Abstracts
Fish damage in hydroelectric stations has been extensively reported, but further investigations focusing on Brazilian migratory species, especially large fish, are needed. The present study describes swimbladder abnormalities in piapara Leporinus obtusidens, a migratory physoclistous species, collected downstream from the Funil Hydroelectric station dam, Minas Gerais, Brazil. The 117 specimens collected were dissected for swimbladder evaluation in the coelomic cavity. The abnormalities detected in 14 of the fish (12%) were swimbladder rupture with exteriorization (N = 12) and formation of sac-like projections (N = 2). The specimens that had swimbladder abnormalities weighed over 1300 g for both sexes. These abnormalities may be caused by bladder infections, however, inflammation process were not detected in these fish. The presence of anomalies in swimbladder suggests that the individuals were subjected to sudden depressurization, possibly related to passage through the turbines. Further comparative studies are needed to corroborate this hypothesis.
Fish mortality; Migratory fish; Pressure changes; Reservoir
Os danos que as usinas hidrelétricas causam na fauna aquática tem sido relatados, mas novos estudos focando as espécies nativas, especialmente os peixes de grande porte são necessários. O presente estudo descreve as anomalias de bexiga natatória em piapara Leporinus obtusidens, uma espécie de peixe migratório, coletados a jusante da barragem da estação Hidrelétrica do Funil, Minas Gerais, Brasil. Os 117 exemplares coletados foram dissecados para avaliação da bexiga natatória na cavidade celomática. As anormalidades detectadas em 14 dos peixes capturados (12%) foram ruptura do tecido com exteriorização da bexiga natatória (N = 12) e formação de projeções da bexiga (N = 2). Os exemplares que apresentavam anormalidades de bexiga, possuíam peso superior a 1300 g em ambos os sexos. Este tipo de anomalia pode ser causado por processo infeccioso da bexiga, no entanto, não foram detectados processos inflamatórios nesses peixes. O quadro de anomalias em bexiga natatória em piapara sugere que os indivíduos foram submetidos a uma brusca despressurização, possivelmente relacionada à passagem pelas turbinas. Outros estudos comparativos são necessários para corroborar esta hipótese.
SCIENTIFIC NOTE
Swimbladder abnormalities in piapara (Leporinus obtusidens) captured downstream of the Funil Dam
Viviane de Oliveira FelizardoI; Estefânia de Souza AndradeII; Luis David Solis MurgasII; Elissandra Ulbricht WinkalerIII; Flademir WoutersII
IDepartamento de Zootecnia, Universidade Federal de Lavras. 37200-000 Lavras, MG, Brazil. viviofbio@yahoo.com.br
IIDepartamento de Medicina Veterinária, Universidade Federal de Lavras. Lavras, MG, Brazil
IIICentro de Ciências Agrárias, Ambientais e Biológicas, Universidade Federal do Recôncavo da Bahia. Cruz das Almas, BA, Brazil
ABSTRACT
Fish damage in hydroelectric stations has been extensively reported, but further investigations focusing on Brazilian migratory species, especially large fish, are needed. The present study describes swimbladder abnormalities in piapara Leporinus obtusidens, a migratory physoclistous species, collected downstream from the Funil Hydroelectric station dam, Minas Gerais, Brazil. The 117 specimens collected were dissected for swimbladder evaluation in the coelomic cavity. The abnormalities detected in 14 of the fish (12%) were swimbladder rupture with exteriorization (N = 12) and formation of sac-like projections (N = 2). The specimens that had swimbladder abnormalities weighed over 1300 g for both sexes. These abnormalities may be caused by bladder infections, however, inflammation process were not detected in these fish. The presence of anomalies in swimbladder suggests that the individuals were subjected to sudden depressurization, possibly related to passage through the turbines. Further comparative studies are needed to corroborate this hypothesis.
Key words: Fish mortality, Migratory fish, Pressure changes, Reservoir.
