Abstracts
The mucous cells (MC) of traíra, Hoplias malabaricus, and jeju, Hoplerythrinus unitaeniatus, two ecologically distinct erythrinid species, were analyzed in fish exposed to deionized (DW) and high ion concentration (NaCl and Ca2+) water (HIW) during 15 days to evaluate the MC responses to ion challenge. MCs are localized in the leading and trailing edge and, interlamellar region of the gill filament epithelium but, in H. unitaeniatus, they are also found in the breathing or lamellar epithelium. MC density is lower in H. malabaricus, the exclusively water-breathing fish, than in H. unitaeniatus, a facultative air-breathing fish. The transference to DW or HIW did not change the MC density and surface area, excepting in H. malabaricus, in the first day of exposure to DW. A single MC containing three types of glycoproteins (neutral, acidic and sulphated) was identified in the gill epithelium of both, H. malabaricus and H. unitaeniatus. The amount (based on the intensity of histochemistry reaction) of these glycoproteins differed between the species and were altered after exposure to DW and HIW showing little adjustments in the amount of mucosubstances in the MC of H. malabaricus and reduction of acidic and sulphated glycoproteins in H. unitaeniatus. The decreasing of these glycoproteins in H. unitaeniatus reduced the mucus protection against desiccation of gill tissue when change the ion concentration in water.
Epithelium; Mucus; Glycoproteins; Deionized water; Ion-rich water
As células mucosas (CM) em traíra, Hoplias malabaricus, e jeju, Hoplerythrinus unitaeniatus, dois eritrinídeos ecologicamente distintos, foram analisadas em espécimes expostos a água deionizada (DW) e água com alta concentração de íons (NaCl + Ca2+, HIW) durante 15 dias para avaliar as respostas das CMs. As CMs estão localizadas no lado aferente e eferente e região interlamelar no epitélio do filamento branquial, mas em H. unitaeniatus, CMs estão presentes no epitélio respiratório ou lamelar. A densidade de CMs é menor em H. malabaricus, espécie com respiração exclusivamente aquática, do que em H. unitaeniatus, espécie com respiração aérea facultativa. A transferência para AD e HIW não alterou a densidade de CMs e a sua superfície, exceto em H. malabaricus, no primeiro dia de exposição à AD. Um único tipo de CM contendo três tipos de glicoproteínas (neutra, ácida e ácida sulfatada) foi identificado no epitélio branquial de H. malabaricus e H. unitaeniatus. A quantidade das glicoproteínas (avaliada pela intensidade da reação histoquímica) diferiu nas duas espécies e foi alterada após a transferência para AD e HIW mostrando pequeno ajuste na quantidade de cada mucosubstância secretada pelas CMs em H. malabaricus e redução de glicoproteínas ácidas e sulfatadas em H. unitaeniatus. A diminuição dessas glicoproteínas em H. unitaeniatus reduz a proteção do muco sobre o tecido branquial quando é alterada a concentração de íons na da água.
Response of mucous cells of the gills of traíra (Hoplias malabaricus) and jeju (Hoplerythrinus unitaeniatus) (Teleostei: Erythrinidae) to hypo- and hyper-osmotic ion stress
Sandro Estevan MoronI; Cássio Arilson de AndradeII; Marisa Narciso FernandesII
IUniversidade Federal do Tocantins, Campus Araguaína, BR 153, km 112, P.O. Box 132, 77807-0160 Araguaína, Tocantins, Brazil. sandromoron@uft.edu.br
IIDepartment of Physiological Sciences, Universidade Federal de São Carlos, P.O. Box 676, 13565-905 São Carlos, São Paulo, Brazil. dmnf@ufscar.br
ABSTRACT
The mucous cells (MC) of traíra, Hoplias malabaricus, and jeju, Hoplerythrinus unitaeniatus, two ecologically distinct erythrinid species, were analyzed in fish exposed to deionized (DW) and high ion concentration (NaCl and Ca2+) water (HIW) during 15 days to evaluate the MC responses to ion challenge. MCs are localized in the leading and trailing edge and, interlamellar region of the gill filament epithelium but, in H. unitaeniatus, they are also found in the breathing or lamellar epithelium. MC density is lower in H. malabaricus, the exclusively water-breathing fish, than in H. unitaeniatus, a facultative air-breathing fish. The transference to DW or HIW did not change the MC density and surface area, excepting in H. malabaricus, in the first day of exposure to DW. A single MC containing three types of glycoproteins (neutral, acidic and sulphated) was identified in the gill epithelium of both, H. malabaricus and H. unitaeniatus. The amount (based on the intensity of histochemistry reaction) of these glycoproteins differed between the species and were altered after exposure to DW and HIW showing little adjustments in the amount of mucosubstances in the MC of H. malabaricus and reduction of acidic and sulphated glycoproteins in H. unitaeniatus. The decreasing of these glycoproteins in H. unitaeniatus reduced the mucus protection against desiccation of gill tissue when change the ion concentration in water.
Key words: Epithelium, Mucus, Glycoproteins, Deionized water, Ion-rich water.
