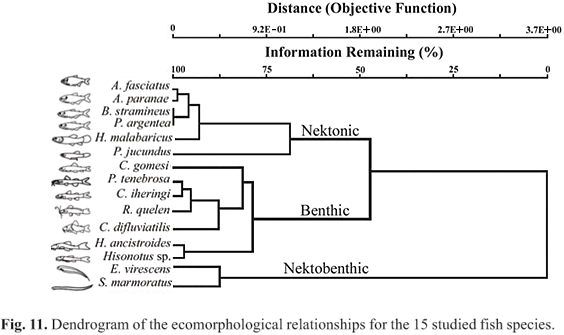
Ecomorphology. The average values of each one of the 16 ecomorphological attributes analyzed for all studied species are presented in Table 4. The first two axes of the PCA explained together 67.8% of the accumulated variance (Table 5).
Three ecomorphological groups were obtained: a) nektonic fishes, b) benthic fishes, and c) the nektobenthic fishes (Figs. 11-12). Three ecomorphological attributes contributed to distinguish nektonic from benthic fishes: compression index, index of ventral flattening, and mouth orientation. Only one attribute contributed to distinguish nektonic from nektobenthic fishes: the caudal fin aspect ratio. The relative head width permits to distinguish benthic from nektobenthic fishes. The same two attributes contributed to distinguish nektonic from benthic fishes, nektonic from nektobenthic fishes, and benthic from nektobenthic fishes: the relative area of the dorsal and pectoral fins. Four attributes contributed to distinguish benthic from nektobenthic fishes and nektonic from nektobenthic fishes: the relative depth, relative area of the caudal fin, relative head length, and relative mouth height (Table 5; Fig. 12).
Discussion
Environmental factors. The water temperature did not varied in the three studied stretches, probably as a result of a thermal balance provided by tree shadows (Gregory et al., 1992 apud Lima & Zakia, 2000). The dissolved oxygen variation also was low in the three studied stretch, probably in function of the thermal balance of the water, since oxygen solubility varies inversely with temperature. The high value of dissolved oxygen in the lower stretch is probably due to the action of the topographical steepness, small depth, rocky bottom, and higher current speed. Those factors can cause an increase in the water agitation, and consequently, a renewal of the interface water layer, increasing the gaseous exchanges.
The horizontal water transparency depends of the amount of suspended material. In none of the studied stretches there was seasonal variation of this characteristic, probably as a function of the presence of riparian vegetation that filters the sediments that would otherwise be discharged by the stream during the rainy season (Lima & Zakia, 2000).
Similar conductivity was found between the upper and middle stretches, but in the lower stretch there was an increase in conductivity values. This increase is probably related to the utilization of agricultural defensive practices in the sugar cane plantations, besides an increase of water volume due to a tributary, the Água Santa stream.
Ichthyofauna. The characiform and siluriform predominance found in the Paulicéia stream corroborates Lowe-McConnell's (1987) previous observation.
Uieda (1984) provided three explanations to justify the occurrence of accessory and accidental species: a) that these species are probably migratory, joining the local community at certain periods of the year; b) that these species present low population density; c) or that these species take shelter in places in which sampling is difficult, resulting in low sample frequencies. Due the applied collecting effort during the entire study period, I believe that the accessory and accidental species have a low population density.
Several accidental species were found in the lower stretch (C. gomesi, C. iheringi, R. quelen, P. tenebrosa and H. ancistroides) which can be explained by the lack of efficiency of the sampling methodology. When the electrofishing methodology was used in the lower stretch, almost all species that were first registered as accidental were captured in higher abundance, besides the new occurrence of P. tenebrosa. Probably, electrofishing was efficient in the lower stretch because 60% of the species that occur there were benthic, and the rocky bottom provides hiding places among the rocks impeding their capture. The methodology applied during the entire study period was appropriate in the upper and middle stretches; electrofishing did not show differences in number of specimens or species collected in these stretches.
The longitudinal distribution of the species is associated with several factors, that can act separately or together (Garutti, 1988). When the three studied stretches were compared, two different situations concerning substratum type (rocky or sandy), depth (shallow or deep), width (narrow or wide), current speed (slowly and fast), and riparian submerged vegetation (absence or presence), were found. I believe that the combination of these abiotic factors determines the occurrence and distribution of the fish fauna of the Paulicéia stream. For example, Eigenmannia virescens was considered a resident species in the upper stretch and accessory in the middle stretch. This species shelters within submerged vegetation (see Garutti, 1988 and Ferreira & Castro, 2005), which suggests that its absence in the lower stretch may be related to the absence of this kind of microhabitat.
