ABSTRACT
During an ichthyological survey in September 2015 at the Ceará-Mirim River estuary, Rio Grande do Norte State, northeastern Brazil, we collected a male of Kryptolebias hermaphroditus , a cynolebiid species that had been previously described as containing exclusively self-fertilizing hermaphrodites. This is the first record of a male in this species, over 140 years after the discovery of the mangrove rivulid species from Brazil. Our discovery reinforces the need for more studies in K. hermaphroditus , as well as the potential of this species as a model for evolutionary studies due to its unique mating system.
Keywords:
Hermaphroditism; Mangrove killifish; Mid-Northeastern Caatinga ecoregion; Mixed-mating system; Sexual dimorphism
RESUMO
Durante uma amostragem ictiológica em setembro de 2015 no estuário do rio Ceará-Mirim, Estado do Rio Grande do Norte, Nordeste do Brasil, nós coletamos um macho de Kryptolebias hermaphroditus , uma espécie de rivulídeo que foi descrita como contendo apenas hermafroditas auto-fertilizantes. Este é o primeiro registro de um macho dessa espécie, mais de 140 anos depois da descoberta das espécies de rivulídeos de manguezais do Brasil. Nossa descoberta reforça a necessidade de mais estudos em K . hermaphroditus , assim como, o potencial desta espécie como um modelo para estudos evolutivos devido ao seu sistema reprodutivo único.
Introduction
Although the overall taxonomy of Kryptolebias Costa, 2004 is confusing (Costa, 2006Costa, W. J. E. M. 2006. Redescription of Kryptolebias ocellatus (Hensel) and K. caudomarginatus (Seegers) (Teleostei: Cyprinodontiformes: Rivulidae), two killifishes from mangroves of south-eastern Brazil. Aqua Journal of Ichthyology and Aquatic Biology, 11: 5-12.), the K. marmoratus species group is well supported by both molecular (Tartarenkov et al. , 2009Tatarenkov, A., S. M. Q. Lima, D. S. Taylor & J. C. Avise. 2009. Long-term retention of self-fertilization in a fish clade. Proceedings of the National Academy of Sciences, 106: 14456-14459.) and morphological data (Costa et al ., 2010Costa, W. J. E. M., S. M. Q. Lima & R. Bartolette. 2010. Androdioecy in Kryptolebias and the evolution of self-fertilizing hermaphroditism. Biological Journal of the Linnean Society, 99: 344-349.). The species comprising this group, K. marmoratus (Poey, 1880), K. ocellatus (Hensel, 1868) and K. hermaphroditus Costa, 2011, inhabit estuarine areas associated with mangroves, ranging from southeastern United States of America to southern Brazil (Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.). Besides this unusual brackish habitat, these species are also unique among aplocheiloid killifishes due to the absence of females, which are replaced by hermaphrodites (Costa et al. , 2010Costa, W. J. E. M., S. M. Q. Lima & R. Bartolette. 2010. Androdioecy in Kryptolebias and the evolution of self-fertilizing hermaphroditism. Biological Journal of the Linnean Society, 99: 344-349.).
Indeed, self-fertilizing hermaphroditism is the dominant mode of reproduction in Kryptolebias marmoratus distributed from Florida to Venezuela (Tatarenkov et al., 2011Tatarenkov, A., S. M. Q. Lima & J. C. Avise. 2011. Extreme homogeneity and low genetic diversity in Kryptolebias ocellatus from south-eastern Brazil suggest a recent foundation for this androdioecious fish population. Journal of Fish Biology, 79: 2095-2105.) and K. hermaphroditus occurring in the Atlantic Forest mangroves of Brazil (Lira et al., 2015Lira, M. G. S., R. E. C. Paiva, T. P. A. Ramos & S. M. Q. Lima. 2015. First record of Kryptolebias hermaphroditus Costa, 2011 (Cyprinodontiformes: Rivulidae) in the extreme north Atlantic Forest mangroves, Rio Grande do Norte state, Brazil. Check List, 11: 1656.). This clade represents the only occurrence of self-fertilizing hermaphroditism among vertebrates (Tartarenkov et al. , 2009Tatarenkov, A., S. M. Q. Lima, D. S. Taylor & J. C. Avise. 2009. Long-term retention of self-fertilization in a fish clade. Proceedings of the National Academy of Sciences, 106: 14456-14459.; Costa et al., 2010Costa, W. J. E. M., S. M. Q. Lima & R. Bartolette. 2010. Androdioecy in Kryptolebias and the evolution of self-fertilizing hermaphroditism. Biological Journal of the Linnean Society, 99: 344-349.). Because of its reproductive mixed-mated system, K. marmoratus has been a popular model species for embryological, physiological, behavioral and genetic studies (Avise & Tatarenkov, 2015Avise, J. C. & A. Tatarenkov. 2015. Population genetics and evolution of the mangrove rivulus Kryptolebias marmoratus , the world's only self-fertilizing hermaphroditic vertebrate. Journal of Fish Biology, 87: 519-538.). While males are rare in K. marmoratus , they are considered to be absent in K. hermaphroditus (Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.; Tatarenkov et al. , 2011Tatarenkov, A., S. M. Q. Lima & J. C. Avise. 2011. Extreme homogeneity and low genetic diversity in Kryptolebias ocellatus from south-eastern Brazil suggest a recent foundation for this androdioecious fish population. Journal of Fish Biology, 79: 2095-2105.), despite this species having been known at least since 1868 (previously known as K. ocellatus ) and numerous museum and unpreserved specimens have been examined (Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.). Due to the absence of males in fish collections, and following a re-examination of the holotype of K. ocellatus , the monomorphic hermaphrodite species from Brazil was described as K. hermaphroditus (Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.).
