Abstracts
Cytogenetic analysis of Potamotrygon aff. motoro and P. falkneri indicated the occurrence of an X1X1X2X2/X1X2 Y multiple sex chromosome system in both species, with 2n = 66 chromosomes for females and 2n = 65 chromosomes for males. The nucleolus organizer regions (NORs) identified using Ag-NOR technique showed that both species have multiple Ag-NORs (5 to 7 chromosomes stained). C-banding technique indicated the presence of heterochromatic blocks in the centromeric regions of almost all chromosomes in both species. Through this study there was evidence of heterogeneity in the karyotypes, which suggests that chromosomal rearrangements such as inversions and/or translocations occurred during the chromosomal evolution in two species of this genus.
Cytogenetic; NOR; C-band; Fish; Stingrays
Análises citogenéticas de Potamotrygon aff. motoro e P. falkneri identificaram a ocorrência de um sistema múltiplo de cromossomos sexuais do tipo X1X1X2X2/X1X2Y, em ambas as espécies, com 2n = 66 cromossomos em fêmeas e 2n = 65 cromossomos nos machos. As regiões organizadoras de nucléolos (RONs) identificadas pela reação Ag-RON, evidenciaram marcações múltiplas em ambas as espécies (com variações de 5 a 7 RONs). A técnica de bandamento C, revelou a presença de blocos heterocromáticos localizados nas regiões centromérica em quase todos os cromossomos nas duas espécies em estudo. Através do presente estudo foi evidenciada uma heterogeneidade nos cariótipos, permitindo sugerir que rearranjos cromossômicos, como inversões e/ou translocações, ocorreram durante a evolução cromossômica nas duas espécies desse gênero.
Karyotype description and evidence of multiple sex chromosome system X1X1X2X2/X1X2Y in Potamotrygon aff. motoro and P. falkneri (Chondrichthyes: Potamotrygonidae) in the upper Paraná River basin, Brazil
Vanessa Paes da Cruz; Cristiane Kioko Shimabukuro-Dias; Claudio Oliveira; Fausto Foresti
IDepartamento de Morfologia, Instituto de Biociências, Universidade Estadual Paulista - UNESP, Câmpus de Botucatu, 18618-000 Botucatu, SP, Brazil. fforesti@ibb.unesp.br
ABSTRACT
Cytogenetic analysis of Potamotrygon aff. motoro and P. falkneri indicated the occurrence of an X1X1X2X2/X1X2 Y multiple sex chromosome system in both species, with 2n = 66 chromosomes for females and 2n = 65 chromosomes for males. The nucleolus organizer regions (NORs) identified using Ag-NOR technique showed that both species have multiple Ag-NORs (5 to 7 chromosomes stained). C-banding technique indicated the presence of heterochromatic blocks in the centromeric regions of almost all chromosomes in both species. Through this study there was evidence of heterogeneity in the karyotypes, which suggests that chromosomal rearrangements such as inversions and/or translocations occurred during the chromosomal evolution in two species of this genus.
Key words: Cytogenetic, NOR, C-band, Fish, Stingrays.
RESUMO
Análises citogenéticas de Potamotrygon aff. motoro e P. falkneri identificaram a ocorrência de um sistema múltiplo de cromossomos sexuais do tipo X1X1X2X2/X1X2Y, em ambas as espécies, com 2n = 66 cromossomos em fêmeas e 2n = 65 cromossomos nos machos. As regiões organizadoras de nucléolos (RONs) identificadas pela reação Ag-RON, evidenciaram marcações múltiplas em ambas as espécies (com variações de 5 a 7 RONs). A técnica de bandamento C, revelou a presença de blocos heterocromáticos localizados nas regiões centromérica em quase todos os cromossomos nas duas espécies em estudo. Através do presente estudo foi evidenciada uma heterogeneidade nos cariótipos, permitindo sugerir que rearranjos cromossômicos, como inversões e/ou translocações, ocorreram durante a evolução cromossômica nas duas espécies desse gênero.
Introduction
Although cytogenetic studies in fish have considerably increased lately, the Chondrichthyes remain among the least studied groups (Rocco et al., 2004), with only about 6% of approximately 1,100 currently living species karyotyped (Stingo & Rocco, 2001). Cytogenetic data on marine rays suggest a variation in the diploid number from 2n = 28 for Narcine brasiliensis (Donahue, 1974) to 2n = 104 for Okamejei meerdervoortii (Makino, 1937). The cytogenetic data available in literature for freshwater rays of the family Potamotrygonidae suggest that the species Potamotrygon motoro and P. orbignyi have the same diploid number of 2n = 66 , whereas Paratrygon aiereba presents 2n = 90 (Valentim et al., 2006).