The discharge in the lower stretch was three times larger than the value registered in the other two stretches. The occurrence and addition of species in the lower stretch corroborates Lowe-McConnell's (1999) observation in which the water level increase is a factor responsible for the expansion of the microhabitat, increasing food availability, spawning places, and areas that will serve as shelter, since six, six and 12 species were collected in the upper, middle, and lower stretches, respectively.
The fish fauna in the upper and middle stretches were very similar. However, the abundance in the middle stretch was smaller than the abundance in the upper stretch. In the middle stretch, the vegetation and submerged tree branches were not so abundant as in the upper stretch. The large heterogeneity in aquatic vegetation is an important factor in the determination of the species richness, because it increases the offer of refuge areas. The riparian vegetation is very important to nektonic species that use it as temporary shelter between displacements, and benthic species that use it for hiding (Caramaschi, 1986; pers. obs.).
Diet. The three omnivore species with a tendency for insectivory (A. paranae, A. fasciatus and P. argentea) belong to family Characidae. In general, characin species are omnivores, visually dependent to feed, showing high feeding plasticity, capturing prey both at the water column as well as in the substratum. There is a wide literature on the feeding habits in different environments corroborating the results presented herein for the genus Astyanax (e.g. Uieda et al., 1997; Esteves & Galetti, 1995; Esteves, 1996; Castro & Casatti, 1997) and for P. argentea (Godoy, 1975; Ferreira et al., 2002).
The majority of the studied species are larvivores. The importance of the aquatic insects for tropical stream fishes was reported by many authors. Previous studies concerned with the species studied herein or its congeners corroborate the results of the present study: see Obara & Mendes (1990), Aranha et al. (1993) and Castro & Casatti (1997) for C. difluviatilis; see, Casatti & Castro (1998) and Obara & Mendes (1990) for C. iheringi; see Uieda (1984), Costa (1987), Sabino & Castro (1990), Casatti & Castro (1998) and Aranha et al. (1998) for C. gomesi; see Knöppell (1970), Soares (1979), Obara & Mendes (1990) and Castro & Casatti (1997) for E. virescens; see Knöppel (1970), Soares (1979), Costa (1987), Castro & Casatti (1997), Casatti & Castro (1998) and Casatti et al. (2001) for R. quelen.
Algae are a common feeding item present in the diet of neotropical stream fishes. Especially for Loricariidae, algae are the most important item (see Power, 1984 a, b; Lowe-McConnell, 1987; Buck & Sazima, 1995). The loricariids are morphologically specialized for scraping attached material from flat surfaces (Power, 1990). The digestion process in loricariids is facilitated by the elongate coiled intestine. Green algae and blue algae are found intact in their feces, while diatom tests are usually empty, suggesting that they are more easily digested by these species (Power, 1984). The diatoms were the most abundant item found in the guts of Hisonotus sp. and H. ancitroides, which is in accordance to the literature.
Synbranchus marmoratus is known as insectivorous species (see Mérigoux & Ponton, 1998; Meschiatti & Arcifa, 2002). In the present study this species was considered to feed on periphyton, probably because only one specimen was analyzed and the algae found in its stomach were probably ingested accidentally.
Hoplias malabaricus was the unique piscivorous species in the Paulicéia stream. Several authors studied its feeding behavior (Knöppel, 1970; Winemiller, 1989; Silva, 1993; Castro & Casatti, 1997), observing that young individuals feed on small crustaceans and insects while the adults are fish feeders.
Food overlap. The food overlap found among 40% of the species pair does not necessarily invoke the existence of competition. It could be a reflex of the availability of resources that are partitioned (Ross, 1986) or the employment of broad taxonomic categories for such analysis.
There was food overlap between E. virescens and C. difluviatilis, and between E. virescens and A. paranae in the upper stretch. However, these three species have distinct morphological features, which occupy different habitat and use different feeding strategies. Eigenmannia virescens is a nocturnal species, A. paranae and C. difluviatilis are both diurnal predators. Astyanax paranae is a nektonic species, actively swimming during the day at the water column in groups of three to seven individuals using "drift feeding" (cf. Grant & Noakes, 1987) and "surface picking" (cf. Sazima, 1986) as feeding strategies (Ferreira & Castro, 2005). Corydoras difluviatilis is a benthic species, which was observed always solitary or in couples practicing "the hunting by speculation" tactic (cf. Curio, 1976; Ferreira & Castro, 2005).