RESUMO
As células mucosas (CM) em traíra, Hoplias malabaricus, e jeju, Hoplerythrinus unitaeniatus, dois eritrinídeos ecologicamente distintos, foram analisadas em espécimes expostos a água deionizada (DW) e água com alta concentração de íons (NaCl + Ca2+, HIW) durante 15 dias para avaliar as respostas das CMs. As CMs estão localizadas no lado aferente e eferente e região interlamelar no epitélio do filamento branquial, mas em H. unitaeniatus, CMs estão presentes no epitélio respiratório ou lamelar. A densidade de CMs é menor em H. malabaricus, espécie com respiração exclusivamente aquática, do que em H. unitaeniatus, espécie com respiração aérea facultativa. A transferência para AD e HIW não alterou a densidade de CMs e a sua superfície, exceto em H. malabaricus, no primeiro dia de exposição à AD. Um único tipo de CM contendo três tipos de glicoproteínas (neutra, ácida e ácida sulfatada) foi identificado no epitélio branquial de H. malabaricus e H. unitaeniatus. A quantidade das glicoproteínas (avaliada pela intensidade da reação histoquímica) diferiu nas duas espécies e foi alterada após a transferência para AD e HIW mostrando pequeno ajuste na quantidade de cada mucosubstância secretada pelas CMs em H. malabaricus e redução de glicoproteínas ácidas e sulfatadas em H. unitaeniatus. A diminuição dessas glicoproteínas em H. unitaeniatus reduz a proteção do muco sobre o tecido branquial quando é alterada a concentração de íons na da água.
Introduction
The structure and function of fish gills have been well described by several authors (Munshi, 1960; Hughes, 1966; Hughes & Munshi, 1979; Hughes et al., 1979; Laurent, 1984; Roy & Munshi, 1986; Munshi & Singh, 1992; Fernandes et al. 1994; Evans et al., 2005; Moraes et al., 2005; Banerjee, 2007; Fernandes et al. 2007). The main cells that constitute the filament epithelium from the inner to the outer cell layer are non-differentiated, neuroepithelial, chloride, mucous and pavement cells. The lamellar or respiratory epithelium has two cell layers which consist of pavement cells in the outermost layer and undifferentiated cells in the innermost one. Several studies on the teleost gill epithelium have emphasized the pavement cells (PVCs) of the lamellar epithelium which are directly related to gas exchange and the chloride cells (CCs) which are related to the ion regulation as well as the changes of these cells in response to the internal and/or external ionic or acid-base environment (Munshi, 1964; Perry & Laurent, 1993; Goss et al., 1994; Moron & Fernandes, 1996; Fernandes & Perna-Martins, 2001; Moron et al., 2003; Sakuragui et al., 2003). Mucous cells (MCs) in the epithelial lining of the gill filament and their responses to environment have been intensively studied by Munshi, Hughes and their associates (Hughes & Munshi, 1979; Singh & Munshi, 1996; Parashar & Banerjee, 1999a, 1999b, 1999c, 2002; Chandra & Banerjee, 2003, 2004; Diaz et al., 2004, 2005; Banerjee, 2007).
The mucus, secreted by the MCs, on the gill surface has been considered an important protection against abrasive injuries by solid materials suspended in water, pathogenic bacteria, parasites (McCahon et al., 1987; Deszfuli et al., 2003), air-exposure (Chandra & Banerjee, 2003, 2004; Banerjee, 2007) and pollutant (Hughes et al., 1979; Hemalatha & Banerjee, 1997a, 1997b; Singh & Munshi, 1996; Ledy et al., 2003; Roberts & Powell, 2005; Singh & Banerjee, 2008). Furthermore, mucus may play a role in the ion regulation (Handy et al., 1989; Roberts & Powell, 2003; Powel, 2007). Mucous cells present in the gill filament epithelium and their secretion may be a mechanism for adaptation to different conditions of the aquatic environment. Changes in the density of the mucous cells of gills and skin (Paul & Banerjee, 1997) have been considered as a response to the changes in the ion concentration in water such as sodium, calcium and chloride (Perry & Wood, 1985; Laurent & Hebibi, 1989) and xenobiotics (Wendelaar Bonga et al., 1989).
Most of the Brazilian freshwater is soft and ion-poor (Esteves, 1998) and fish inhabiting such environment exhibit great diversity in the distribution and density of some gill cells as a compensatory adjustments to maintain the gill functions (Fernandes & Perna-Martins, 2001, 2002; Moron et al., 2003). For example, the traíra, Hoplias malabaricus, a water-breathing fish, and the jeju, Hoplerythrinus unitaeniatus, a facultative air-breathing fish, living in the same environment show different chloride cell density in the filament and lamellar epithelia of gills and also in the response to different ion concentration in water (Fernandes & Moron, 1996; Moron & Fernandes, 1996; Moron et al., 2003). The distribution of CMs in the lamellae of H. unitaeniatus is probably related to air breathing mode of respiration and fish behavior which may be found moving on land to reach water ponds during the dry season.