Fish represents an extremely heterogeneous group regarding to sex determination presenting eight systems of sex determination controlled by sex chromosomes (Devlin & Nagahama, 2002). In the Neotropical region species with morphologically distinct chromosomes show a great variety of systems (Oliveira et al., 2009). However, cytogenetic studies with freshwater rays have not shown the occurrence of heteromorphic sex chromosome systems in this group (Valentim et al., 2006).
The present study aimed to investigate the karyotypic structure of P. aff. motoro and P. falkneri, as well as to identify chromosome differences involved in the diversification process of these species, using chromosome banding methods.
Material and Methods
Cytogenetic studies were carried out with Potamotrygon aff. motoro and P. falkneri specimens from the upper Paraná River basin, collected in Porto Rico, Paraná State (22º47'42.4"S 53º20'29.7"W) and Ilha Solteira, São Paulo State (20º47'42.4"S 51º38'29.7"W), Brazil. We collected 30 P. aff. motoro specimens, of which 10 females and 13 males were from Porto Rico and 3 females and 4 males from Ilha Solteira. As regards P. falkneri, we collected 34 specimens, 12 females and 14 males from Porto Rico and 3 females and 5 males from Ilha Solteira. Voucher specimens were deposited at collection of fish the Laboratório de Biologia e Genética de Peixes (LBP 5202 , LBP 5203, LBP 6716, LBP 6717).
Chromosomes were obtained from gill and kidney tissues using the technique described by Foresti et al. (1993). Meiotic metaphases were obtained using the technique described and adapted for fish by Bertollo et al. (1978). Silver staining of the nucleolus organizer regions (NOR) followed the technique of Howell & Black (1980), and C-banding was performed according to Sumner (1972). Chromosome morphology was determined based on arm ratio, as proposed by Levan et al. (1964), and chromosomes were classified into metacentric (m), submetacentric (sm), subtelocentric (st), and acrocentric (a). Fundamental numbers (FN) were calculated considering metacentric, submetacentric, subtelocentric chromosomes as biarmed and acrocentric as uniarmed.
Results
The species Potamotrygon aff. motoro and P. falkneri had 2n = 66 chromosomes in females and 2n = 65 chromosomes in males. This heteromorphic chromosome found, is due to the occurrence of a sex chromosomes system of the type X1X1X2X2/X1X2Y and as also evidenced by the data of meiosis. The two analyzed species showed different fundamental numbers, as shown in Table 1.
Analysis of meiotic preparation of P. falkneri males revealed the presence of a trivalent, besides the 31 bivalents (Fig. 1), reinforcing the hypotheses of occurrence of multiple sex chromosomes.
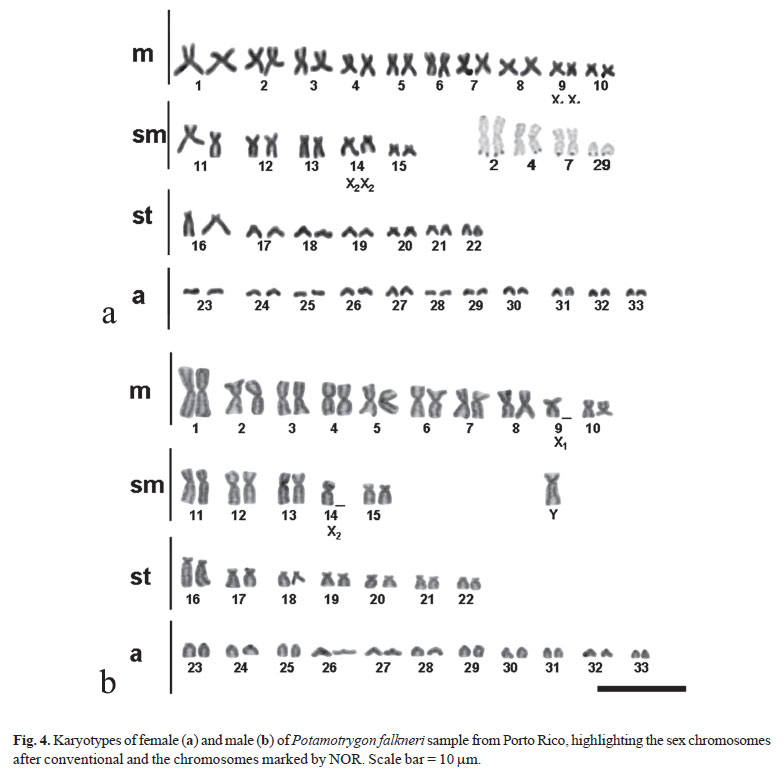
The Ag-NORs technique indicated the presence of multiple Nucleolus Organizer Regions in both species. The samples of P. aff. motoro from Porto Rico have stains in the terminal regions of five pairs of chromosomes; they were identified in the short arm of pair two, in the short arm of pair four, in the long arm of pair six, in the long arm of pair sixteen, and in the pair twenty-six (Fig. 2). The samples from Ilha Solteira have stains in the terminal regions of six chromosomes: in the short arm of pair two, in the long arm of pair six, in the long arm of pair twenty, and in of pair twentysix (Fig. 3). The samples of P. falkneri from Porto Rico have stains in the terminal regions of six metacentric chromosomes and one acrocentric chromosome (Fig. 4). In the sample from Ilha Solteira, stains were also found in the terminal regions of five metacentric and two acrocentric chromosomes (Fig. 5). C-banding pattern indicated that P. aff. motoro and P. falkneri (Fig. 6) have large C-band positive segments located in the centromeric region of most chromosomes.