The food overlap in the middle stretch was between C. iheringi, C. difluviatilis and E. virescens, and between Hisonotus sp. and S. marmoratus. Corydoras difluviatilis is a diurnal species and also practices "the hunting by speculation" tactic (cf. Curio, 1976; see Ferreira & Castro, 2005). Eigenmannia virescens and C. iheringi have nocturnal habits and practice "substratum speculation". However, these two species presented low population densities during the study period (four specimens for both). The food overlap found between Hisonotus sp. and S. marmoratus was probably due to the fact that only one specimen of S. marmoratus was analyzed. Loricariids and S. marmoratus possess different spatial distributions and do not use the same tactics to obtain food (see Silva, 1993; Buck & Sazima, 1995).
In the lower stretch, C. difluviatilis, P. tenebrosa, P. argentea, C. gomesi and R. quelen also presented food overlap. However, these species present different feeding activity periods, spatial distribution and alimentary tactics. Corydoras difluviatilis and C. gomesi are diurnal and benthic species; however, C. difluviatilis was sampled in lentic waters with sandy bottoms, while C. gomesi was sampled from bottom rocks in area of stronger current. Piabina argentea is also a diurnal species, but a nektonic one. Rhamdia quelen and P. tenebrosa are nocturnal and benthic species (see Casatti et al., 2001), however, R. quelen was sampled in lentic waters with sandy bottoms, while P. tenebrosa was sampled in bottom-rock areas of high current.
In the lower stretch also have food overlap between Astyanax paranae, A. fasciatus and P. argentea. These three characins species are diurnal, use similar feeding tactics and occupy similar micro habitats (see Uieda, 1984; Casatti & Castro, 1998; Casatti et al., 2001). Analyzing in more detail the items ingested by these three species, I could observe differences among their diets. Piabina argentea presented a diet more diversified when compared to Astyanax species. The diets of A. paranae and A. fasciatus were similar; however, these species were represented by just a few individuals, decreasing the degree of food competition.
Hypostomus ancistroides and Hisonotus sp. were sympatric in the lower stretch and had a maximum food overlap. This is related to the feeding tactic used by loricariids to feed on periphyton. Probably algae are not a limited food resource in the Paulicéia stream and the preference for certain types of micro habitats probably minimizes the competition for food (Buck & Sazima, 1995; Uieda et al., 1997).
Reproduction. The simple presence of females that are mature or in maturation does not mean that a species is reproducing in the Paulicéia stream. If environmental conditions are not favorable, the oocytes can be absorbed and spawning will not be executed, or spawning cna occur but the eggs and embryos will not survive. The simple presence of young individuals as a sign of reproduction in certain places should also be carefully analyzed because recruitment can occur from adjacent areas (Suzuki & Agostinho, 1997).
The reproductive success of a species can be indicated by the presence of females with signs of recent spawning together with juvenile individuals in a same region (Suzuki & Agostinho, 1997). At the study site, only A. paranae, A. fasciatus and Hisonotus sp. complete the reproductive cycle in Paulicéia stream. I believe that such place is a favorable environment for the development of complete ontogenetic stages only for A. paranae and Hisonotus sp. These species do not accomplish extensive migrations and they occur only in small streams. According to Nomura (1975) and Agostinho et al. (1995), A. fasciatus lives in rivers, realizing migrations and presenting total spawning. The spawn of the larger-sized migratory fishes occur in small streams during the rainy season and the eggs flow for great distance; where they reach flooded areas with favorable conditions for survived (Suzuki & Agostinho, 1997). For such reasons, I believe that A. fasciatus probably swim up the Paulicéia stream to spawn, as it is a direct tributary of the Mogi-Guaçu River and this species was only sampled in the lower stretch studied.
The results indicate that A. paranae and Hisonotus sp. reproduce throughout the whole year in the Paulicéia stream. Barbieri (1992 a, b) and Veregue & Orsi (2003) corroborate the present study with respect to A. paranae. This prolonged reproductive season may be explained as an important strategy to assure the survival in a stochastic environment, because headwaters are not stable as rivers, and their physiography can be changed by erosive actions (Garutti, 1988). This phenomenon causes massive loss of eggs, larvae or juveniles by heavy rain, with sand accumulation on the bottom, suspended material, and more accumulation of predators at headwaters.
In the present study, the expected sexual proportion 1:1 was observed only for Hisonotus sp. No other Hypoptopomatinae species has presently been studied, except for Pseudotothyris obtusa from southeastern Brazil. This species have a proportion of four females to one male (Takeuti et al., 1999).
