Abstracts
Reproductive seasonality in tropical freshwater fishes is strongly influenced by rainfall. In lowlands, floods spill laterally to floodplains and fishes usually breed during the flooding season. In mountain rivers, floods are sudden and flush out aquatic organisms. Fishes in mountain rivers usually breed during dry seasons, what has been hypothesized as a strategy to reduce mortality due to strong floods. If that is the case, mouth-brooding fishes should suffer less from strong floods and should have more prolonged breeding seasons in mountain rivers. Here we investigated the breeding activity of a mouth-brooding cichlid (Geophagus steindachneri) in a mountain river in Colombia using three kinds of evidence: monthly variation of gonad weight, macroscopic and histological observations of the gonads, and occurrence of mouth-brooding females. Analysis was made on adults captured monthly throughout a year. The results indicate that G. steindachneri breeds during the dry season in the mountain river studied. Female mouth brooding was related with a halt in the maturation of their ovaries. Other factors than the flushing-out effect of floods on offspring may be determining dry-season breeding of fishes in tropical mountain rivers.
Limnology; Parental care; Phenology; Rainfall; Spawning
La estacionalidad reproductiva de los peces tropicales de agua dulce esta influida por los patrones de lluvia. En tierras bajas, las aguas se expanden hacia el plano de inundación y los peces generalmente se reproducen en aguas altas. En ríos de montaña las inundaciones son repentinas y arrastran a los organismos acuáticos. Los peces de ríos de montaña usualmente muestran que se reproducen durante las temporadas secas, posiblemente como estrategia para reducir la mortalidad producto de las inundaciones repentinas. Si esta hipótesis es cierta, los peces que realizan incubación bucal estarían menos afectados por las inundaciones repentinas y tendrían una temporada reproductiva más prolongada en los ríos de montaña. En este trabajos estudiamos la temporada reproductiva de un cíclido con incubación bucal (Geophagus steindachneri) en un río de montaña en Colombia, usando tres tipos de evidencia: la variación mensual del peso gonadal, observaciones macroscópicas e histológicas de las gónadas y la frecuencia de las hembras incubando. Los análisis se realizaron en adultos capturados mensualmente durante un año. Nuestros resultados indican que la temporada reproductiva de G. steindachneri en este río de montaña ocurre en la época seca. Se observó una pausa en el desarrollo ovárico durante la incubación bucal. Otros factores, diferentes al arrastre producido por las inundaciones, deben estar determinando la reproducción durante la temporada seca de los peces en los ríos de montaña.
Introduction
Periodic changes in environmental conditions determine phenology of species (Chapman et al., 2005Chapman, C. A., L. J. Chapman, T. T. Struhsaker, A. E. Zanne, C. J. Clark & J. R. Poulsen. 2005. A long-term evaluation of fruiting phenology: importance of climate change. Journal of Tropical Ecology, 21: 31-45.), which is mostly influenced by day length and temperature in higher latitudes and by rainfall and biotic factors in the tropics (Bronson, 2009Bronson, F. H. 2009. Climate change and seasonal reproduction in mammals. Philosophical Transactions of the Royal Society of London B, Biological Sciences, 364: 3331-3340.; Payne, 1986Payne, A. I. 1986. The ecology of tropical lakes and rivers. Chichester, J. Wiley. 301p.; Morales-Nin & Panfili, 2005Morales-Nin, B. & J. Panfili. 2005. Seasonality in the deep sea and tropics revisited: what can otoliths tell us? Marine and Freshwater Research, 56: 585-598.). In fishes, rainfall determines hydrological patterns that condition reproductive activity (Lowe-McConnell, 1987Lowe-McConnell, R. H. 1969. The cichlid fishes of Guyana, South America, with notes on their ecology and breeding behaviour. Zoological Journal of the Linnean Society, 48: 255-302.). Lowland floodplain rivers have large watersheds with low slopes and banks. These rivers flood gradually their lateral plains, making terrestrial resources on these plains available for aquatic organisms (Welcomme, 1979Welcomme, R. 1979. The fisheries ecology of floodplain rivers. London.; Lowe-McConnell, 1987). Inversely, mountain rivers, those with ≥0.002 m/m of slope (Wohl & Merritt, 2008Wohl, E. & D. M. Merritt. 2008. Reach-scale channel geometry of mountain streams. Geomorphology, 93: 168-185.), usually have smaller and steeper watersheds with banks that confine water, resulting in sudden and quick floods (Chapman & Kramer, 1991Kramer, D. L. 1978. Reproductive seasonality in the fishes of a tropical stream. Ecology, 59: 976-985.). Such flash-floods have a flushing-out effect that decimates fish populations and their food (Chapman & Kramer, 1991; Grether et al., 2001Grether, G. F., D. F. Millie, M. J. Bryant, D. N. Reznick & W. Mayea. 2001. Rain forest canopy cover, resource availability, and life history evolution in guppies. Ecology, 82: 1546-1559.). These contrasting flow patterns between floodplain and mountain rivers determine the patterns of breeding of fishes from each type of river. Fishes from floodplain rivers usually breed during the rainy season (Kramer, 1978; Lowe-McConnell, 1987; Munro, 1990Munro, A. D. 1990. Tropical freshwater fish. Pp. 145-239. In: Munro, A. D., A. P. Scott & T. J. Lam. (Eds.). Reproductive seasonality in Teleosts: environmental influences. Boca Raton, CRC.; Menezes & Vazzoler, 1992Menezes, N. A. & A. E. A. de M. Vazzoler. 1992. Reproductive characteristics of characiformes. Pp. 60-70. In: Hamlett, W. C. (Ed.). Reproductive biology of South American vertebrates. New York, Springer., Alkins-koo, 2000Alkins-koo, M. 2000. Reproductive timing of fishes in a tropical intermittent stream. Environmental Biology of Fishes, 57: 49-66.; Lewis Jr. et al., 2006Lewis Jr., W. M., S. K. Hamilton & J. F. Saunders III. 2006. Rivers of Northern South America. Pp: 219-256. In: Cushing, C. E., K. V. Cummins & G. W. Minshall (Eds.). River and stream ecosystems of the world. University of California Berkeley Press.), whereas mountain river fishes tend to do so during the dry season (Sazima, 1980Sazima, I. 1980. Behavior of two Brazilian species of parodontid fishes, Apareiodon piracicabae and A. ibitiensis. Copeia, 1: 166-169.; Flecker et al., 1991Flecker, A. S., D. C. Taphorn, J. A. Lovel & B. P. Feifarek. 1991. Drift of characin larvae, Bryconamericus deuterodonoides, during the dry season from Andean Piedmont streams. Environmental Biology of Fishes, 31: 197-202.; Harikumar et al., 1994Harikumar, S., K. G. Padmanabhan, P. A. John & K. Kortmulder. 