Discussion
Although many vertebrate species have sex chromosomes morphologically different between males and females, most fish species show no chromosomal difference between sexes; thus, there is great difficulty in identifying sex using karyotypic analysis.
In this study, all samples of Potamotrygon aff. motoro and P. falkneri have 2n = 66 chromosomes for females and 2n = 65 chromosomes for males. Such difference in the chromosome number is due to a multiple system of sex determination of the type X1X1X2X2/X1X2Y.
Both species have similar distribution of C-band positive segments in the centromeric regions of almost all chromosomes, as already evidenced by Valentim et al. (2006) for Paratrygon aiereba, Potamotrygon motoro, and P. orbigny.
In some fish species such as Pseudotocinclus tietensis (Andreata et al., 1992), Microlepidogaster leucofrenatus (Andreata et al., 1993), and Characidium cf. fasciatum (Maistro et al., 1998) sex chromosomes are easily identified using techniques like C-banding. However, in this study, the chromosomes involved in sex determination could not be identified through the constitutive heterochromatin patterns obtained for P. aff. motoro and P. falkneri since in their sex chromosomes positive C-band segments are restricted to the centromeric regions.
The analyzed P. aff. motoro samples have several karyotypic formulae and fundamental numbers, but the sample from Ilha Solteira had a karyotypic formula different from that of the remaining samples due to the presence of different numbers of acrocentric and subtelocentric chromosomes. These variations in the fundamental number may be the result of chromosomal rearrangements such as pericentric inversions since the diploid number is conserved. This kind of rearrangement has been pointed as very important in fish chromosome differentiation (Oliveira et al., 2009).
Potamotrygon aff. motoro and P. falkneri had eight to ten chromosomes stained with silver nitrate (Ag-NORs) always in terminal regions of similar size chromosomes. Multiple NORs were also observed in other rays (Valentim et al., 2006) and are very common in fishes (Oliveira et al., 2007). The differences in NOR sites found in these samples could be explained by Robertsonian rearrangements, resulting in dispersion and/or loss of ribosomal genes.
The occurrence of ribosomal DNA in sex chromosomes has been observed in several groups of organisms. Cistrons of 5.8 S, 18S and 28S ribosomal genes, which form the NORs in animals and are also stained with silver nitrate, were found in sex chromosomes of insects, such as Drosophila (Parise-Maltempi & Avancini, 2001), beetles (Juan et al., 1993), mammals (Yonenaga-Yassuda et al., 1983; Oshida et al., 1999), and plants (Nakayama et al., 2001). In fish, reports of NORs in sex chromosomes are restricted to Fundulus diaphanus (Howell & Black, 1979), Salvelinus alpinus (Reed & Phillips, 1995), Triportheus guentheri (Artoni & Bertollo 1999), Hoplias malabaricus (Born & Bertollo, 2000) and Hisonotus sp. A (Andreata, 2002).
The formation of a multiple sex chromosome system in fish is usually due to a preexisting simple system in a particular group, such as ZZ/Z0, XX/X0, ZZ/ZW, or XX/XY. In the studied ray species, the found X1X1X2X2/X1X2Y system could have arisen from reciprocal translocations or from an ancestral system with a simple such as XY where the Y chromosomes fused with an autossomal chromosome giving origin to new Y and X2 chromosomes, and the old X chromosome is identified as X1 chromosome. In some fish species, the origin of Y is clear since it has the same characteristics of chromosomes (X1) and (X2), such as in Eigenmannia species (Almeida-Toledo et al., 1984, 2000).