As the MCs are active cells present in the gills and there are evidences that they respond to environmental changes (Banerjee, 2007), the aim of the present study was to quantify the mucous cells and identify the mucosubstances (glycoproteins) present in these cells in the gills in the two ecologically distinct erythrinid species, H. malabaricus and H. unitaeniatus as well as their responses to ion challenge consisted of exposure to absence and high concentration of Na+, Cl - and Ca2+ in the freshwater environment.
Material and Methods
Experimental animals and holding conditions
Specimens of H. malabaricus (Bloch) (n = 24) weighing 117.8 ± 5.3 g and H. unitaeniatus (Spix) (n = 24) weighing 70.5 ± 2.1 g were collected from the Mogi-Guaçu basin, São Paulo, Brazil and kept in 1000 L aquarium supplied with continuously flowing aerated water (water composition: in mM L-1: Na+ = 0.034 ± 0.002, Ca2+ = 0.043 ± 0.001, K+ = 0.023 ± 0.001, Mg2+ = 0.073 ± 0.003, Cl- = 0.014 ± 0.0004, conductivity = 6.2 ± 1.0 mS, hardness 26.0 as mg L-1 CaCO3, pH 6.8 ± 0.2) at 25 ± 1°C for 1 month. The photoperiod was 12 h light and fish were fed once a week with live fish (Astyanax sp.). During the experiment, no feeding was allowed.
Experimental protocols
The fish were divided into three groups (eight animals in each group). One group served as the control and was transferred to aquarium with water having the same Na+, Ca2+ and Cl- concentration as the acclimation water. The second group was transferred to aquarium with deionized water (Na+ , K+, Ca2+, Mg2+ and Cl- were not detected and pH was 6.5-6.7). The third group was transferred to aquarium containing water enriched with NaCl and Ca2+ (water ion composition in mML-1: Na+ = 0.50 ± 0.01, Ca2+ = 2.13 ± 0.09, K = 0.023 ± 0.01, Mg2+ = 0.0073 ± 0.001, Cl- = 0.678 ± 0.015; conductivity 20.4 mS, hardness 94.7 as mg L-1 CaCO3, pH 6.7-6.8).
The temperature of water of the aquaria were maintained at 25°C and renewed daily without disturbing the fish. After one, two, seven and 15 days, eight fish of each group were randomly removed from their respective experimental aquariums and killed by spinal dislocation. The gill arches were excised, individually separated and fixed in 2.5% glutaraldehyde buffered to pH 7.4 with 0.1 M phosphate buffer, at 4°C. Systematic random samples of filament were taken from dorsal, middle and ventral regions of the each gill arch and processed for light microscopy (LM) and scanning (SEM) and transmission (TEM) electron microscopy.
Mucous cell histochemistry
Fixed gill filament samples were dehydrated in graded ethanol, cleared in xylene and embedded in paraffin and sectioned at 3 µm. Mucins were demonstrated using Alcian blue (AB) at pH 1.0 (sulphated glycoproteins, Lev & Spicer, 1964) and pH 2.5 (acid glycoproteins, Mowry, 1956; Pearse, 1985) and, periodic acid-Schiff (neutral glycoproteins, McManaus, 1948). To identification of MC containing more than one type of glycoproteins PAS technique was also employed in combination with AB pH 1.0 and pH 2.5. Methylation and saponification processes were used to ascertain the chemical nature of the mucosubstances. The reagent groups of the carbohydrate hydroxyl, carboxyl and sulphate ester was chemically blocked. Sections were treated with a solution of HCl in methanol to evidence carboxyl groups and remove O-sulphate esters, n-sulphate groups and amino groups. The methylation was used to block carboxyl groups and to remove ester-sulfate groups. The saponification was an unblock procedure used after the methylation (Kiernan, 1999).
The quantification of MCs and the identification of the glycoproteins were based on the intensity of histochemistry reactions against a background stain, whenever necessary, for better visualization of the gill epithelium. Stained sections were examined under a light microscope Olympus-Micronal CBA-K using systematic 10 random fields in each section. Positive-staining MCs were assessed semi quantitatively as 0 unstained, + weak, ++ moderated, +++ strong.
Transmission (TEM) and scanning (SEM) electron microscopy
Glutaraldehyde fixed samples for TEM analyses were post fixed with 1.0 % osmium tetroxide buffered to pH 7.4 with the same buffer, dehydrated using a graded acetone series and embedded in Araldite 6005 (Ladd Research). Semi-thin sections (0.5 mm) were stained with toluidine blue and examined under an Olympus photomicroscope. Ultra-thin sections (~60 nm) were stained with 2.0% uranyl acetate and 0.5% lead citrate and examined with a JEOL 100 CX transmission electron microscope at 60 or 80 kV.