1994. Dry-season spawning in a cyprinid fish of southern India. Environmental Biology of Fishes, 39: 129-136.; Wang et al., 1995Wang, J., M. Liu & L. Fang. 1995. The reproductive biology of an endemic cyprinid, Zacco pachycephalus in Taiwan. Environmental Biology of Fishes, 43: 135-143.; Pusey et al., 2002Pusey, B. J., A. H. Arthington, P. G. Close & J. R. Bird. 2002. Larval fishes in rainforest streams: recruitment and microhabitat use. Proceedings of the Royal Society of Queensland, 110: 27-46.; Román-Valencia & Botero, 2006; Torres-Mejica & Ramírez-Pinilla, 2008Torres-Mejica, M. & M. P. Ramírez-Pinilla. 2008. Dry-season breeding of a characin in a neotropical mountain river. Copeia, 1: 99-104.). However, these patterns are based on a biased sampling because most studies on the reproductive activity of Neotropical cichlids have investigated species living in floodplain rivers (Lowe-McConnell, 1969; Munro, 1990; Señaris & Lasso-A., 1993; Cala et al., 1996Cala, P., E. Gonzáles & M. P. Varona. 1996. Aspectos biológicos y taxonoomicos del tucunare, Cichla monoculus (Pisces: Cichlidae). Dahlia, 1: 23-37.; Gonzalez et al., 1996; Crampton, 2008Crampton, W. G. R. 2008. Ecology and life history of an Amazon floodplain cichlid: the discus fish Symphysodon (Perciformes: Cichlidae). Neotropical Ichthyology, 6: 599-612.; Rossoni et al., 2010Rossoni, F., S. Amadio, E. Ferreira, & J. Zuanon. 2010. Reproductive and population parameters of discus fish Symphysodon aequifasciatus Pellegrin, 1904 (Perciformes: Cichlidae) from Piagaçu-Purus Sustainable Development Reserve (RDS-PP), lower Purus River, Amazonas, Brazil. Neotropical Ichthyology, 8: 379-383.). We provide in this study the first analysis on the reproductive activity of any mountain cichlid.
Dry-season breeding in mountain rivers may be explained as a result of the favorable conditions for newborn offspring during that season, which has been coined as the "low-flow recruitment hypothesis" (Humphries et al., 1999Humphries, P., A. J. King & J. D. Koehn. 1999. Fish , flows and flood plains: links between freshwater fishes and their environment in the Murray-Darling River system, Australia. Environmental Biology of Fishes, 56: 129-151.). This hypothesis states that dry-season breeding would be favorable for fishes because of the absence of floods that flush poorly swimming juveniles and their food, small-size plankton. If dry-season breeding is a mechanism for avoiding catastrophic flushing-down of juveniles during floods, fishes that carry their offspring, such as mouth-brooding cichlids, should show less constrained reproductive seasons in mountain rivers. Here we tested whether Geophagus steindachneri Eigenmann & Hildebrand, 1922, a cichlid that performs mouth brooding, shows dry-season breeding in a tropical mountain river.
Geophagus steindachneri is distributed in the Atlantic drainages from río Sinú in Colombia to Lago Maracaibo basin in Venezuela (Kullander, 2003Kullander, S. O. 2003. Family Cichlidae (Cichlids). Pp. 605-654. In: Reis, R. E., S. O. Kullander & C. J. Ferraris, Jr. (Orgs.). Check list of the freshwater fishes of South and Central America. Porto Alegre, Edipucrs.). Adults of this species show sexual dimorphism, males have more colored fins and a red post-occipital cephalic hump. This species shows mouth brooding (Wimberger, 1991Wimberger, P. H. 1991. Plasticity of jaw and skull morphology in the Neotropical cichlids Geophagus brasiliensis and G. steindachneri. Evolution, 45: 1546-1563.), a characteristic of other Geophagines and other cichlids (Perrone & Zaret, 1979Perrone, M., Jr. & T. M. Zaret. 1979. Parental care patterns of fishes. The American Naturalist, 113: 351-361.; López-Fernández et al., 2012). Mating occurs as follows (Wimberger, 1991; pers. obs.). A male guards a nest in the streambed substrate. A reproductive female reaches the male nest and, after courtship displays, the female spawns on the streambed. After each pass of spawning, the female picks up her eggs with her mouth. Once all eggs are spawn, the female reaches the male gonopore, stimulates it with her mouth, and obtains sperm to fertilize the eggs intrabucally. For two to three weeks, females starve while eggs hatch and embryos develop. After that period, embryos start taking short trips outside mother's mouths to feed on exogenous sources and mothers resume eating as well. Interestingly, it is unknown what is occurring to the ovary development and energy reserve of cichlid females while they are breeding their offspring in their mouths because most studies focus on one reproductive aspect, not multiple aspects in conjunction (but see Esmaeili et al., 2010Esmaeili, H. R., Z. Ganjali & M. Monsefi. 2010. Gonad morphology and histology of the endemic Hormuz cichlid, Iranocichla hormuzensis Coad, 1982 from Mehran River, Southern Iran. IUFS Journal of Biology, 69: 1-12.). Here we investigated the breeding cycle of G. steindachneri using multiple sources of evidence, including gonadal, liver, and fat weights, macro- and microscopic observations of gonads, and observations of offspring in mouth-brooding females.
To summarize, our main objective was to determine the annual breeding cycle of a population of G. steindachneri in a mountain river. We analyzed the seasonality of different interrelated variables such as body, liver, fat, and gonadal weights. We also observed gonads micro- and macroscopically, and recorded embryo development in brooding females. With this information, we inspected the relationship between the occurrence of mouth brooding and body, liver, fat, and gonadal weights. Our results are the first on the reproduction of this species, the first report on the breeding cycle of a mountain cichlid, and one of the handful to study the correlation of mouth brooding and reproductive aspects in a cichlid.
Material and Methods
Study area and sampling. Samples were collected in río Fonce (río Magdalena system) in the department of Santander, Colombia (N 6º 25'-31', W 73º 7'- 9'), between 1,100 and 1,350 m of altitude. In this area, the western slope of the Eastern Cordillera of Colombian Andes, the rainfall pattern shows two rainy seasons (April-May and October-November), and two dry seasons (June-September and December-March, Fig. 1a). Monthly samplings were performed from December 2003 to November 2004, using cast nets, seines, and dip nets. On each month, a minimum of 25 specimens was collected, individually separated in zipper bags, immersed in an overdose of sedative (Lidocaine), and fixed by injecting them with Bouin's solution into their body cavities and by keeping them in buffered formalin (10%) for a month. Females of this species rarely abandon brooding offspring as suggested by the observations that only one female expulsed its offspring during capture and none of the females showed a reduced number of fry for its size. In any case, specimens were preserved in individual plastic bags to prevent embryo loss. Water temperature and pH were measured with a pH-meter (Schott Handylab) and conductivity with a conductivity-meter (Hanna Instruments HI 8033). Water transparency was determined using a secchi disk.