Hermaphrodites of both species (K. marmoratus and K. hermaphroditus ) typically have a black ocellus on the caudal peduncle and a dark-grey body side (Soto & Noakes, 1994Soto, C. G. & D. L. G. Noakes. 1994. Coloration and gender in the hermaphroditic fish Rivulus marmoratus Poey (Teleostei: Rivulidae). Ichthyological Exploration of Freshwaters, 5: 79-90.; Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.). In K. marmoratus , males exhibit a bright orange coloration on the body flank and fins, an absent or poorly-defined ocellus on the caudal peduncle, and black margins on the caudal fins (Davis et al., 1990Davis, W. P., D. S. Taylor & B. J. Turner. 1990. Field observations of the ecology and habits of mangrove rivulus (Rivulus marmoratus ) in Belize and Florida (Teleostei: Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 1: 123-134.; Soto & Noakes, 1994Soto, C. G. & D. L. G. Noakes. 1994. Coloration and gender in the hermaphroditic fish Rivulus marmoratus Poey (Teleostei: Rivulidae). Ichthyological Exploration of Freshwaters, 5: 79-90.; Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.).
In a recent collection in the Ceará-Mirim River estuary, we found a Kryptolebias hermaphroditus individual with a male phenotype. Considering that this species has been described as being completely composed of hermaphrodites, the main objective of the present study is to record and describe the first occurrence of a male K. hermaphroditus .
Material and Methods
In September 2015, during a nocturnal exploratory fieldwork being conducted with the objective of designing an ecological study on Kryptolebias hermaphroditus at the Ceará-Mirim River estuary (05°40'25.9"S 35°14'14.5"W, see Lira et al., 2015Lira, M. G. S., R. E. C. Paiva, T. P. A. Ramos & S. M. Q. Lima. 2015. First record of Kryptolebias hermaphroditus Costa, 2011 (Cyprinodontiformes: Rivulidae) in the extreme north Atlantic Forest mangroves, Rio Grande do Norte state, Brazil. Check List, 11: 1656. for a map and detailed description of the area), we observed a bright orange individual of Kryptolebias (Fig. 1). This specimen together with specimens exhibiting a more typical hermaphroditic appearance, were captured by hand and with the help of a hand sieve (50 x 60 cm, 1 mm mesh), under permit #30532-1/2011 issued by ICMBio/SISBIO. The fishes were euthanized using clove oil, fixed in formalin solution, transferred to an 70% alcohol solution and then deposited in the ichthyological collection of Universidade Federal do Rio Grande do Norte (UFRN 3774, 8 ex.). Some other individuals, as well as a small fin-clip (TIUFRN 3481; tissue collection of UFRN), were directly stored in 96% ethanol for molecular analysis.
Measurements and counts for the orange individual follow Costa (1995Costa, W. J. E. M. 1995. Pearl killifishes: the Cynolebiatinae: systematics and biogeography of the Neotropical annual fish subfamily. Neptune city, TFH Publications, 128p.). Kryptolebias species identification was carried out using the key proposed by Costa (2009Costa, W. J. E. M. 2009. Peixes aploqueilóideos da Mata Atlântica brasileira: história, diversidade e conservação. Rio de Janeiro, Museu Nacional. 171p.), and later confirmed using Costa (2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.). To identify male traits, we used the information described for the closely related species K. marmoratus (Davis et al., 1990Davis, W. P., D. S. Taylor & B. J. Turner. 1990. Field observations of the ecology and habits of mangrove rivulus (Rivulus marmoratus ) in Belize and Florida (Teleostei: Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 1: 123-134.; Soto & Noakes, 1994Soto, C. G. & D. L. G. Noakes. 1994. Coloration and gender in the hermaphroditic fish Rivulus marmoratus Poey (Teleostei: Rivulidae). Ichthyological Exploration of Freshwaters, 5: 79-90.; Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.).