The proportion among females and males of A. paranae found in the Paulicéia stream corroborates the studies of Barbieri (1992 a, b) in the Fazzari stream, São Paulo State. Barbieri (1992 a, b) also observed that the females are larger than the males and that the adult individuals are ready for reproduction during the whole year. However, Veregue & Orsi (2003), also studying A. paranae in Marrecas stream (Tibagi basin, Paraná State), found a prevalence of males during the studied period. This difference in the sexual proportion can be explained in two ways: by inadequate methodology (the mesh of sieves and hoop nets may be too big, allowing the escape of males that are smaller than females), or by environmental pressures that can alter this proportion of males and females, either positively or negatively (Nikolsky, 1969).
The great size of the mature females of A. paranae and Hisonotus sp. compared to males when reaching the first sexual maturation is probably a reproductive strategy of this species. It should be an advantage for females to be larger than males because they could produce more oocytes. Besides, I also believe that the males are smaller than females because their energy investment for reproduction is lower and they lack parental care.
In spite of several studies on reproductive aspects of neotropical fish have been developed in the last years, the volume of available information is still quite reduced when compared to the number of existing species, and this type of study is restricted to species with some commercial interest. It is important to point out that information on the reproductive period and life cycle are of great value for the understanding of the importance of small streams in the ecology of larger rivers (Garutti, 1988).
Ecomorphology. The goal of an ecomorphological study is to understand the interaction between the morphology of organisms and their ecology (Norton et al., 1995).
The ecomorphological attributes of each species may reflect important aspects of their ecology and can be used as indicators of theirs life habits or adaptations to different habitats. The attributes related to swimming, feeding and habitat were the most important for the distribution of the studied species in the ecomorphological analysis.
a) Nektonic: In this group are included the mid-water swimmers, with adaptive characters to weak to moderate current environments. Small-sized characins present morphological characters related on continuous swimming, such as, compressed bodies, lateral eyes and small pectorals fins. Piabina argentea and B. stramineus presented high values in the pectoral fin ratios when compared to species of Astyanax. This attribute is related to their capacity to travel long distances (Keast & Webb, 1966 apud Mahon, 1984) and to habitats of fast waters. Piabina argentea and B. stramineus were sampled only in the lower stretch where the water velocity is three times larger than in the upper stretch. The small-sized characin species are omnivorous in the Paulicéia stream and they possess subdorsal mouth. Except for P. jucundus and H. malabaricus, the other nektonic species presented high values of caudal fin ratios, indicating active and continuous swimming (Balon et al., 1986). There is a vast literature about the preference of the poecilids and erythrinids for lentic habitats.
The analyzed specimens of H. malabaricus presented high values in head length, width and height relative to the mouth, suggesting that they feed on items that are relatively big. Hoplias malabaricus practice "sit-and-wait predation" strategies to obtain food (cf. Sazima, 1986). Usually they stay immobile in marginal areas in which prey is captured in fast spurts, starting initially from a state of immobility. This species can explore all of the water column to capture prey. I observed this species in the half water column, in rest close to the margin in the upper stretch of Paulicéia stream.
b) Benthic: In this group are fishes with a morphology related to life close to the bottom, typical of riffles. With exception of C. difluviatilis and Hisonotus sp., the other species were sampled only in the lower riffle stretch. In general, this group presents a high caudal peduncle, depressed body, dorsal eyes and large pectoral and caudal fins. Characidium gomesi has a fusiform body, wide pectoral fins, high caudal peduncle, low indexes of ventral flattening, and a small dorsal fin, characteristics that are fundamental to live in a fast water habitat (Balon et al., 1986). Besides, the individuals of this species present small and narrow mouths, suggesting microphagous habits, which were verified in the diet analysis.
Heptapterinae comprise catfishes with slender bodies, sensorial barbells and dorsal eyes, characters that facilitate the exploration of small spaces among the bottom rocks, where they probably feed on aquatic insect larvae. Rhamdia quelen is different than the other heptapterines for presenting a caudal peduncle that is extremely high, indicating little capacity to accomplish maneuvers (Balon et al., 1986). It also present high values in the relative head length and relative mouth height and width, suggesting the ingestion of big prey items.
In the present study, only a single specimen of Rhamdia was collected, having in its stomach only small aquatic insect larvae; however, several studies show the occurrence of larger items in their diet (fishes for example).
Corydoras difluviatilis was collected in the three studied stretches of the Paulicéia stream. Callichthyidae are benthic fishes with typical characteristics as sensorial barbells, eyes dorsolaterally located, high body and large caudal fin that increases their capacity for vertical displacements (Balon et al., 1986). This species also presents a short caudal peduncle, which is related to life in lentic habitats, a slightly dorsoventrally flattened body and wide pectoral fins, allowing these benthic species to remain motionless on the substratum (Balon et al., 1986). Corydoras difluviatilis also possesses a small and subterminal mouth, suggesting that it feeds on small bottom prey, which in fact was observed during the underwater observations (Ferreira & Castro, 2005) and in the diet study.