Monthly variation of environmental variables: (a) rainfall pattern, (b) pH and temperature, and (c) conductivity and transparency.
Laboratory procedures. All specimens were deposited in the Colección Ictiológica, Universidad Industrial de Santander (UIS-T, 500 and 1526). One month after fixation, mouths of all specimens were inspected for embryos, specimens were transferred to 70% ethanol and embryos were counted and preserved separately in 5% formalin. Stage of offspring development was classified in three periods, following Balon (1975)Balon, E. K. 1975. Reproductive guilds of fishes: a proposal and definition. Journal of the Fisheries Research Board of Canada, 32: 821-864.. The embryonic period ranges from egg fertilization to complete hatching, the larval period ends when nutrition is fully exogenous (yolk sac is fully reabsorbed), and the juvenile period is characterized by fully differentiated fins (what allows juveniles to swim and forage) and ends with gonadal maturation. We measured standard length (SL, ±0.02 mm) and the weight of eviscerated body, liver, mesenteric fat, and gonads (±0.001 g). Macroscopic observations were done under a stereo-microscope. Microscopic observation of gonads was performed on histological preparations stained with hematoxylin-eosin (Luna, 1968Luna, L. G. (Ed.) 1968. Manual of histologic staining methods of the Armed Forces Institute of Pathology. 3rd ed. New York, McGraw Hill, 258p.). Some samples were also stained with PAS-Alcian Blue 2.5% (Luna, 1968) to distinguish yolk vesicles, which are indicators of the onset of vitellogenesis (Selman et al., 1986Selman, K., R. A. Wallace & V. Barr. 1986. Oogenesis in Fundulus heteroclitus IV. Yolk-vesicle formation. Journal of Experimental Zoology Part A, 239: 277-288.).
Female maturity was classified following the scale of Smith & Walker (2004)Smith, B. B. & K. F. Walker. 2004. Spawning dynamics of common carp in the River Murray , South Australia, shown by macroscopic and histological staging of gonads. Journal of Fish Biology, 64: 336-354., with a modification to adapt the microscopic terminology of Coward & Bromage (1998)Coward, K. & N. R. Bromage. 1998. Histological classification of oocyte growth and the dynamics of ovarian recrudescence in Tilapia zillii. Journal of Fish Biology, 53: 285-302.. Immature females have thin ovaries and small and tightly packed oocytes at early perinuclear or previtelogenic stages. Mature females have ovaries that are opaque and granular, with small yellow oocytes that are most frequently vitellogenic (with small yolk granules at oocyte periphery). Ripe females show ovaries distinctly bulging and lobular with large yellow opaque oocytes that are preovulatory follicles (with yolk granules accumulating into large yolk globules). Spent females have translucent, flaccid, and bloodshot ovaries with scattered residual mature oocytes and abundant post-ovulatory follicles. Regressing females have flaccid ovaries that show abundant post-ovulatory and atretic oocytes, the later looking small, yellow, and opaque. The type of spawning (i.e. the synchrony in egg maturation, ranging from continuous to multiple sequential batches) was determined from the relative frequency distribution of oocyte diameters (Vazzoler, 1996). These diameters were measured using an ocular micrometer. Batch fecundity, the number of pre-ovulatory follicles in ripe ovaries was quantified from macroscopic observations of dissected ovaries. Spawning season was determined using three kinds of evidence: the monthly variation in ovarian mass, the monthly frequencies of stages of ovary maturity, and the monthly presence of brooding females.
Male maturity was classified using histological observations of testes, following the testis classification with three stages of Smith & Walker (2004). Immature males have testes with small and distinct lobules, formed by peripheral cysts in spermatogenesis, and lumens lacking sperm and relatively small in proportion to cysts. Mature males have testes with large lobules with relatively large lumens filled with sperm and surrounded by thin interstitial tissue. Spent males have testes with thick interstitial tissue and reduced lumens with scarce or no sperm.
Data analysis. Monthly variation in standard length (SL) was determined with ANOVAs for each sex. Monthly variation in weights of eviscerated body, liver, mesenteric fat, and gonads was determined with ANCOVAs for each dependent variable and sex, with log-transformed SL as the covariate. In these linear models, the dependent variables were log-transformed and the independent variable was the month of capture. For males, in order to increase group size in months with low sample size, monthly samples were merged in two groups (December-January and February-March). Assumptions of normality, linearity, homocedasticity, homogeneity of slopes, and absence of outliers were satisfied for all analyses. Immature specimens and females smaller than the size of the smallest brooding female were excluded from analyses. The relationship between mouth brooding and energy storage (fat and liver weights), body condition (WE), and reproduction (gonadal weight) was estimated using separate ANCOVAS for each dependent variable. In these ANCOVAS, the dependent variables and the covariate were log-transformed and the independent variable was the stage of maternal care (grouped in five categories: mature females, ripe females without embryos, and females with embryos, larvae, or juveniles). To visualize results of ANCOVAs, the size-independent variation in dependent variables was shown in figures as the residuals of the allometric regressions. The departure of a 1:1 sex-ratio was tested with a goodness-of-fit test. Monthly differences in the frequencies of the operational sex ratio, ovarian maturity stages, and maternal care stages were tested with G-tests on the respective contingency tables (Zar, 1999Zar, J. H. 1999. Biostatistical analysis. 4th ed. Upper Saddle River, NJ, Prentice Hall.). All statistical analyses were performed using R (R Development Core Team, 2014R Development Core Team. 2014. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna.).
Results
Physical and chemical characteristics of the river during sampling months are shown in Fig. 1. These characteristics varied with rainfall, showing bimodal patterns where river during dry months showed higher pH, temperature, conductivity, and transparency values than during rainy months. A total of 374 adult specimens were collected. There was about twice as many females as males (239:135, χ 2 = 28.366, df = 1, p< 0.0001). Sex ratio varied along the year (G0.05, 11= 31.144, p=0.001), with proportionally less males collected during the main dry season (Dec-Mar).
Females. The minimum size of a mouth-brooding female (minimum size at maturity) was 66.88 mm of SL. Female SL was highest in the main dry season (Dec-Mar) and decreased throughout the year (Fig. 2a, Table 1). Female SL was significantly related with body, liver, fat, and ovarian weights (Table 1), supporting the use of SL as a covariate in ANCOVA analyses.
Monthly variation in adult females: (a) SL, standard length and WE, eviscerated weight, (b) liver and fat weight, and (c) ovary weight. Barplot behind the lines represents the rainfall pattern.