For SEM analyses, the samples from each gill arch consisted of two to three filament pairs of hemibranchs attached by the septum and fixed with 2.5% glutaraldehyde buffered to pH 7.4 with 0.1 M phosphate buffer. The samples were dehydrated using a graded ethanol series up to absolute ethanol, soaked of 1,1,1,3,3,3-hexamethyldisilazane (Aldrich) and air dried. Filament pairs were glued with silver paint onto the specimen stub, coated with gold (99%, Degussa) in a vacuum sputter and examined on a DSM 940 ZEISS Scanning Microscope at 25 kV. Mucous cell density in contact with the external medium was determined using random SEM photographs of epithelial surface of the filaments at a magnification of 3000x (5 non-contiguous fields). The mucous cell apical surface area was determined by tracing the apical perimeters of all MCs visible in the microphotographs, using a morphometric software program (Sigmascan Pro 3.0 Jandel Scientific Inc) as described by Bindon et al. (1994) and Moron et al. (2003). All cell (complete and partial) areas were used for the determination of the MC density and MC fractional area (MCFA). MCFA represents the fraction of the gill filament epithelial surface of apical surface of MCs. MCFA and MC densities were calculated using the following equations:
MC density = MCFA/average of apical area of whole MCs in microphotograph
The MCFA data were presented as a percentage (%) of filament surface area and MC density data were presented as number of cells/mm2 of filament surface (n/mm2). The mean values for each fish were then calculated and these values used to determine the average of each experimental condition.
Statistical analyses
The statistical analyses were done using the program GraphPad Instat (GraphPad Software, version 3.0, San Diego, CA). The variance analysis (ANOVA) was applied to determine differences among groups. Tuckey's test with 95% confidence limit was applied to compare the mean values whenever a level of significance occurred.
Results
No fish died during the experimental period. Mucous cells number is higher in H. unitaeniatus than in H. malabaricus and did not alter after transfer to deionized water and/or water with high ion concentration. Mucus release into water was not quantified but it was more copious in H. unitaeniatus than in H. malabaricus during exposure to deionized water and in ion-rich water.
The MCs of H. malabaricus and H. unitaeniatus are located mainly in the afferent and efferent gill filament borders. Few mucous cells were also distributed between the secondary lamellae (Fig. 1a, b, c, d). In H. unitaeniatus, the MCs were also found in the lamellar epithelium (Fig. 1b, d).
A typical MC is characterized by a basal irregular nucleus, numerous secretion vesicles with different diameters and electron density which occupy the entire cytoplasm of the cell (Fig. 2a, b). The secretion granules are liberated directly into the environmental water (Fig. 2a, b, c).
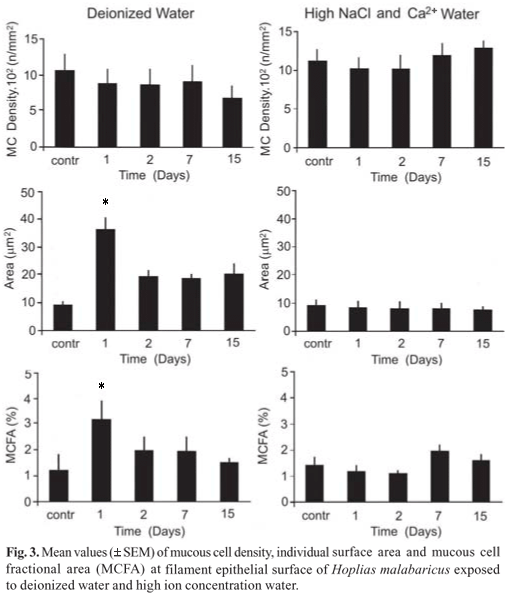
The MC density in the edge of the gill filaments is higher in H. unitaeniatus comparing to H. malabaricus (Fig. 3 and 4). H. malabaricus maintained in DW increased significantly (p < 0.05) the individual MC surface area and fractional area on the 1st day exposure (p < 0.05), but no significant change in the MC density (Fig. 3). No alteration was observed in the MC number of H. malabaricus exposed in water with high ion concentration (Fig. 3). Hoplerythrinus unitaeniatus transferred and maintained in DW and in HIW did not present any alterations in the mucous cell number and density (Fig. 4).
All the three glycoprotein types (acid, acid sulphated and neutral) was identified in the same MC of the gill epithelium of H. malabaricus and H. unitaeniatus however, the amount of each variety of the carbohydrate (intensity of reaction) differed in the two species (Table 1). Following the transfer of the fishes to DW and HIW the amount of glycoproteins in the MCs of the gill epithelium altered (Table 1). The MCs of H. malabaricus however showed slight increase in its neutral glycoproteins content during the two first days of exposure in the deionized water. On the other hand, MCs of H. unitaeniatus presented a reduction in their acidic and sulphated glycoproteins on the 2nd, 7th and 15th day of exposure. In water having high ion concentration there was a reduction of sulphated glycoproteins (7th and 15th day) in H. malabaricus and acidic and sulphated glycoproteins throughout the experimental period. A reduction in neutral glycoproteins was however noticed only in the 7th day of experiment in H. unitaeniatus.
Discussion
In general, the MC density and mucus production vary among different fish species. They also vary at different environmental conditions (Laurent, 1984). Handy & Eddy (1991) demonstrated the absence of mucus in the surface of the gills of unstressed rainbow trout, Oncorhynchus mykiss. Numerous studies have shown that increased amounts of mucus secretion is induced by stresses of acid (Berntssen et al., 1997), salinity (Roberts & Powell, 2003), gill diseases (Powell, 2007), pollutants (Parashar & Banerjee, 2002; Fernandes & Mazon, 2003; Singh & Banerjee, 2008), air exposure and desiccation (Olson, 1996; Parashar & Banerjee, 1999a, 1999b, 1999c; Chandra & Banerjee, 2003, 2004; Banerjee, 2007).