The two species of Loricariidae are morphologically specialized for life in riffles. They possess ventral lips modified as adhesive organs, depressed body, eyes dorsally located, long caudal peduncle, and relatively large pectorals and caudal fins. Hisonotus sp. and H. ancistroides forage on the surface of rocks and submerged leaves and logs (see Casatti et al., 2001; Ferreira & Castro, 2005).
c) Nektobenthic: In this group are two species phylogenetically distant but that present similar morphological characters (a example of ecomorphological convergence). These two species present elongated bodies, lack dorsal and caudal fins, and their pectoral fins are reduced. These features decrease their capacity to actively swim allowing them to live only in lentic waters. Usually, they are captured among the riparian vegetation, where they remain stationary. The relative head length, and the width and height of their mouth are low, indicating theirs preference for small food items, which was verified in the diet analysis.
The ecomorlogical analysis corroborates the underwater observations, as well as natural history data from the literature. The development of this type of study is very important for understanding the niches occupied by stream fishes in the Neotropical region (Uieda, 1995).
General considerations. The substratum type, current speed, and the presence or absence of submerged marginal vegetation are environmental factors of great importance in constraining the occurrence and distribution of the ichthyofauna of the Paulicéia stream. The riparian vegetation is also very important as food or prey habitat for the studied fish species. Alterations in the riparian vegetation will certainly result in a negative impact for the biotic integrity of a stream. São Paulo is probably the most degraded state of Brazil and there are few studies on the dynamics of fish communities that inhabit the Upper Paraná River streams. Studies like this are useful for future work about evaluation and recovery of degraded areas.
Acknowledgments
I am very grateful to Hertz F. Santos, Luiz S.F. Martins, Lilian Casatti, Renata Stopiglia and Bruno Meireles for help with field work. Alexandre C. Ribeiro and Alex L.A. Melo for help with field work, photos and suggestions. Alex L.A. Melo also helped with riparian plants identification. Marcelo R. de Carvalho provided a detailed review of the English. Murilo de Carvalho helped with the statistical analysis. Léa A. Biagione supplied me with data on pluviometric of the studied area. Instituto Florestal, in special to Heverton J. Ribeiro, for allowed to work in the Parque Estadual de Vassunga (COTEC 266/2000 and SMA 040.643/2000). This study was made possible by funding from the Biota/Fapesp through the grant "Fish diversity of the headwaters and streams of the upper Paraná River system in the State of São Paulo, Brazil" (98/05072-8). The manuscripts benefited from the comments and suggestions of Virginia S. Uieda and Fernando Z. Gibran. I received financial support from Fundação de Amparo à Pesquisa do Estado de São Paulo-FAPESP (00/01918-1). This manuscript was extracted from a master dissertation present to the graduate program in Comparative Biology of the FFCLRP-USP.
Literature Cited
Submitted June 2006
Accepted August 2007
- Agostinho, A. A., A. E. A. M. Vazzoler & S. M. Thomaz. 1995. The high River Paraná basin: limnological and ichthyological aspects. Pp. 59-103. In: Tundisi, J. G., C. E. M. Bicudo & T. Matsumura-Tundisi (Eds.). Limnology in Brazil. Rio de Janeiro, ABC/SBL.
- Aranha, J. M., E. P. Caramaschi & U. Caramaschi. 1993. Ocupação espacial, alimentação e época reprodutiva de duas espécies de Corydoras Lacépède (Siluriformes, Callichthyidae) coexistentes no rio Alambari (Botucatu, São Paulo). Revista Brasileira de Zoologia, 10(3): 453-466.
- Aranha, J. M. R., D. F. Takeuti & T. M. Yoshimura. 1998. Habitat use and food partitioning of the fishes in a coastal stream of Atlantic Forest, Brazil. Revista de Biologia Tropical, 46(4): 951-959.
- Ayres, M. & M. Ayres Jr. 2000. BioEstat 2.0: Aplicações estatística nas áreas das Ciências Biológicas e Médicas. Belém: Sociedade Civíl Mamirauá: CNPq, xii, 272p.
- Balon, E. K., S. S. Crawford & A. Lelek. 1986. Fish communities of the upper Danube River (Germany, Austria) prior to the new Rhein-Main-Donau connection. Environmental Biology of Fish, 15(4): 242-271.
- Barbieri, G. 1982a. Biologia de Astyanax scabripinnis paranae (Characiformes, Characidae) do Ribeirão do Fazzari. São Carlos. Estado de São Paulo. I. Estrutura Populacional e Crescimento. Revista Brasileira de Biologia, 52(4): 579-588.