Analysis of variance and covariance for monthly variation of log-transformed standard length (SL), eviscerated weight (WE), liver, fat, and gonad weights of females and males. Effect size measured as the partial η2.
Female WE had its highest values in the middle of the main dry season (Jan) and decreased to its lowest values the following month, remaining constant in later months (Fig. 2a, Table 1). Liver weight decreased during the dry season, falling to its lowest values in February and gradually increased through the year. The pattern of fat weight variation followed closely that of liver weight; its lowest values were observed during the dry season and progressively increased along the year (Fig. 2b, Table 1). Ovarian weight showed a bi-seasonal pattern, with highest values observed at the beginning of each dry season (Fig. 2c, Table 1).
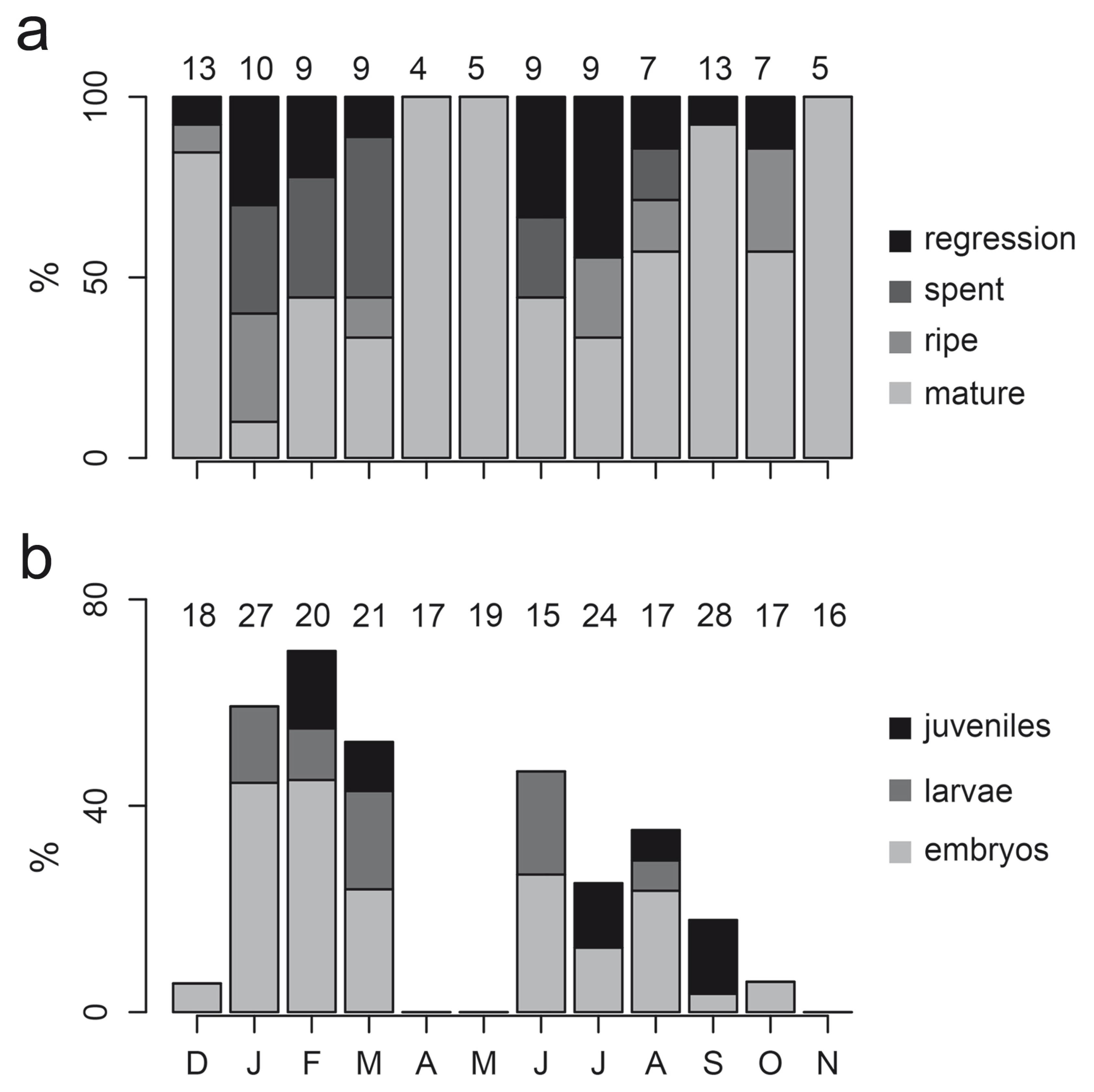
Histological and mouth-brooding analysis also showed a bimodal pattern of female reproduction. The frequencies of ovarian histological stages varied among months (G0.05,33=811.753, p< 0.001). Mature females were observed all year long, but ripe, spent, and regressing females were observed more frequent during dry season months (Fig. 3a). Oocytes matured synchronously as a single batch. The largest oocytes were preovulatory and had a maximum of 1.9 mm of diameter. There was a positive relationship between SL and batch fecundity (y=5.391x-312.8, n=21, r2=0.544, p<0.001). The average batch fecundity was 210 eggs, and fecundity ranges from 110 to 456 eggs in females of 78.96 mm and 122 mm SL, respectively. Brooding females were more frequent during dry season months and less frequent or absent during rainy months (G0.05,11=82.183, p<0.001; Fig. 3b). Females carrying embryos and larvae were more frequent at the beginning of each dry season; later in the dry seasons they were more frequently carrying juveniles (Fig. 3b).
Monthly variation in (a) the frequency of ovarian development stages and (b) the frequency of mouthbrooding females carrying embryos, larvae, or juveniles.
The contrast of stage of embryo development and the stage of ovary maturity, in conjunction with the decrease of liver and gonad weights in brooding females, evidenced a halt in ovarian development early in the mouth-brooding process (Fig. 4). Ovaries of brooding females never were ripe (Fig. 4a). Ovaries of females with embryos or larvae were mostly spent or in regression (Fig. 4a). Moreover, females with larvae in their mouths never showed mature ovaries (i.e., with preovulatory oocytes); mature ovaries were seen again in females carrying juveniles (Fig. 4a). Females with embryos at different stages of development did not vary in body size (log-SL: F3,235=1.921, p=0.127), body condition (log-WE, F3,234=0.792, p=0.499), or fat weight (F3,234=2.006, p=0.114). In contrast, liver (F3,234=16.408, p<0.001) and gonad weights (F3,234=9.374, p<0.001) were lower in females with offspring in their mouths (Fig. 4b).
Differences between females that were not performing parental care and mouth-brooding females: (a) stage of ovary maturity, with circle size indicating number of females in each condition; (b) liver and ovary weight.