The higher MC density in the gills of the air-breathing erythrinid, H. unitaeniatus than those in the water-breathing, H. malabaricus, may be related to mode of respiration and a possible protection against desiccation when fish is out of water (Parashar & Banerjee, 1999b, 1999c). H. unitaeniatus is known for its moving between ponds (Saul, 1975) during dry season. This hypothesis is supported by the presence of MCs even in its lamellar epithelium which rarely occurs in water-breathing species but is reported in other air-breathing fish such as Clarias batrachus (L.) (Olson, 1996) and Channa striata (Bloch) (Chandra & Banerjee, 2003, 2004) and the knowledge that mucus has low permeability to water (Shepherd, 1989).
Other hypothesis that may explain the differences of MC density between these two erythrinid fish is related to ion regulation. H. unitaeniatus has larger number of chloride cells, including those in the lamellae while H. malabaricus, living in the same environment, has lower chloride cell density and that is restricted only to filament epithelium (Moron & Fernandes, 1996; Moron et al., 2003; Fernandes et al., 2007). The interspecific chloride cell variability between these erythrinid was related to interspecific rate of ion uptake and may reflect differences in their efficiency to maintain ion balance (ratio between ion gain/loss) as suggested by Moron et al. (2003). Chloride and mucous cells in the lamellar epithelium increase the water blood barrier for respiratory gases diffusion (Fernandes et al., 1998; Fernandes & Perna-Martins, 2002), and consequently reduces O2 uptake (Sakuragui et al., 2003) and CO2 excretion (Powell & Perry, 1997). Conversely, the mucous layer covering lamellae seems to have no significant effect on oxygen transfer (Lusts & Gross, 1979) and provides unstirred layer mucus which limits ion movements to diffusion fluxes and also contributes for epithelial ion impermeability. The polyanionic nature of mucus helps to trap neighboring cations at the gill surface resulting in an ionic gradient from mucosal layer to flowing water on the gills (Shephard, 1989). Thus, the higher MC density with increased mucus production in H. unitaeniatus may help this species to reduce water loss when the fish is out of water and the loss of ion when it is within water.
Ion challenge often induces MC responses. Anguilla anguilla has large number of MCs in the gill filament and the variation in the ion concentration in the water alters the number and the distribution of MCs in this species (Laurent & Dunel-Erb, 1980). The MC number in freshwater acclimated Oncorhynchus mykiss was higher in the efferent side of filament around the marginal channel of lamellae (Morgan & Tovell, 1973). However, it decreased when fish was transferred to sea water (Laurent, 1984). Later studies with the same species showed no significance in the MC density following adaptation to sea water although, these MCs increased in the size (Laurent & Hebibi, 1989). Hypostomus plecostomus adapted to water with high ion concentration (NaCl and Ca2+) had a significant increase in the density of the MC while their exposure to distilled water resulted in hypoplasia and hypotrophy of these cells (Fernandes & Perna-Martins, 2002). In H. malabaricus and H. unitaeniatus the responses of MCs to ion challenge were not reflected in their density. However, alterations in their histochemical properties were quite evident.
Neutral glycoproteins have low viscosity and may protect and lubricate gill epithelium against the physical injuries while the acidic and sulphated acid glycoproteins provide high viscosity and may help the adhesion of particles in suspension in water (Sibbing & Uribe, 1985) or prevent the proliferation of potentially pathogenic microorganisms in the epithelial surface (Mittal et al., 1994b). In the case of air-breathing fish, sulphated glycoproteins are also related to the protection against desiccation during emersion to take the atmospheric air (Mittal et al., 1994a; Parashar & Banerjee, 1999a, b; Chandra & Banerjee, 2003, 2004).The transference of the erythrinids, H. unitaeniatus and H. malabaricus, to deionized water (absence of ions) or water with high ion concentration resulted in changes of the chemical characteristics of glycoproteins produced by the MC.
In conclusion, the transfer H. malabaricus and H. unitaeniatus to deionized water and water with high ion concentration caused minor alterations in histochemical nature of the glycoproteins. The MC density however, did not show such fluctuation. The reduction of acidic and sulphated glycoproteins in the gills of H. unitaeniatus may be considered as a disadvantage for this species, mainly in deionized water as the reduction of unstirred mucus layer may reduce the ion gradient close to epithelial surface and favor ion loss. However in the nature fishes never face the stress of deionized water exposure.
Acknowledgements
Financial support was provided by the Brazilian research funding, FAPESP and CNPq to M. N. Fernandes. The authors are grateful to the Laboratory of Electron Microscopy, Department of Morphology of the University of São Paulo, at Ribeirão Preto, and the Laboratory of Structural Characterization of Department of Materials Engineering of Federal University of São Carlos for the use of their facilities and technical assistance and to Nelson Santos (in memorian) for the capture and maintenance of the fish in the laboratory. S. E. Moron and C. A. Andrade thank CNPq for their scholarships.