- Barbieri, G. 1982b. Biologia de Astyanax scabripinnis paranae (Characiformes, Characidae) do Ribeirão do Fazzari. São Carlos. Estado de São Paulo. II. Aspectos Quantitativos da Reprodução. Revista Brasileira de Biologia, 52(4): 589-596.
- Beaumord, A. C. & M. Petrere Jr. 1994. Fish Communities of Manso River, Chapada dos Guimarães, MT, Brazil, Acta Biológica Venezuelana, 15(2): 21-35.
- Bowen, S. H. 1992. Quantitative description of the diet. Pp. 325-336. In: Nielsen, L. A. & D. L. Johnson (Eds.). Fisheries techniques. American Fisheries Society, Bethesda, 468p.
- Buck, S. & I. Sazima. 1995. An assemblage of mailed catfishes (Loricariidae) in southeastern Brazil: distribution, activity and feeding. Ichthyological Exploration of Freshwaters, 6(4): 325-332.
- Caramaschi, E. P. 1986. Distribuição da ictiofauna de riachos das bacias do Tietê e do Paranapanema, junto ao divisor de águas (Botucatu, SP). Unpublished Ph.D. Thesis, Universidade Federal de São Carlos, São Carlos. 245p.
- Casatti, L. & R. M C. Castro. 1998. A fish community of the São Francisco River headwaters riffles, southeastern Brazil. Ichthyological Exploration of Freshwaters, 9(3): 229-242.
- Casatti, L., F. Langeani & R. M. C. Castro. 2001. Peixes de riachos do Parque Estadual Morro do Diabo, bacia do Alto Rio Paraná, SP. Biota Neotropica, 1: 1-15
- Castro, R. M. C. & N. A Menezes. 1998. Estudo diagnóstico da diversidade de peixes do estado de São Paulo. Pp. 1-13. In: R. M. C. Castro (Ed.), C. A. Joly & C. E. M. (Orgs.), Biodiversidade do Estado de São Paulo, Brasil: Síntese do conhecimento ao final do século XX, 6: Vertebrados. Winner Graph - FAPESP, São Paulo, 71p.
- Castro, R. M. C. & L. Casatti. 1997. The fish fauna from a small forest stream of the upper Paraná River basin, Southeastern Brasil. Ichthyological Exploration of Freshwather, 7(4): 337-352.
- Castro, R.M.C. ; L. Casatti; H. F. Santos; K. M. Ferreira; A. C. Ribeiro; R. C. Benine; G. Z. P. Dardis; A. L . A Melo; R. Stopiglia; T. X. Abreu; F. A. Bockmann; M. Carvalho; F. Z. Gibran & F. C. T. Lima. 2003. Estrutura e composição da ictiofauna de riachos do rio Paranapanema, Sudeste e Sul do Brasil. Biota Neotropica, Campinas, 3(1): 1-31.
- Colwell, R. K. 1997. EstimateS 5. Statistical estimation of species richness and shared species from samples. Version 5.0.1. University of Connecticut. (http://viceroy.eeb.uconn.edu/estimates).
- Costa, W. J. E. M. 1987. Feeding habits of a fish community in a tropical coastal stream, rio Mato Grosso, Brasil. Studies on Neotropical Fauna and Environmental, 22(3): 145-153.
- Curio, E. 1976. The ethology of predation. Springer, Belin, 249p.
- Dajoz, R. 1983. Ecologia Geral. Ecologia Geral. São Paulo, Editora Vozes e EDUSP, 472p.
- Esteves, K. E. & P. M. Galetti Jr. 1995. Food partitioning among some characids of a small Brazilian floodplain lake from the Paraná River basin. Environmental Biology of Fishes, 42: 375-389.
- Esteves, K. E. 1996. Feeding ecology of three Astyanax species (Characidae, Tetragonopterinae) from a floodplain lake of Mogi-Guaçu River, Paraná River Basin, Brazil. Environmental Biology of Fishes, 46: 83-101.
- Ferreira, A., N. S. Hahn & R. L. Delariva. 2002. Ecologia alimentar de Piabina argentea (Teleostei, Tetragonopterinae) nas fases de pré e pós-represamento do rio Corumbá, GO. Acta Limnologica Brasiliensia, 14: 45-52.
- Ferreira, K. M. & R. M. C. Castro. 2005. História Natural dos Peixes do córrego Paulicéia, Pp 149-162. In: Varanda, E. M & V. R. Pivello (Eds). Cerrado Pé-de-Gigante: Ecologia e Conservação, São Paulo, Editora, 312p.