Males. The minimum size of a male with developed testes was 43.8 mm SL. Males varied in SL throughout the year, showing a peak in size at the end of the main dry season (Feb-Mar), and the lowest sizes right afterwards (Fig. 5a, Table 1). SL was related with liver, fat, testis, and eviscerated body weights, supporting the use of ANCOVA models with SL as a covariate (Table 1). There was monthly variation in body, liver, fat, and testis weights (Table 1, Fig. 5). WE showed a bimodal pattern, with two peaks during dry months (Dec-Jan and Aug), and two valleys, one in the dry month of June and another in the rainy month of October (Fig. 5a). As in females, liver and fat weights showed very close patterns, increasing from low values in the main dry season (Dec-Mar) to highest values in the rainiest months later in the year (Fig. 5b). Testis weights showed a peak in Dec-Jan, the beginning of the main dry season, decreasing abruptly in Feb-Mar, and slowly increasing the later months (Fig. 5c). The testes showed a histological arrangement of the unrestricted spermatogonial testis type. All mature males had sperm in their lobular lumens, evidencing that spermatogenesis occurred continuously throughout the year.
Monthly variation in adult males: (a) SL, standard length and WE, eviscerated weight, (b) liver and fat weight, and (c) testis weight. Barplot behind the lines represents the rainfall pattern.
Discussion
Four pieces of evidence indicate that females of G. steindachneri breed during the dry season in río Fonce. A bimodal peak was observed in the variation of ovary weight, in the relative abundance of ripe females and spent females, and in the occurrence of mouth-brooding females. The peaks of all these bimodal patterns coincided with the driest months in the region. Therefore, reproductive activity of females was more intense during dry seasons. Interestingly, we observed a discontinuity in ovarian development during mouth brooding, a characteristic that has been rarely reported. Although mature males had sperm in their testes all year long, their gonadal weight showed a peak during dry months, coinciding with the reproductive peak of females.
The variation in the breeding patterns of tropical freshwater fishes has been explained with several hypotheses. Humphries et al. (1999) suggested that dry-season breeding in mountain rivers avoids floods that flush-out younger fish and their food. Here we observed the dry-season breeding in G. steindachneri, a species that presumably has a reduced mortality of offspring due to flush out because it cares for its offspring with mouth brooding. This assumption is reasonable because parental care, in general, reduces mortality of offspring (Sargent & Gross, 1985Gross, M. R. & R. C. Sargent. 1985. The evolution of male and female parental care in fishes. American Zoologist, 25: 807-822.). Our results suggest that other factors than the flushing-out of offspring by floods may be determining the breeding season in this species. Floods also may be flushing out items reported as food for younger tropical fishes (Winemiller, 1989Winemiller, K. O. 1989. Patterns of variation in life history among South American fishes in seasonal environments. Oecologia, 81: 225-241.; Esteves, 1996Esteves, K. E. 1996. Feeding ecology of three Astyanax species (Characidae, Tetragonopterinae) from a floodplain lake of Mogi-Guaçú River, Paraná River basin, Brazil. Environmental Biology of Fishes, 46: 83-101.; Mérigoux & Ponton, 1998), such as zooplankton (Saunders III & Lewis Jr., 1988Saunders III, J. F. & W. M. Lewis, Jr. 1988. Zooplankton abundance and transport in a tropical white-water river. Hydrobiologia, 162: 147-155.), macroinvertebrates (Lytle, 2000Lytle, D. A. 2000. Biotic and abiotic effects of flash flooding in a montane desert stream. Archiv für Hydrobiologie, 150: 85-100.), and epilithon (Grether et al., 2001; Kohler et al., 2012Kohler, T. J., T. N. Heatherly, R. W. El-Sabaawi, E. Zandonà, M. C. Marshall, A. S. Flecker, C. M. Pringle, D. N. Reznick & S. A. Thomas. 2012. Flow, nutrients, and light availability influence Neotropical epilithon biomass and stoichiometry. Freshwater Science, 31: 1019-1034.). At least for adults, food reduction does not explain the breeding pattern of the population of G. steindachneri studied here because food was apparently available during non-breeding months, according to the heavy body, fat, and liver weights observed during wet months in both males and females. Turbidity is another, potentially key factor that may be determining the timing of breeding in G. steindachneri and in other tropical fishes living in mountain rivers. High turbidity and sediment load are characteristic of stream floods (Wood & Armitage, 1997Wood, P. & P. D. Armitage. 1997. Biological effects of fine sediment in the lotic environment. Environmental Management, 21: 203-217.; Restrepo & Kjerfve, 2000Restrepo, J. D. & B. Kjerfve. 2000. Magdalena river: interannual variability (1975-1995) and revised water discharge and sediment load estimates. Journal of Hydrology, 235: 137-149.). High sediment load affects the food web (Newcombe & MacDonald, 1991Newcombe, C. P. & D. D. MacDonald. 1991. Effects of suspended sediments on aquatic ecosystems. North American Journal of Fisheries Management, 11: 72-82.; Davies-Colley et al., 1992Davies-Colley, R. J., C. W. Hickey, J. M. Quinn & P. A. Ryan. 1992. Effects of clay discharges on streams. 1. optical properties and epilithon. Hydrobiologia, 248: 215-234.; Wood & Armitage, 1997) and the foraging and reproductive behavior of fishes (Bruton, 1985Bruton, M. N. 1985. The effects of suspensoids on fish. Hydrobiologia, 125: 221-241.; Gray et al., 2011Gray, S. M., S. Sabbah & C. W. Hawryshyn. 2011. Experimentally increased turbidity causes behavioural shifts in Lake Malawi cichlids. Ecology of Freshwater Fish, 20: 529-536.). We suggest that future studies should pay close attention to turbidity and its associated factors as plausible key parts to understand the patterns of breeding in tropical freshwater fishes.