Literature Cited
Accepted June 14, 2009
Published September 30, 2009
- Banerjee, T. K. 2007. Histopathology of respiratory organs of certain air-breathing fishes of India. Fish Physiology Biochemistry, 33: 441-454.
- Berntssen, M. H. G., F. Kroglund, B. O. Rosseland & S. E. Wendelaar Bonga. 1997. Responses of skin mucous cells to aluminum exposure at low pH in Atlantic salmon (Salmo salar) smolts. Canadian Journal of Fisheries and Aquatic Science, 54(5): 1039-1045.
- Bindon, S. D., J. C. Fenwick & S. F. Perry. 1994. Branchial chloride cell proliferation in the rainbow trout, Oncorhynchus mykiss: implications for gas transfer. Canadian Journal of Zoology, 72: 1395-1402.
- Chandra, S. & T. K. Banerjee. 2003. Histopathological analysis of the respiratory organs of Clarias batrachus (Linnaeus) subjected to the stress of air-exposure. Acta Zoologica Taiwanica, 14(1): 45-64.
- Chandra, S. & T. K. Banerjee. 2004. Histopathological analysis of the respiratory organs of Channa striata subjected to air exposure. Veterinarski Arhiv, 74(1): 37-52.
- Dezfuli, B. S., L. Giari, R. Konecny, P. Jaeger & M. Manera. 2003. Immunohistochemistry, ultrastructure and pathology of gills of Abramis brama from Lake Mondsee, Austria, infected with Ergasilus sieboldi (Copepoda). Diseases of Aquatic Organisms, 53: 257-262.
- Diaz, A. O., A. M. Garcia, C. V. Devincenti & A. L. Goldemberg. 2005. Ultrastructure and Histochemical Study of Glycoconjugates in the Gills of the White Croaker (Micropogonias furnieri). Anatomia, Histologia, Embryologia, 34: 117-122.
- Diaz, A. O., A. M. Garcia, A. H. Escalante & A. L. Goldemberg. 2004. Gycoconjugates in the gills of Odontesthes bonariensis (Teleostei, Atherinopsidae). Biocell, 28(2): 241-245.
- Esteves, F. A.1998. Fundamentos de Limnologia. Rio de Janeiro, Interciência/FINEP, 601p.
- Evans, D. H., P. M. Piermarini & K. P. Choe. 2005. The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous wastes. Physiology Review, 85: 97-177.
- Fernandes, M. N. & A. F. Mazon. 2003. Environmental pollution and fish gill morphology. Pp. 203-231. In: Val, A. L. & B. G. Kapoor (Eds.). Fish Adaptation. Enfield, Science Publishers, 418p.
- Fernandes, M. N. & S. E. Moron. 1996. Respiratory organs in erythrinid fishes. Pp. 93-100. In: Val, A. L., D. J. Randall & D. MacKinley (Eds.). Proceeding of Physiology of Tropical Fishes. San Francisco, American Fisheries Society, 141p.
- Fernandes, M. N., S. E. Moron & M. M. Sakuragui. 2007. Gill morphological adjustments to environment and the gas exchange function. Pp. 93-120. In: Fernandes, M. N., M. L. Glass, F. T. Rantin & B. G. Kapoor (Eds.). Fish Respiration and Environment. Enfield, Science Publisher, 392p.
- Fernandes, M. N. & S. A. Perna-Martins. 2001. Epithelial gill cells in the armoured catfish, Hypostomus cf. plecostomus (Loricariidae). Revista Brasileira Biologia, 61: 69-78.
- Fernandes, M. N. & S. A. Perna-Martins. 2002. Chloride cells responses to long-term exposure to distilled and hard water in the Gill of the armoured catfish, Hypostomus plecostomus (Loricariidae). Acta Zoologica, 83: 312-328.
- Fernandes, M. N., S. A. Perna & S. E. Moron. 1998. Chloride cells apical surface changes in gill epithelia of the armoured catfish, Hypostomus plecostomus L. during exposure to distilled water. Journal of Fish Biology, 52: 844-849.
- Fernandes, M. N., F. T. Rantin, A. L. Kalinin & S. E. Moron. 1994. Comparative study of gill dimensions of three erythrinid species in relation to their respiratory function. Canadian Journal of Zoology, 72: 160-165.
- Goss, G. G., C. M. Wood, P. Laurent & S. F. Perry. 1994. Morphological responses of the rainbow trout (Oncorhynchus mykiss) gill to hiperoxia, base (NaHCO3) and acid (HCl) infusions. Fish Physiology and biochemistry, 12(6): 465-477.
- Handy, R. D. & F. B. Eddy. 1991. The absence of mucus on the secondary lamellae of unstressed rainbow trout, Oncorhynchus mykiss (Walbaum). Journal of Fish Biology, 38: 153-155.
- Handy, R. D., F. B. Eddy & G. Romain. 1989. In vitro evidence for the ionoregulatory role of rainbow trout mucus in acid, acid/aluminum and zinc toxicity. Journal of Fish Biology, 35: 737-747.