Valentim (2001) reported a possible sex chromosome system of the type XX/X0 in Potamotrygon sp. from the middle rio Negro, Amazonas State, where the female had 2n = 68 chromosomes and the male 2n = 67 chromosomes due to a putative absence of a homologous pair 2 (metacentric) in the male. Different mechanisms of sex determination in phylogenetically related groups are common in Neotropical fish, as evidenced in several groups such as Gymnotiformes (Almeida-Toledo et al., 1984, 2000, 2001) and Loricariidae (Andreata et al., 1992, 1993), in which different sex chromosome systems were identified among its representatives.
This study indicated that there is great uniformity concerning the diploid number in the studied species. However, variations in the number of chromosome arms suggest that extensive chromosomal rearrangements such as pericentric inversions occurred during the chromosomal evolution in the two analyzed species. If, on the one hand, the uniformity in diploid numbers and mechanisms of chromosome heteromorphisms related to sex showed a closer relationship between what is shown for P. aff. motoro and P. falkneri, it can be assumed that the occurrence of such chromosomal polymorphism may reflect the existence of not only two freshwater ray species in this environment, but also a complex group of species that need to be further studied. A consistent understanding of the evolutionary relationships between variable karyotypes and the mechanisms of P. aff motoro and P. falkneri sex chromosomes will depend on new cytogenetic and molecular information from other species and samples of freshwater rays.
Acknowledgements
The authors are grateful to Renato Devidé properly for technical assistance, to Fernando F. Mendonça and Jefferson M. Henriques for the review, financial support granted by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Brazil and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) Brazil.
Literature Cited
Accepted December 14, 2010
Published March 31, 2011
- Almeida-Toledo, L. F., F. Foresti & S. A. Toledo-Filho. 1984. Complex sex chromosome system in Eigenmannia sp. (Pisces, Gymnotiformes). Genetica, 64: 165-169.
- Almeida-Toledo, L. F., F. Foresti & S. A. Toledo-Filho. 2000. Karyotipic evolution in Neotropical freshwater fish. In Chromosome Today, Switzerland: Birkhauer Verlag, 13: 169-182.
- Almeida-Toledo, L. F. & F. Foresti. 2001. Morphologically differentiated sex chromosomes in Neotropical freshwater fish. Genetica, 111: 1-3.
- Almeida-Toledo, L. F., F. Foresti, E. V. Péquignot & M. F. Z. Daniel-Silva. 2001. XX:XY sex chromosome system with X heterocromatinization: an early stage of sex chromosome differentiation in the Neotropic electric eel Eigenmannia virescens. Cytogenetics and Cell Genetics, 95: 73-78.
- Andreata, A. A. 2002. Estudos citogenéticos no gênero Microlepidogaster (Pisces, Loricariidae, Hypoptopomatinae). Unpublished Ph.D. Dissertation. Instituto de Biociências, Universidade de Estadual Paulista, UNESP, Botucatu, SP, 196p.
- Andreata, A. A., L. F. Almeida-Toledo & C. Oliveira. 1992. Chromosome studies in Hypoptopomatinae (Pisces, Siluriformes, Loricariidae): XX/XY sex chromosome heteromorphism in Pseudotocinclus tietensis Cytologia, 57: 369-372.
- Andreata, A. A., L. F. Almeida-Toledo & C. Oliveira. 1993. Chromosome studies in Hypoptomatinae (Pisces, Siluriformes, Loricariidae). II. ZZ/ZW sex-chromosome system, B chromosomes, and constitutive heterochromatin differentiation in Microlepdogaster leucofrenatus Cytogenetics and Cell Genetics, 63: 215-220.
- Artoni, R. F. & L. A. C. Bertollo. 1999. Nature and distribution of constitutive heterochromatin in fishes, genus Hypostomus (Loricariidae). Genetica, 106: 209-214.
- Bertollo, L. A. C., C. S. Takahashi & O. Moreira-Filho. 1978. Cytotaxonomic consideration on Hoplias lacerdae (Pisces, Erythrinidae). Revista Brasileira de Genética, 1: 103-120.
- Born, G. G. & L. A. C. Bertollo. 2000. An XX/XY sex chromosome system in a fish species, Hoplias malabaricus, with a polymorphic NOR-bearing X chromosome. Chromosome Research, 8: 111-118.
- Devlin, R. H. & Y. Nagahama. 2002. Sex determination and sex differentiation in fish: an overview of genetic, physiological, and environmental influences, Aquaculture, 208: 191-364.
- Donahue, W. H. 1974. A karyotypic study of three species of Rajiformes (Chondrichthyes, Pisces). Canadian Journal of Genetics Cytology, 16: 203-211.