Several reproductive traits observed in this species may be related with the mouth brooding. The low fecundity observed in G. steindachneri (220 oocytes) is similar to that observed in other mouth-brooding cichlids (e.g., G. surinamensis (Bloch, 1791), 250 oocytes; Satanoperca jurupari (Heckel, 1840), 300 eggs; Keith et al., 2000Keith, P., P.- Y. Le Bail & P. Planquette. 2000. Atlas des poissons d'eau douce de Guyane. Paris, Muséum National D'historie Naturelle. Institut d'Ecologie et de gestion de la Biodiversité. Tome 2: Fascicule I. Batrachoidiformes, Mugiliformes, Beloniformes, Cyprinodontiformes, Synbranchiformes, Perciformes, Pleuronectiformes, Tetraodontiformes.), a low value relative to other fishes (Godinho et al., 2010Godinho, A. L., I. R. Lamas & H. P. Godinho. 2010. Reproductive ecology of Brazilian freshwater fishes. Environmental Biology of Fishes, 87: 143-162.; Lowe-McConnell, 1987). Such relative low fecundities are typical of species that show parental care, what can be explained as a trade-off between fecundity and parental investment when resources are limited (Okuda et al., 1998Okuda, N., I. Tayasu & Y. Yanagisawa. 1998. Determinate growth in a paternal mouthbrooding fish whose reproductive success is limited by buccal capacity. Evolutionary Ecology, 12: 681-699.). The analysis of frequencies of oocytes diameters indicates group-synchronous oogenesis in this species, what has been previously observed in other cichlids (e.g., G. brasiliensis (Quoy & Gaimard, 1824); Mazzoni & Iglesias-Rios 2002Mazzoni, R. & R. Iglesias-Rios. 2002. Environmentally related life history variations in Geophagus brasiliensis. Journal of Fish Biology, 61: 1606-1618.). Histological analyses allowed us to notice synchronization between a discontinuity in oogenesis and the development of offspring in female's mouths, what has been observed before in other cichlid (Tubert et al., 2012Tubert, C., F. Lo Nostro, V. Villafañe & M. Pandolfi. 2012. Aggressive behavior and reproductive physiology in females of the social cichlid fish Cichlasoma dimerus. Physiology & Behavior, 106: 193-200.). Similarly, the interspawning interval in Oreochromis niloticus (Linnaeus, 1758) is longer in mouth brooding females than in females with broods experimentally removed, indicating that mouth brooding slows gonadal development in this species (Tacon et al., 1996Tacon, P., P. Ndiaye, C. Cauty, F. Le Menn & B. Jalabert. 1996. Relationships between the expression of maternal behaviour and ovarian development in the mouthbrooding cichlid fish Oreochromis niloticus. Aquaculture, 146: 261-275.). That pause should allow females to conclude the process of caring for one batch of offspring before engaging in a new reproductive episode. The presence of developing oocytes in some females with juveniles in their mouths indicates that at least some females are able to have two reproductive events during the same breeding season, similar to what occurs in G. brasiliensis (Barbieri et al., 1981Barbieri, M. C., G. Barbieri & M. de. A. Marins. 1981. Sobre a anatomia e histologia de ovário de Geophagus brasilliensis (Quoy & Gaimard, 1824) na represa do Lobo, estado de São Paulo. Revista Brasileira de Biología, 41: 163-168.).
Monthly variation in liver and fat weights revealed energy investment in reproduction. For females, the energy expenditure may be most related with hepatic vitellogenesis (Fontaínhas-Fernandes et al., 2002) and egg recruitment (Lucey, 2009Lucey, S. M. 2009. Characteristics of fish yolk proteins and a method for inducing vitellogenin. Unpublished Ph. d. Dissertation, University of Massachusetts, 47p.), as revealed by histological analysis. Mouth brooding seems to be costly for females as indicated by the reduction of their fat reserves during reproductive months, which may be caused by energy invested in egg production, courtship, and the reduction in food ingestion while brooding. Moreover, mouth brooding has another cost for females, it seem to be delaying the following reproductive event of females until their juveniles become independent.
Females of G. steindachneri showed at least two breeding events during breeding seasons, according to the observation of recruitment of vitellogenetic eggs in females with juveniles in their mouths. However, more data is needed to confirm the number of reproductive events of the females of this species in a given season, in a hydrological cycle, and in general in their lifetime. Males, on the other hand, seem to be readily available for reproductive events all year long, what is not surprising given that spermatogenesis does not require a large investment in energy resources of males (Huntingford et al., 2001Huntingford, F. A., S. Chellappa., A. C. Taylor & R. H. C. Strang. 2001. Energy reserves and reproductive investment in male three-spined sticklebacks, Gasterosteus aculeatus. Ecology of Freshwater Fish, 10: 111-117.). However, males heavily invested their energy reserves during dry months, possibly on performing the energetically costly activities of defending their territory or courting females (Chellappa et al., 1989Chellappa, S., Huntingford, R. H. C. Strang & R. Y. Thomson. 1989. Annual variation in energy reserves in male three-spined stickleback, Gasterosteus aculeatus L. (Pisces, Gasterosteidae). Journal of Fish Biology, 35: 275-286.; Suriyampola & Eason, 2014Suriyampola, P. S. & P. K. Eason. 2015. Sex differences in territorial defense in a shell-breeding cichlid, Neolamprologus multifasciatus. Acta Ethologica, 18: 59-68.).
This study is one of the few on the breeding patterns of fishes in mountain streams. It is remarkable that this basic piece of natural history is still unknown for most of the highly diverse tropical freshwater fishes living in mountain rivers. These rivers are subject to strong anthropic pressures, including drastic changes in watershed soil use, loss of riparian forests, water diversion, and stream damming, all of which modify the natural stream flow and function. Learning more about breeding patterns of fishes in mountain rivers may reveal their importance in these ecosystems. For example, annual reproductive migrations of Prochilodus mariae Eigenmann, 1922 are closely tied with the carbon cycle in piedmont streams of the Venezuelan Andes (Taylor et al., 2006Taylor, B. W., A. S. Flecker & R. O. Hall, Jr. 2006. Loss of a harvested fish species disrupts carbon flow in a diverse tropical river. Science, 313: 833-836.). More of this kind of knowledge can also be used to improve river management, for example determining the natural stream flow that a species needs to complete its breeding cycle.
Acknowledgements
We want to thank Martha Patricia Ramírez-Pinilla for her guidance in the different aspects of this work well as provide facilities and equipment of Laboratorio de Biología Reproductiva de Vertebrados. Many thanks to M. Valderrama-Vernaza and P. Rueda-Cediel for all their support and parental care of our respective offspring during this work. Fieldwork was assisted by N. Pinto-Sánchez, J. Santander, O. Rojas, Myyo, M. Rojas, M. Quijano-Abril, and A. Motta. Limnological equipment was kindly facilitated by R. A. Gavilán-Díaz and Escuela de Biología (UIS). Special thanks to the members of the LBRV-UIS. The Bucaramanga office of INCODER issued the permission to capture the specimens. This study was supported by LBRV-UIS and our beloved families.
References
- Alkins-koo, M. 2000. Reproductive timing of fishes in a tropical intermittent stream. Environmental Biology of Fishes, 57: 49-66.
- Balon, E. K. 1975. Reproductive guilds of fishes: a proposal and definition. Journal of the Fisheries Research Board of Canada, 32: 821-864.
- Barbieri, M. C., G. Barbieri & M. de. A. Marins. 1981. Sobre a anatomia e histologia de ovário de Geophagus brasilliensis (Quoy & Gaimard, 1824) na represa do Lobo, estado de São Paulo. Revista Brasileira de Biología, 41: 163-168.
- Bronson, F. H. 2009. Climate change and seasonal reproduction in mammals. Philosophical Transactions of the Royal Society of London B, Biological Sciences, 364: 3331-3340.