- Hemalatha, S. & T. K. Banerjee. 1997a. Histopathological analysis of acute toxicity of zinc chloride to the respiratory organs of the air-breathing catfish Heteropneustes fossilis (Bloch). Veterinarski Arhiv, 67: 11-24.
- Hemalatha, S. & T. K. Banerjee. 1997b. Histopathological analysis of sublethal toxicity of zinc chloride to the respiratory organs of the air-breathing catfish Heteropneustes fossilis (Bloch). Biological Research, 30: 11-21.
- Hughes, G. M. 1966. The dimensions of gills in relation to their function. Journal of Experimental Biology, 45: 177-195.
- Hughes, G. M. & J. S. D.Munshi. 1979. Fine structure of the gills of some Indian air-breathing fishes. Journal of Morphology, 160: 169-194.
- Hughes, G. M., S. F. Perry & V. M. Brown. 1979. A morphometric study of the effects of nickel, chromium and cadmium on secondary lamellae of rainbow trout. Water Resources, 13: 665-679.
- Kiernan, J. A. 1999. Histological and Histochemical Methods: Theory & Practice. Oxford, Butterworth-Heinemann, 502p.
- Laurent, P. 1984. Internal morphology of the gill. Pp. 73-183. In: Hoar W. S. & D. J. Randall (Eds.). Fish Physiology, vol X. Orlando, Academic Press, 454p.
- Laurent, P. & S. DuneL-Erb. 1980. Morphology of gill epithelia in fish. American Journal of Physiology, 238: 147-519.
- Laurent, P. & N. Hebibi. 1989. Gill morphometry and fish osmoregulation. Canadian Journal of Zoology, 67: 3055-3063.
- Ledy, K., L. Giamberini & J. C. Pihan. 2003. Mucous cell responses in gill and skin of brown trout Salmo trutta fario in acidic, aluminium-containing stream water. Diseases of Aquatic Organisms, 56(3): 235-240.
- Lev, R. & S. S. Spicer. 1964. Specific staining of sulphate groups with alcian blue at low pH. Journal of Histochemical and Cytochemical, 2: 309-309.
- McCahon, C. P., D. Pascoe & M. Kavanagh. 1987. Histochemical observations on the salmonids Salmo salar L. and Salmo trutta L. and the ephemeropterans Baetis rhodani (Pict.) and Ecdyonurus venosus (Fabr.) following a simulated episode of acidity in an upland stream. Hydrobiology, 153: 3-12.
- McManus, J. F. A. 1948. Histological and histochemical uses of periodic acid. Stain Technology, 23: 99-108.
- Mittal, A. K., T. Ueda, O. Fujimori & K. Yamada.1994a. Histochemical analysis of glycoproteins in the epidermal mucous cells and sacciform cells of an Indian swamp eel Monopterus cuchia (Hamilton) (Synbranchiformes, Pisces). Acta Histochemical and Cytochemmical, 27(3): 193-204.
- Mittal, A. K., T. Ueda, O. Fujimori & K. Yamada. 1994b. Histochemical analysis of glycoproteins in the unicellular glands in the epidermis of an Indian freshwater fish Mastacembelus pancalus (Hamilton). Histochemical Journal, 26: 666-677.
- Moraes, M. F. P. G., S. Höller, O. T. F. Costa, M. L. Glass, M. N. Fernandes & S. F. Perry. 2005. Morphometric comparison of the respiratory organs of the South American lungfish, Lepidosiren paradoxa (Dipnoi). Physiological and Biochemical Zoology, 78: 546-559.
- Morgan, M. & P. W. A. Tovell. 1973. The structure of gill of the trout, Salmo gairdneri (Richardson). Zellforsch Mikrosk Anatomic, 142: 147-162.
- Moron, S. E. & M. N. Fernandes. 1996. Pavement cell structural differences on Hoplias malabaricus gill epithelia. Journal of Fish Biology, 49: 357-362.
- Moron, S. E., E. T. Oba, C. A. Andrade & M. N. Fernandes. 2003. Chloride cell responses to ion challenge in two tropical freshwater fish, the erythrinids Hoplias malabaricus and Hoplerythrinus unitaeniatus Journal of Experimental Zoology, 298A: 93-104.
- Mowry, R. W. 1956. Alcian blue techniques for the histochemical study of acidic carbohydrates. Journal of Histochemical and Cytochemmical, 4: 407-408.
- Munshi, J. S. D. 1960. The structure of the gills of certain fresh water teleosts. Indian Journal Zoology Memory, 4: 1-40.
- Munshi, J. S. D. 1964. Chloride cell in the gills of freshwater telosts. Quantitative Microscopy Science, 105: 179.
- Munshi, J. S. D. & A. Singh. 1992. Scanning electron microscopic evaluation of effects of low pH on gills of Channa punctata (Bloch). Journal of Fish Biology, 41: 83-89.
- Olson, K. R. 1996. Scanning electron microscopy of the fish gill. Pp. 31-45. In: Munshi, J. S. D. & H. M. Dutta (Eds.). Fish Morphology. Horizon of New Research. Brookfield, CRC Press, 300p.