RESUMO
Os danos que as usinas hidrelétricas causam na fauna aquática tem sido relatados, mas novos estudos focando as espécies nativas, especialmente os peixes de grande porte são necessários. O presente estudo descreve as anomalias de bexiga natatória em piapara Leporinus obtusidens, uma espécie de peixe migratório, coletados a jusante da barragem da estação Hidrelétrica do Funil, Minas Gerais, Brasil. Os 117 exemplares coletados foram dissecados para avaliação da bexiga natatória na cavidade celomática. As anormalidades detectadas em 14 dos peixes capturados (12%) foram ruptura do tecido com exteriorização da bexiga natatória (N = 12) e formação de projeções da bexiga (N = 2). Os exemplares que apresentavam anormalidades de bexiga, possuíam peso superior a 1300 g em ambos os sexos. Este tipo de anomalia pode ser causado por processo infeccioso da bexiga, no entanto, não foram detectados processos inflamatórios nesses peixes. O quadro de anomalias em bexiga natatória em piapara sugere que os indivíduos foram submetidos a uma brusca despressurização, possivelmente relacionada à passagem pelas turbinas. Outros estudos comparativos são necessários para corroborar esta hipótese.
River impoundment for hydroelectric generation is a common practice in the large hydrographic basins of South America. Currently, almost all of these basins have dams or are affected by the construction of dams. Recent studies carried out in Brazil estimate that more than 700 large water reservoirs have been built (Agostinho et al., 2007). River regulation by dams is one of the primary causes of fish diversity reduction and depletion of migratory species (Oldani et al., 2007). Populations of migratory species may indeed be reduced by the interruption of their natural routes (Larinier & Marmulla, 2004; Okada et al., 2005).
Migratory species can travel hundreds of kilometers to find sites that are adequate for each phase of their life cycle; e.g. some species swim upstream to spawn (Carolsfeld et al., 2003). Upstream fishways, such as fish lift and fish ladders, are used to restore the connectivity of habitats that are separated by dams along migration routes. However, reservoir hydrodynamics is quite different from that of the original river and may itself provide an additional internal barrier to migratory transit, especially downstream (Agostinho et al., 2007).
Besides blocking migration routes, dams may cause fish injury and even death as a result of contact with physical components of the powerhouse generator (e.g. turbines and spillways) as well as the conditions created during hydroelectric station operation (Agostinho et al., 2007). When fish pass through the generator unit they are subjected to a range of adverse conditions caused by water flow, such as turbulence, mechanical shock, cavitation, shear bond strength and changes in water pressure (Cada et al., 1997).
Fish may suffer a pressure change from 5-10 atm to negative values while passing through a Francis-type turbine (Pavlov et al., 2002). The effects of sudden pressure variation may cause generalized hemorrhage, stomach eversion or rupture, dilatation of the ocular globe and swimbladder gas embolism, distension or rupture (Albernethy et al., 2002).
Physical abnormalities may compromise swimbladder function, resulting in serious consequences for fish. Located in the coelomic cavity, the swimbladder is a gas-filled sac that contracts and expands, promoting changes in body density and allowing the animal to achieve neutral buoyancy, float or dive in the water column (Strand et al., 2005; Robertson et al., 2008). This structure also plays an important role for sound production and perception as well as in the respiratory processes (Peruzzi et al., 2007). Albernethy et al. (2002) reported that serious injuries such as distention and rupture in the swimbladders of physostomous fish that pass through Kaplan type turbines may occur. Further studies are needed to investigate whether hydropower stations also affect swimbladder integrity in physoclistous fish.
Piapara Leporinus obtusidens ( Valenciennes, 1837) is a large migratory physoclistous fish species that may suffer the negative effects of dam construction in its natural habitats in South and Southeast Brazil (Glusczak et al., 2006). It lives in lotic and lentic environments, small streams and migrates upstream once a year to spawn (Oldani et al., 1992). Piaparas are omnivorous and because of their intermediary position in the food chain, are important for ecosystem equilibrium (Araya et al., 2005). They reach 40 cm in length, a weight of 6 kg and are economically important - it is indeed one of the most captured species in South Brazil (Gioda et al., 2007; Taitson et al., 2008).
Although fish damage in hydroelectric stations has been extensively reported, further investigations focusing on Brazilian migratory species are needed, especially considering physoclistous fish of ecological and economic importance. Accordingly, the present study describes swimbladder abnormalities in piaparas (Leporinus obtusidens) collected downstream from the Funil Hydroelectric station dam, Minas Gerais, Brazil.