- Foresti, F., C. Oliveira & L. F. Almeida-Toledo. 1993. A method for chromosome preparations from large specimens of fishes using in vitro short treatment with colchicine. Experientia, 49: 810-813.
- Howell, W. M. & D. A. Black. 1979. Location of the nucleolus organizer regions on the sex chromosomes of the banded killifish, Fundulus diaphanus Copeia, 3: 544-546.
- Howell, W. M. & D. A. Black. 1980. Controlled silver-staining of nucleolus organizer regions with a protective colloidal developer: a 1-step method. Experientia, 36: 1014-1015.
- Juan, C., J. Pons & E. Petitpierre. 1993. Localization of tandemly repeated DNA sequences in beetle chromosomes by fluorescence in situ hybridization. Chromosome Research, 1: 167-174.
- Levan, A., K. Fredga & A. A. Sandbreg. 1964. Nomeclature for centromeric position on chromosomes. Hereditas, 52: 201-220.
- Maistro, E. L., E. P. Mata, C. Oliveira & F. Foresti. 1998. Unusual occurrence of a ZZ/ZW sex-chromosome system and supernumerary chromosomes in Characidium cf. fasciatum (Pisces, Characiformes, Characidiinae). Genetica, 104: 1-7.
- Makino, S. 1937. The chromosomes of two elasmobranch fishes. Cytologia, 2: 867-876.
- Nakayama, S., M. Fujishita, T. Sone & K. Ohyama. 2001. Additional locus of rDNA sequence specific to the X chromosome of the liverwort, Marchantia polymorpha Chromosome Research, 9: 469-473.
- Oliveira, C., F. Foresti & L. F. Almeida-Toledo. 2007. Karyotypic evolution in Neotropical fishes. Pp. 111-164. In: Fish Cytogenetics. Pisano, E., C. Ozouf-Costaz, F. Foresti & B. G. Kapoor (Eds.). Enfield, Science Publisher, 502p.
- Oliveira, C., F. Foresti & A. W. S. Hilsdorf. 2009. Genetics of neotropical fish: from chromosomes to populations. Fish Physiology and Biochemistry, 35: 81-100.
- Oshida, T., J. Shindo & M. C. Yoshida. 1999. NOR-bearing Y chromosome in american beaver, Castor canadensis (Rodentia, Castoridae). Chromosome Science, 3: 117-118.
- Parise-Maltempi, P. P. & R. M. P. Avancini. 2001. C-banding and FISH in chromosomes of the blow flies Chrysomya megacephala and Chrysomya putoria (Diptera, Calliphoridae). Memórias do Instituto Oswaldo Cruz, 96: 371-377.
- Reed, K. M. & R. B. Phillips. 1995. Molecular cytogenetic analysis of the double-CMA3 chromosome of lake trout, Salvelinus namaycush Cytogenetics and Cell Genetics, 70: 104-107.
- Rocco, L., D. Costagliola, M. Fiorillo, F. Tinti & V. Stingo. 2004. Molecular and chromosomal analysis of ribosomal cistrons in two cartilaginous fish, Taenura lymma and Raja montagui (Chondrichthyes, Batoidea). Genetica, 123: 245-253.
- Stingo, V. & L. Rocco. 2001. Selachian cytogenetics: a review. Genetica, 111: 329-347.
- Sumner, A. T. 1972. A simple technique for demonstrating centromeric heterochromatin. Experimental Cell Research, 75: 304-306.
- Valentim, F. C. S. 2001. Caracterização cromossômica em espécies de peixes da família Potamotrygonidae (Chondrichthyes, Rajiformes) do médio rio Negro. Unpublished Ph.D. Dissertation. Universidade Federal de São Calos, São Carlos, 77p.
- Valentim, F. C. S., J. N. Falcão, J. I. R. Porto & E. Feldberg. 2006. Chromosomes of three freshwater stingrays (Rajiformes, Potamotrygonidae) from the Rio Negro basin, Amazon, Brazil. Genetica, 128: 33-39.
- Yonenaga-Yassuda, Y., M. F. L. Assis, S. Kasahara, M. L. L'abbate & M. J. Souza. 1983. Nucleolar organizer regions in Akodon arviculoides (Cricetidae, Rodentia): evidence for the activity of rDNA genes in both X chromosomes of females. Cytogenetics and Cell Genetics, 35: 143-147.
Publication Dates
-
Publication in this collection
24 May 2011 -
Date of issue
Mar 2011
History
-
Accepted
31 Mar 2011 -
Received
14 Dec 2010