- Garutti, V. 1988. Distribuição longitudinal da ictiofauna de um córrego na região noroeste do Estado de São Paulo, Bacia do Rio Paraná. Revista Brasileira de Biologia, 48(4): 747-759.
- Gatz Jr., A. J. 1979. Ecological morphology of freshwater stream fishes. Tulane Studies in Zoology and Botany, 21(2): 91-124.
- Godoy, M. P. 1975. Peixes do Brasil: Subordem Characoidei, bacia do Rio Moji Guaçú. Piracicaba, São Paulo, Editora Franciscana, 4 vols, 846p.
- Grant, J. W. A. & D. L. G. Noakes. 1987. A simple model of optimal territory size for drift-feeding fish. Canadian Journal of Zoology, 65: 270-276.
- GraphPad Software, InStat guide to choosing and interpreting statistical tests , 1998, GraphPad Software, Inc., San Diego California USA, www.graphpad.com
- Hynes, H. B. N. 1950. The food of fresh-water sticklebacks (Gasterosteus aculatus e Pygosteus pungitius), with a review of methods used in studies of the food of fishes. Journal of Animal Ecology, 19: 36-57.
- Knöpell, H. A. 1970. Food of Central Amazonian fishes: contribution to the nutrient ecology of Amazonian rain forest streams. Amazoniana, 2(3): 257-352.
- Lee, S. M. & A. Chao. 1994. Estimating population size via sample coverage for closed capture-recapture models. Biometrics, 50: 88-97.
- Lima, W. de P. & M. J. B. Zakia. 2000. Hidrologia de matas ciliares. Pp. 33-44. In: Rodrigues, R. R. & H. F. Leitão-Filho (Eds.). Matas Ciliares: conservação e recuperação, São Paulo, EDUSP, 320p.
- Lincoln, R. J., G. A. Boxshall & P. F. Clark. 1995. Diccionario de ecología, evolución y taxonomía. México, Fondo de Cultura Económica, 488p.
- Linton, L. R., R. W. Davies & F. J. Wrona. 1981. Resource utilization indices: an assessment. The Journal of Animal Ecology, 50: 283-292.
- Lowe-McConnell, R. H. 1987. Ecological studies in tropical fish Communities. Cambridge, Cambridge Univ. Press, XIII + 382p.
- Lowe-McConnell, R. H. 1999. Estudos ecológicos de comunidades de peixes tropicais. São Paulo, EDUSP, 536p.
- Magurran, A. 1998. BIO~DAP Ecological Diversity and its measurement, Resource Conservation Fundy National Park, Alma New Brunswich, Canada.
- Mahon, R. 1984. Divergent structure in fish taxocenes of north temperate stream. Canadian Journal of Fisheries and Aquatic Sciences, 41: 330-350.
- Maret, T. R., C. T. Robinson & G. W. Minshall. 1997. Fish assemblages and environmental correlates in least-disturbed streams of the Upper Snake River Basin. Transactions of the American Fisheries Society, 126: 200-216.
- McCune, B. & M. J. Mefford. 1999. PC-ORD Multivariate analysis of ecological data, version 4. MjM Software Design, Gleneden Beach, Oregon, 237p.
- Mérigoux, S. & D. Ponton. 1998. Body shape, diet and ontogenetic diet shifts in young fish of the Sinnamary River, French Guiana, South America. Journal of Fish Biology, 52: 556-569.
- Meschiatti, A.J., M. S. Arcifa. 2002. Early life stages of fish and the relationships with zooplankton in a tropical brazilian reservoir: lake Monte Alegre. Brazilian Journal Biology, 62(1): 41-50.
- Nikolsky, G. V. 1969. Theory of fish population dynamics. Edinburgh, Oliver & Boyd, 323p.
- Nimer, E. 1989. Climatologia do Brasil. Secretaria de Planejamento e Coordenação da Presidência da República e IBGE, Rio de Janeiro.
- Nomura, H. 1975. Fecundidade, maturação sexual e índice gônado-somático de lambaris do gênero Astyanax Baird & Girard, 1854 (Osteichthyes, Characidae), relacionados com fatores ambientais. Revista Brasileira de Biologia, 35(4): 775-798.
- Norton, S. F., J. L. Luczkovich & P. J. Motta.1995. The role of ecomorphological studies in the comparative biology of fishes. Environmental Biology of Fishes, 44: 287-304.
- Obara, E. & L. F. Mendes. 1990. Aspectos da biologia dos peixes de um trecho da cabeceira do ribeirão Tamanduá, bacia do Rio Pardo, SP. Unpublished Monograph, Universidade de São Paulo, Ribeirão Preto, 68p.