Study Area
The Grande River, covers 143,000 km2 in Southeast Brazil. A number of hydroelectric stations built in this river basin or in its contributing basins are main energy sources in the country.
The Funil Hydroelectric station (FHS) is located on the Grande River between the City of Perdões and Lavras (MG), with 21º05'S and 44º55'W. The construction of FHS began in 2000 and operations commenced in 2003, it uses Kaplan type turbines, in which a flow of 191 m3.s-1 produces an output of 61.5 MW per turbine, with three turbines.
The FHS has a fish transposition system of the type elevator designed to allow migration and reproduction processes of the different fish species from downstream to upstream of the dam.
Specimens collection
We captured 47 males (200 to 3500 g) and 70 females (200 to 2800 g) piaparas Leporinus obtusidens between September 2006 and August 2007 in the Grande River, downstream from the FHS dam. The downstream part of the Funil Dam has a mean depth of 12 meters. Fish collection was carried out at 15 day-intervals, in the morning, using rod and reel with cow heart and worms as bait. Mean temperature during the fish collection period was 20.5ºC (sd = 3.2) and the highest rainfall indexes were observed from November to February, with a peak of 17.9 mm occurring in January. Although dissolved oxygen was not measured during fish capture, data from 2005 report variations from 6.0 to 6.6 mg.L-1 over the year.
The captured fish were anesthetized with benzocaine (80 mg.L-1) and transported to the laboratory inside ice-filled coolers. We identified the specimens and measured total weight, total length, and standard length.
We made a wide ventral incision in each animal to observe the internal organs and sex was determined by macroscopic gonad examination. Swimbladder was evaluated macroscopically for deformities and microorganism infection immediately after the other contents in the coelomic cavity were removed, because swimbladder infections can cause swelling which can lead to increased swimbladder. To verify the presence of inflammation was performed histopathological evaluation of tissue removed from the swimbladder. We collected swimbladder fragments, fixed them in 10% formalin and processed them using routine histological techniques. The histological sections underwent HE (hematoxylin and eosin) and Masson trichrome staining H for further evaluation under optical microscope.
The estimated age of the specimens showed abnormal swimbladder was calculated from the length of the specimens, according to the methodology employed by Araya et al. (2005).
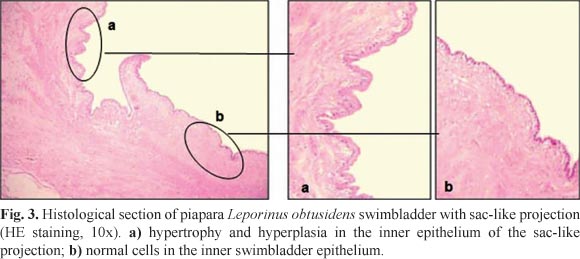
The biometric data of piaparas captured during the experimental collection period are presented in Table 1. Compared to the normal specimens (Figs. 1a, 2a), 12% of the specimens analyzed (6 males and 8 females) had swimbladder abnormalities. The most frequent abnormality, observed in 12 fish, consisted of exteriorization of the posterior extremity of the swimbladder across a rupture in the coelomic cavity. This rupture was posterior to the urogenital opening and anterior to the anal fin base (Figs. 1b, 2b). Another abnormality found in 2 fish was a sac-like projection in the ventral median portion of the organ without exposure to the external medium (Figs. 1c, 2c).
Inflammatory processes were not observed in the fresh samples. When healthy swimbladders were compared to those with sac-like projections (Fig. 3), the histological sections from the abnormal organs revealed a rupture in the muscle layer and replacement with fibrous connective tissue. The epithelium lining the inner portion of the sac-like projection had hypertrophy and hyperplasia (Fig. 3a) along with normal cells.
The estimated age of the specimens with abnormal swimbladder was 7 to 8 years old.