- Bruton, M. N. 1985. The effects of suspensoids on fish. Hydrobiologia, 125: 221-241.
- Cala, P., E. Gonzáles & M. P. Varona. 1996. Aspectos biológicos y taxonoomicos del tucunare, Cichla monoculus (Pisces: Cichlidae). Dahlia, 1: 23-37.
- Chapman, C. A., L. J. Chapman, T. T. Struhsaker, A. E. Zanne, C. J. Clark & J. R. Poulsen. 2005. A long-term evaluation of fruiting phenology: importance of climate change. Journal of Tropical Ecology, 21: 31-45.
- Chapman, L. J. & D. L. Kramer. 1991. The consequences of flooding for the dispersal and fate of poeciliid fish in an intermittent tropical stream. Oecologia, 87: 299-306.
- Chellappa, S., Huntingford, R. H. C. Strang & R. Y. Thomson. 1989. Annual variation in energy reserves in male three-spined stickleback, Gasterosteus aculeatus L. (Pisces, Gasterosteidae). Journal of Fish Biology, 35: 275-286.
- Coward, K. & N. R. Bromage. 1998. Histological classification of oocyte growth and the dynamics of ovarian recrudescence in Tilapia zillii. Journal of Fish Biology, 53: 285-302.
- Crampton, W. G. R. 2008. Ecology and life history of an Amazon floodplain cichlid: the discus fish Symphysodon (Perciformes: Cichlidae). Neotropical Ichthyology, 6: 599-612.
- Davies-Colley, R. J., C. W. Hickey, J. M. Quinn & P. A. Ryan. 1992. Effects of clay discharges on streams. 1. optical properties and epilithon. Hydrobiologia, 248: 215-234.
- Esmaeili, H. R., Z. Ganjali & M. Monsefi. 2010. Gonad morphology and histology of the endemic Hormuz cichlid, Iranocichla hormuzensis Coad, 1982 from Mehran River, Southern Iran. IUFS Journal of Biology, 69: 1-12.
- Esteves, K. E. 1996. Feeding ecology of three Astyanax species (Characidae, Tetragonopterinae) from a floodplain lake of Mogi-Guaçú River, Paraná River basin, Brazil. Environmental Biology of Fishes, 46: 83-101.
- Flecker, A. S., D. C. Taphorn, J. A. Lovel & B. P. Feifarek. 1991. Drift of characin larvae, Bryconamericus deuterodonoides, during the dry season from Andean Piedmont streams. Environmental Biology of Fishes, 31: 197-202.
- Fontaínhas-Fernandes, A., E. Gomes, Ma. A. Reis-Henriques & J. Coimbra. 2002. Plasma thyroid hormones and hepatic nucleic acids in relation to sex of tilapia Oreochromis niloticus. Journal of Applied Ichthyology, 18: 185-191.
- Godinho, A. L., I. R. Lamas & H. P. Godinho. 2010. Reproductive ecology of Brazilian freshwater fishes. Environmental Biology of Fishes, 87: 143-162.
- González, E., M. P. Varona & P. Cala. 1996. Datos biológicos del oscar, Astronotus ocellatus (Pisces: Cichlidae), en los alrededores de Leticia, Amazonia. Dahlia, 1: 51-62.
- Gray, S. M., S. Sabbah & C. W. Hawryshyn. 2011. Experimentally increased turbidity causes behavioural shifts in Lake Malawi cichlids. Ecology of Freshwater Fish, 20: 529-536.
- Grether, G. F., D. F. Millie, M. J. Bryant, D. N. Reznick & W. Mayea. 2001. Rain forest canopy cover, resource availability, and life history evolution in guppies. Ecology, 82: 1546-1559.
- Harikumar, S., K. G. Padmanabhan, P. A. John & K. Kortmulder. 1994. Dry-season spawning in a cyprinid fish of southern India. Environmental Biology of Fishes, 39: 129-136.
- Humphries, P., A. J. King & J. D. Koehn. 1999. Fish , flows and flood plains: links between freshwater fishes and their environment in the Murray-Darling River system, Australia. Environmental Biology of Fishes, 56: 129-151.
- Huntingford, F. A., S. Chellappa., A. C. Taylor & R. H. C. Strang. 2001. Energy reserves and reproductive investment in male three-spined sticklebacks, Gasterosteus aculeatus. Ecology of Freshwater Fish, 10: 111-117.
- Keith, P., P.- Y. Le Bail & P. Planquette. 2000. Atlas des poissons d'eau douce de Guyane. Paris, Muséum National D'historie Naturelle. Institut d'Ecologie et de gestion de la Biodiversité. Tome 2: Fascicule I. Batrachoidiformes, Mugiliformes, Beloniformes, Cyprinodontiformes, Synbranchiformes, Perciformes, Pleuronectiformes, Tetraodontiformes.
- Kohler, T. J., T. N. Heatherly, R. W. El-Sabaawi, E. Zandonà, M. C. Marshall, A. S. Flecker, C. M. Pringle, D. N. Reznick & S. A. Thomas. 2012. Flow, nutrients, and light availability influence Neotropical epilithon biomass and stoichiometry. Freshwater Science, 31: 1019-1034.
- Kramer, D. L. 1978. Reproductive seasonality in the fishes of a tropical stream. Ecology, 59: 976-985.
- Kullander, S. O. 2003. Family Cichlidae (Cichlids). Pp. 605-654. In: Reis, R. E., S. O. Kullander & C. J. Ferraris, Jr. (Orgs.). Check list of the freshwater fishes of South and Central America. Porto Alegre, Edipucrs.
- Lewis Jr., W. M., S. K. Hamilton & J. F. Saunders III. 2006. Rivers of Northern South America. Pp: 219-256. In: Cushing, C. E., K. V. Cummins & G. W. Minshall (Eds.). River and stream ecosystems of the world. University of California Berkeley Press.
- López-Fernández, H., K. O. Winemiller, C. Montaña & R. L. Honeycutt. 2012. Diet-morphology correlations in the radiation of South American geophagine cichlids (Perciformes: Cichlidae: Cichlinae). PloS One, 7: 1-14.
- Lowe-McConnell, R. H. 1969. The cichlid fishes of Guyana, South America, with notes on their ecology and breeding behaviour. Zoological Journal of the Linnean Society, 48: 255-302.
- Lowe-McConnell, R. H. 1987. Ecological studies in tropical fish communities. Cambridge, University Press, 382p. (Cambridge tropical biology series).
- Lucey, S. M. 2009. Characteristics of fish yolk proteins and a method for inducing vitellogenin. Unpublished Ph. d. Dissertation, University of Massachusetts, 47p.
- Luna, L. G. (Ed.) 1968. Manual of histologic staining methods of the Armed Forces Institute of Pathology. 3rd ed. New York, McGraw Hill, 258p.