- Parashar, R. S. & T. K. Banerjee. 1999a. Effect of dehydration stress on the accessory respiratory organs of the air-breathing catfish Heteropneustes fossilis (Bloch). Pp. 413-425. In: Val, A. L. & V. M. F. Almeida-Val (Eds.). Biology of Tropical Fishes. Manaus, INPA.
- Parashar, R. S. & T. K. Banerjee. 1999b. Response of the aerial respiratory organs of the air-breathing catfish Heteropneustes fossilis (Bloch) to extreme stress of desiccation. Veterinarski Arhiv, 69: 63-68.
- Parashar, R. S. & T. K. Banerjee. 1999c. Response of the gill of the air-breathing catfish Heteropneustes fossilis (Bloch) to acute stress of desiccation. Journal of Experimental Zoology, 2: 169-174.
- Parashar, R. S. & T. K Banerjee. 2002. Toxic impact of lethal concentration of lead nitrate on the gills of air-breathing catfish Heteropneustes fossilis (Bloch). Veterinarski Arhiv, 72: 167-183.
- Paul, I. & T. K. Banerjee. 1997. Histopathological changes induced by ambient ammonia (ammonium sulphate) on the opercular linings of the catfish Heteropneustes fossilis Diseases of Aquatic Organisms, 28: 151-161.
- Pearce, A. G. E. 1985. Histochemistry-theoretical and applied. Vol. II. Edinburgh/London, Churchill Livingston Inc., 441-1055p.
- Perry, S. F. & P. Laurent. 1993. Environmental effects on fish gill structure and function. Pp. 231-264. In: Fish Ecophysiology. J. C. Rakin & F. B. Jensen (Eds.). London, Chapman and Hall, 421p.
- Perry, S. F. & C. M. Wood. 1985. Kinetics of brachial calcium uptake in the rainbow trout: effects of acclimation to various external calcium levels. Journal of Experimental Biology, 116: 411-433.
- Powel, M. D. 2007. Respiration in infectious and non-infectious gill diseases. Pp. 317-339. In: Fernandes, M. N., M. L. Glass, F. T. Rantin & B. G. Kapoor. (Eds.). Fish Respiration and Environment. Enfield, Science Publisher, 392p.
- Powell, M. D. & S. F. Perry. 1997. Respiratory and acid-base- disturbances in rainbow trout blood during exposure to chloramine-T under hypoxia and hyperoxia. Journal of Fish Biology, 50: 418-428.
- Roberts, S. D. & M. D. Powell. 2003. Comparative ionic flux and gill mucous cell histochemistry: effects of salinity and disease status in Atlantic salmon (Salmo salar L.). Comparative Biochemistry and Physiology A, 134(3): 525-537.
- Roberts, S. D. & M. D. Powell. 2005. The viscosity and glycoprotein biochemistry of salmonid mucus varies with species, salinity and the presence of amoebic gill disease. Journal Comparative Physiology B, 175: 1-11.
- Roy, P. K. & J. S. D. Munshi. 1986. Scanning electron microscopic evaluation of effects of saponin on gills of the climbing perch Anabas testudineus (Bloch) (Anabantidae, Pisces). Indian Journal of Experimental Biology, 24: 511-516.
- Sakuragui, M. M., J. R. Sanches & M. N. Fernandes. 2003. Gill chloride cell proliferation and respiratory responses to hypoxia of the neotropical erythrinid fish Hoplias malabaricus Journal of Comparative Physiology, B173: 309-317.
- Saul, W. G. 1975. An ecological study of fishes at a site in upper Amazonian Ecuador. Proceedings of Academy of National Science, Phyladelphia, 127: 93-134.
- Shephard, K. L. 1989. The effect of mucus and mucilaginous materials on ion distributions at epithelial surfaces. Pp. 123-130. In: Chantler, E. & N. A. Ratcliffe (Eds.). Mucus and Related Topics. Cambridge, Company of Biologist Limited, 471p.
- Sibbing, F. A. & R. Uribe.1985. Regional specialization in the oro-pharyngeal wall and food processing in the carp Cyprinus carpio Netherlands Journal of Zoology, 35: 377-422.
- Singh, A. K. & T. K. Banerjee. 2008. Toxic effects of sodium arsenate (Na2HAsO4 7H2O) on the skin epidermis of air-breathing catfish Clarias batrachus (L.). Veterinarski Arhiv, 78(1): 73-88.
- Singh, A. & J. S. D. Munshi. 1996. Scanning electron microscopic evaluation of the effects of short and long term exposure of copper and mercury on the gills of Channa punctata (Bloch). Journal Freshwater Biology, 8: 51-53.
- Wendelaar-Bonga, S. E. & C. J. M. Van Der Meij. 1989. Degeneration and death, by apoptosis and necrosis the pavement and cells the gills of the teleost Oreochromis mossambicus Cell and Tissue Research, 255: 235-243.
Publication Dates
-
Publication in this collection
30 Oct 2009 -
Date of issue
Sept 2009
History
-
Accepted
30 Sept 2009 -
Received
14 June 2009