We found deformed swimbladder in 12% of the Leporinus obtusidens specimens analyzed, and installation of the FHS is likely one of the causal factors of this abnormality. The abnormal specimens collected between 2006 and 2007 were 7 to 8 years old, who also report that female L. obtusidens are longer than same-age males. They were thus occupying the environment before the FHS went into operation in 2003. This suggests that the studied fish may. Have undergoing decompression, possibly by passing through the turbines, which is the most likely. However, although less frequent, turn on the turbines or taken charge of by machine, and the opening of spillways can throw the fish back to the surface abruptly and lead to a similar condition. The swimbladder of healthy piaparas was rectilinear, elongated and contained 2 distinct compartments (Fig. 1). In contrast, the abnormal swimbladders had distended walls or sac-like projections. These could have been caused by microorganism contaminations that trigger infectious processes. The rhabdovirus from the family Rhabdoviridae, for instance, frequently infects young individuals, causing anorexia, weight loss, dilatation of the visceral cavity due to swimbladder dilatation, hemorrhages and necrotic wounds in the swimbladder (OIE, 2008). In addition, contamination by worms from the superfamily Dracunculoidea (Anguillicola crassus) may cause severe lesions and swimbladder inflammation in young specimens (Haenen et al., 1994). However, the piaparas evaluated in the present study showed no evidence of inflammatory process. This type of pathology was therefore ruled out as a source of swimbladder abnormality.
Fish passage through the dam is a major factor to be considered when analyzing the causes of swimbladder deformities. The distension of the swimbladder during fish passage through the dam may rupture its coelomic cavity and muscle layer, thereby forming the deformities of sac-like projections.
Migratory fish are particularly affected by dams (Agostinho et al., 2007). During downstream translocation fish are frequently injured or killed (Cada, 2001) while passing through the turbines or outlet spillways (Larinier & Travade, 2002; Antonio et al., 2007; Pompeu et al., 2009). The sudden pressure drop causes drastic changes in swimbladder volume (Agostinho et al., 2007), often causing them to burst (Therrien & Bourgeois, 2000). Abernethy et al. (2002) evaluating the effect of pressure on physostomous (salmon) and phyisoclistos (Lepomis) fishes, found that regardless of their category, showed distention of wall or rupture of the swimbladder. The sudden pressure changes during dam transposition likely affected the piaparas studied here, accounting for macroscopic and microscopic lesions such as tissue hypertrophy and hyperplasia.
Buoyancy changes can directly affect the swimbladder anatomy of piaparas. The sac-like projection filled the entire coelomic cavity and possibly compressed the channels ending at the urogenital opening. This can affect excretion, gamete release and ultimately the reproductive performance of this species. In conclusion, our findings show the impact of the FHS dam on the piapara population, as evidenced by swimbladder abnormalities. Nevertheless, further studies that evaluate other piapara populations are needed to confirm these results.
Literature Cited
Accepted June 30, 2010
Published September 24, 2010
- Abernethy, C. S., B. G. Amidan & G. F. Cada. 2002. Simulated passage though a modified Kaplan turbine pressure regime: a supplement to "laboratory studies of the effects of pressure and dissolved gas supersaturation on turbine-passed fish". Washington, DC, U. S. Department of Energy, 28p.
- Agostinho, A. A., L. C. Gomes & F. M. Pelicice. 2007. Ecologia e manejo de recursos pesqueiros em reservatórios do Brasil. Maringá, Eduem, 501p.
- Antonio, R. R., A. A. Agostinho, F. M. Pelicice, D. Bailly & J. H. P. Dias. 2007. Blockage of migration routes by dam construction: can migratory fish find alternative routes? Neotropical Ichthyology, 5(2): 177-184.
- Araya, P. R., A. A. Agostinho & J. A. Bechara. 2005. The influence of dam construction on a population of Leporinus obtusidens (Valenciennes, 1847) (Pisces, Anostomidae) in the Yacyretá Reservoir (Argentina). Fisheries Research, 74(1): 198-209.
- Cada, G. F., C. C. Coutant & R. R. Whitney. 1997. Development of Biological Criteria for the Design of Advanced Hydropower Turbines. DOE/ID- 10578, U.S. Department of Energy, Idaho Operations Office, Idaho Falls, ID.
- Cada, F. G. 2001. The development of advanced hydroelectric turbines to improve fish passage survival. Fisheries, 26(1): 14-23.
- Carolsfeld, J., B. Harvey, C. Ross & A. Baer. 2003. Migratory fishes of South America: biology, fisheries and conservation status. World Fisheries Trust, The World Bank, Ottawa, Canada, 380p.