- Lytle, D. A. 2000. Biotic and abiotic effects of flash flooding in a montane desert stream. Archiv für Hydrobiologie, 150: 85-100.
- Mazzoni, R. & R. Iglesias-Rios. 2002. Environmentally related life history variations in Geophagus brasiliensis. Journal of Fish Biology, 61: 1606-1618.
- Menezes, N. A. & A. E. A. de M. Vazzoler. 1992. Reproductive characteristics of characiformes. Pp. 60-70. In: Hamlett, W. C. (Ed.). Reproductive biology of South American vertebrates. New York, Springer.
- Mérigoux, S. & D. Ponton. 1998. Body shape, diet and ontogenetic diet shifts in young fish of the Sinnamary River, French Guiana, South America. Journal of Fish Biology, 52: 556-569.
- Morales-Nin, B. & J. Panfili. 2005. Seasonality in the deep sea and tropics revisited: what can otoliths tell us? Marine and Freshwater Research, 56: 585-598.
- Munro, A. D. 1990. Tropical freshwater fish. Pp. 145-239. In: Munro, A. D., A. P. Scott & T. J. Lam. (Eds.). Reproductive seasonality in Teleosts: environmental influences. Boca Raton, CRC.
- Newcombe, C. P. & D. D. MacDonald. 1991. Effects of suspended sediments on aquatic ecosystems. North American Journal of Fisheries Management, 11: 72-82.
- Okuda, N., I. Tayasu & Y. Yanagisawa. 1998. Determinate growth in a paternal mouthbrooding fish whose reproductive success is limited by buccal capacity. Evolutionary Ecology, 12: 681-699.
- Payne, A. I. 1986. The ecology of tropical lakes and rivers. Chichester, J. Wiley. 301p.
- Perrone, M., Jr. & T. M. Zaret. 1979. Parental care patterns of fishes. The American Naturalist, 113: 351-361.
- Pusey, B. J., A. H. Arthington, P. G. Close & J. R. Bird. 2002. Larval fishes in rainforest streams: recruitment and microhabitat use. Proceedings of the Royal Society of Queensland, 110: 27-46.
- R Development Core Team. 2014. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna.
- Restrepo, J. D. & B. Kjerfve. 2000. Magdalena river: interannual variability (1975-1995) and revised water discharge and sediment load estimates. Journal of Hydrology, 235: 137-149.
- Román-Valencia, C. & A. Botero. 2006. Trophic and reproductive ecology of a species of Hemibrycon (Pisces: Characidae) in Tinajas creek , Quindío River drainage, upper Cauca basin, Colombia. Revista del Museo Argentino de Ciencias Naturales, n.s., 8: 1-8.
- Rossoni, F., S. Amadio, E. Ferreira, & J. Zuanon. 2010. Reproductive and population parameters of discus fish Symphysodon aequifasciatus Pellegrin, 1904 (Perciformes: Cichlidae) from Piagaçu-Purus Sustainable Development Reserve (RDS-PP), lower Purus River, Amazonas, Brazil. Neotropical Ichthyology, 8: 379-383.
- Gross, M. R. & R. C. Sargent. 1985. The evolution of male and female parental care in fishes. American Zoologist, 25: 807-822.
- Saunders III, J. F. & W. M. Lewis, Jr. 1988. Zooplankton abundance and transport in a tropical white-water river. Hydrobiologia, 162: 147-155.
- Sazima, I. 1980. Behavior of two Brazilian species of parodontid fishes, Apareiodon piracicabae and A. ibitiensis. Copeia, 1: 166-169.
- Selman, K., R. A. Wallace & V. Barr. 1986. Oogenesis in Fundulus heteroclitus IV. Yolk-vesicle formation. Journal of Experimental Zoology Part A, 239: 277-288.
- Señaris, J. C. & C. Lasso A. 1993. Ecologia alimentaria y reproductiva de la mojarra de río Caquetaia kraussii (Steindachner 1878) (Cichlidae) en los llanos inundables de Venezuela. Publicaciones de la Asociación Amigos de Doñana, 2: 1-58.
- Smith, B. B. & K. F. Walker. 2004. Spawning dynamics of common carp in the River Murray , South Australia, shown by macroscopic and histological staging of gonads. Journal of Fish Biology, 64: 336-354.
- Suriyampola, P. S. & P. K. Eason. 2015. Sex differences in territorial defense in a shell-breeding cichlid, Neolamprologus multifasciatus. Acta Ethologica, 18: 59-68.
- Tacon, P., P. Ndiaye, C. Cauty, F. Le Menn & B. Jalabert. 1996. Relationships between the expression of maternal behaviour and ovarian development in the mouthbrooding cichlid fish Oreochromis niloticus. Aquaculture, 146: 261-275.
- Taylor, B. W., A. S. Flecker & R. O. Hall, Jr. 2006. Loss of a harvested fish species disrupts carbon flow in a diverse tropical river. Science, 313: 833-836.
- Torres-Mejica, M. & M. P. Ramírez-Pinilla. 2008. Dry-season breeding of a characin in a neotropical mountain river. Copeia, 1: 99-104.
- Tubert, C., F. Lo Nostro, V. Villafañe & M. Pandolfi. 2012. Aggressive behavior and reproductive physiology in females of the social cichlid fish Cichlasoma dimerus. Physiology & Behavior, 106: 193-200.
- Vazzoler, A. E. A. de M. 1996. Biologia da reprodução de peixes teleósteos: teoria e prática. Maringá, Eduem, São Paulo, SBI, 169p.
- Wang, J., M. Liu & L. Fang. 1995. The reproductive biology of an endemic cyprinid, Zacco pachycephalus in Taiwan. Environmental Biology of Fishes, 43: 135-143.
- Welcomme, R. 1979. The fisheries ecology of floodplain rivers. London.
- Wimberger, P. H. 1991. Plasticity of jaw and skull morphology in the Neotropical cichlids Geophagus brasiliensis and G. steindachneri. Evolution, 45: 1546-1563.
- Winemiller, K. O. 1989. Patterns of variation in life history among South American fishes in seasonal environments. Oecologia, 81: 225-241.
- Wohl, E. & D. M. Merritt. 2008. Reach-scale channel geometry of mountain streams. Geomorphology, 93: 168-185.
- Wood, P. & P. D. Armitage. 1997. Biological effects of fine sediment in the lotic environment. Environmental Management, 21: 203-217.
- Zar, J. H. 1999. Biostatistical analysis. 4th ed. Upper Saddle River, NJ, Prentice Hall.
Publication Dates
-
Publication in this collection
26 June 2015 -
Date of issue
Apr-Jun 2015
History
-
Received
24 June 2014 -
Accepted
25 Jan 2015