- Pivello, V. R., C. N. Shida & S. T. Meirelles. 1999. Alien grasses in Brazilian savannas: a threat to the biodiversity. Biodiversity and Conservation, 8: 1281-1294.
- Power, M. 1984a. Grazing responses of tropical freshwater fishes to different scales of variation in their food. Pp. 25-37. In: T.M. Zaret (Ed.). Evolutionary Ecology of Neotropical Freshwater Fishes. Dr. W. Junk Publ., Netherlands, 173p.
- Power, M. 1984b. Depth distributions of armored catfish: predator-induced resource avoidance? Ecology, 65(2): 523-528.
- Power, M. E. 1990. Resource enhancement by indirect effects of grazers: armored catfish, algae, and sediment. Ecology, 71(3): 897-904.
- Reis, R., S. O. Kullander & C. J. Ferraris Jr. 2003. Check list of the freshwater fishes of South and Central America. Porto Alegre, EDIPUCRS, 729p.
- Ross, S. T. 1986. Resource partitioning in fish assemblages: a review of field studies. Copeia 1986: 352-388.
- Sabino, J. & R. M. C. Castro. 1990. Alimentação, período de atividade e distribuição espacial dos peixes de um riacho da Floresta Atlântica (Sudeste do Brasil). Revista Brasileira de Biologia, 50(1): 23-36.
- Sazima, I. 1986. Similarities in feeding behavior between some marine and freshwater fishes in two tropical communities. Journal of Fish Biology, 29: 53-65.
- Silva, C. P. D. 1993. Alimentação e distribuição espacial de algumas espécies de peixes do igarapé do Candirú, Amazonas, Brasil. Acta Amazônica, 23(2-3): 271-285.
- Soares, M. G. M. 1979. Aspectos Ecológicos (alimentação e reprodução) dos peixes do igarapé do Porto, Aripuanã, MT. Acta Amazônica, 9(2): 325-352.
- Suzuki, H. I. & A. A. Agostinho. 1997. Reprodução de peixes do reservatório do Segredo. Pp. 163-181. In: Agostinho, A. A. & L. C. Gomes (Eds.). Reservatório do Segredo: bases ecológicas para manejo. Maringá, Editora da Universidade Estadual de Maringá, 387p.
- Takeuti, D. F., J. R. Verani, J. M. R. Aranha & M. S. Menezes. 1999. Population structure and condition factor of Pseudotothyris obtuse (Hypoptopomatinae) from three costal streams in Southern Brazil. Brazilian Archives of Biology and Technology, 42(4): 397-403.
- Uieda, V. S. 1984. Ocorrência e distribuição dos peixes em um riacho de água doce. Revista Brasileira de Biologia, 44(2):203-213.
- Uieda, V. S. 1995. Comunidade de peixes de um riacho litorâneo: composição, habitat e hábitos. Unpublished Ph.D. Thesis, Universidade Estadual de Campinas, Campinas, 213p.
- Uieda, V. S., P. Buzzato & R. M. Kikuchi. 1997. Partilha de recursos alimentares em peixes em um riacho de serra do sudeste do Brasil. Anais da Academia Brasileira de Ciências, 69(2): 243-252.
- Valentin, J. L. 1995. Agrupamento e ordenação. Pp. 25-55. In: Peres-Neto, P. N., J. L. Valentin & F. A. S. Fernandez (Eds.). Tópicos em tratamento de dados biológicos. Oencologia Brasiliensis, v. II, Rio de Janeiro, PPGE-UFRJ, 161p.
- Vazzoler, A. M. A. de M. 1996. Biologia da reprodução de peixes teleósteos: teoria e prática. Maringá, EDUEM - SBI, 169p.
- Veregue, A. M. L. & M. L. Orsi. 2003. Biologia Reprodutiva de Astyanax scabripinnis paranae (Eigenmann) (Ostheichthyes, Characidae), do ribeirão das Marrecas, bacia do rio Tibagi, Paraná. Revista Brasileira de Zoologia, 20(1): 97-105.
- Watson, D. J. & E. K. Balon. 1984. Ecomorphological analysis of fish taxocenes in rainforest streams of northern Borneo. Journal of Fish Biology, 25: 371-384.
- Winemiller, K. O. 1989. Ontogenetic diet shifts and resource partitioning among piscivorous fishes in the Venezuela llanos. Environmental Biology of Fish, 26: 177-199.
- Winemiller, K. O. 1991. Ecomorphological diversification in lowland freshwater fish assemblages from five biotic regions. Ecological Monographs, 1(4): 343-365.
Publication Dates
-
Publication in this collection
29 Oct 2007 -
Date of issue
Sept 2007
History
-
Accepted
Aug 2007 -
Received
June 2006