- Gioda, C. R., L. A. Lissner, A. Pretto, J. B. T. Rocha, M. R. C. Schetinger, J. R. Neto, V. M. Morsch & V. L. Loro. 2007. Exposure to sublethal concentrations of Zn (II) and Cu (II) changes biochemical parameters in Leporinus obtusidens. Chemosphere, 69(1): 170-175.
- Glusczak, L., S. D. Miron, M. Crestani, B. M. Fonseca, A. F. Pedron, F. M. Duarte & P. L. V. Vieira. 2006. Effect of glyphosate herbicide on acetylcholinesterase activity, metabolic and hematological parameters in piava (Leporinus obtusidens). Ecotoxicology and Environmental Safety, 65(2): 237-241.
- Haenen, O. L. M., P. Banning van & W. Dekker. 1994. Infection of eel Anguilla anguilla (L.) and smelt Osmerus eperlanus (L.) with Anguillicola crassus (Nematoda, Dracunculoidea) in the Netherlands from 1986 to 1992. Aquaculture, 126(1): 219-229.
- Larinier, M. & F. Travade. 2002. Downstream migration: problem and facilities. Bulletin Francais de la Peche et de la Pisciculture, 364: 181-207.
- Larinier, M. & G. Marmulla. 2004. Fish passes: types, principles and geographical distribution - an overview. Pp. 183-206. In: Welcomme, R. L. & T. Petr (Eds.). Proceedings of the Second International Symposium on the Management of Large Rivers for Fisheries. Volume 2. Food and Agriculture Organization, Bangkok, Thailand.
- OIE. 2008. Office International des Epizooties (OIE). (Online). Available at: http://www.oie.int Accessed January 12, 2010.
- Okada, E. K., A. A. Agostinho & L. C. Gomes. 2005. Spatial and temporal gradients in artisanal fisheries of a large Neotropical reservoir, the Itaipu Reservoir, Brazil. Canadian Journal of Fisheries and Aquatic Sciences, 62(3): 714-724.
- Oldani, N. O., J. Iwaszkiw, O. Padín & A. Otaegui. 1992. Fluctuaciones de la abundancia de peces en el Alto Paraná (Corrientes, Argentina). Publicaciones de la Comisión Administradora del Río Uruguay, Série Técnico-Científica, 1(1): 43-55.
- Oldani, N. O., C. R. M. Baigún, J. M. Nestler & R. A. Goodwin. 2007. Is fish passage technology saving fish resources in the lower La Plata River basin? Neotropical Ichthyology, 5(2): 89-102.
- Pavlov, D. S., A. I. Lupandin & V. V. Kostin. 2002. Downstream migration of fish through dams of hydroelectric Power plants. Oak ridge national laboratory. Available at: http://hydropower.id.doe.gov Accessed January 13, 2010.
- Peruzzi, S., J. I. Westgaard & B. Chatain. 2007. Genetic investigation of swimbladder inflation anomalies in the European sea bass, Dicentrarchus labrax L. Aquaculture, 265(1-4): 102-108.
- Pompeu, P. S., L. P. M. Horta & C. B. Martinez. 2009. Evaluation of the effects of pressure gradients on four brazilian freshwater fish species. Brazilian Archives of Biology and Tecnology, 52(1): 111-118.
- Robertson, G. N., B. W. Lindsey, T. C. Dumbarton, R. P. Croll &F. M. Smith.2008. The contribution of the swimbladder to buoyancy in the adult zebrafish (Danio rerio): A morphometric analysis. Journal of Morphology, 269(1): 666-673.
- Strand, E., C. Jorgensen & G. Huse. 2005. Modelling buoyancy regulation in fishes with swimbladders: bioenergetics and behavior. Ecological Modelling, 185(2-4): 309-327.
- Taitson, P. F., E. Chami & H. P. Godinho. 2008. Gene banking of the neotropical fish Leporinus obtusidens (Valenciennes, 1836): a protocol to freeze its sperm in the field. Journal of Animal Science, 105(3): 283-291.
- Therrien, J. & G. Bourgeois. 2000. Fish Passage at Small Hydro Sites. Report by Genivar Consulting Group for CANMET Energy Technology Centre, Ottawa. Québec City, Québec Canada.
Publication Dates
-
Publication in this collection
04 Nov 2010 -
Date of issue
2010