Kryptolebias hermaphroditus , male, UFRN 3774, 30.1 mm SL (three days after sampling); Ceará-Mirim River estuary, Extremoz, Rio Grande do Norte, Brazil.
Results
All specimens were identified as Kryptolebias hermaphroditus on the basis of having 1-4 teeth in the vomer and a color pattern with silvery or pale golden spots on the dorsal part of the flank. The male individual had bright orange coloration on the body flank and fins (mainly dorsal, anal and caudal), and black margins on the anal and caudal fins (Fig. 1), as in males of K. marmoratus .
Comparing male and hermaphroditic phenotypes of the captured specimens, both have a dark ocellus on the caudal peduncle; however, the ocellus is faint on the male specimen. The hermaphrodites consistently exhibit a brown or grey flank coloration, as well as hyaline unpaired fins with pale grey dots on the basal portion, whereas the male individual exhibits unpaired fins with orange coloration (Fig. 2).
Kryptolebias hermaphroditus , male (below) 30.1 mm SL, hermaphrodite (above) 35.5 mm SL, UFRN 3774, Ceará-Mirim River estuary, Extremoz, Rio Grande do Norte, Brazil.
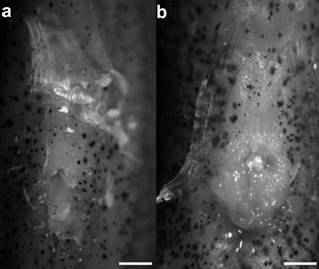
Description. Morphometric data of male (30.1 mm SL) in Table 1. Urogenital papilla cylindrical in male, pocket-like shape in hermaphrodites (Fig. 3). Dorsal fin rounded. Anal fin sub-trapezoidal, distal margin slightly damaged; anal-fin rays 4 and 5 longer than other anal-fin rays. Caudal fin oval, deeper than long. Pectoral fin short and rounded. Pelvic fin elliptical. Pelvic-fin bases medially separated by short interspace. Dorsal-fin origin at vertical between 9th and 10th anal-fin rays. Dorsal-fin rays 8; anal-fin rays 11; caudal-fin rays 30; pectoral-fin rays 13; pelvic-fin rays 6.
Scales small, cycloid. Frontal squamation E-patterned. Longitudinal series of scales 47; transverse series of scales 13; scale rows around caudal peduncle 25. Contact organs absent.
Urogenital papillae of Kryptolebias hermaphroditus in ventral view; UFRN 3374: a, cylindrical shape in male, 30.1 mm SL; b, pocket shape in hermaphrodite, 35.5 mm SL. Scale bars = 1 mm.
Coloration. Flank dark grey on anterior and dorsal portion of head, from greyish to orange on posterior and ventral portions. Side of the body intensely pigmented with faint dark grey stripe between postorbital region and caudal-fin base, and small irregular somewhat zigzag pattern composed of small orange spots more concentrated on anterior half of body side. Inconspicuous dark grey humeral blotch; rounded dark spot with faint yellowish grey margin on dorsal end of caudal peduncle. Side of head light brown on dorsal portion, pale golden with dark grey dots ventrally. Jaws grey. Iris brown, with narrow pale yellow margin around pupil. Dorsal, anal and caudal fins hyaline with dark orangish-brown on basal and medial portions of fins; black stripe on distal anterior margin of anal and lower portion of caudal fin. Pectoral and pelvic fins hyaline. Bright orange coloration faded a little after sampling, probably due to stress and non-natural conditions (Figs. 1-2).
Ecological notes. The sampling locality had clear water and the fishes could be observed using flashlights. The Kryptolebias hermaphroditus male was captured in an ephemeral pool at higher elevations among the estuary's mangroves. This pool had at least four fiddler crabs (Uca maracoani Latreille, 1802) burrow openings and, prior to our approach, the male fish was resting on the water's surface near one of these burrows. During our sampling attempts, the fish repeatedly hid and re-appeared among the crab burrows, appearing periodically in different openings until it was caught. Time between reappearances was about one minute.
Large (23.2-35.5 mm SL) hermaphrodites were also collected in the same area as the male, in shallow pools formed in the edge of the mangroves; in the inner and deeper pools, Poecilia vivipara Bloch & Schneider, 1801 was the dominant species. Guavina guavina (Valenciennes, 1837), potential predator of Kryptolebias hermaphroditus , were also active during the night and observed entering the same large crab burrows. Salinity in these pools, previously recorded in January 2015 at the same site, was 34-36 ppm during the low tide.
Discussion
Over the 140 years, since the description of Kryptolebias ocellatus (as was long called hermaphroditic species who was later described as K. hermaphroditus ) from a mangrove in Rio the Janeiro, no males of this hermaphroditic species from the Brazilian coast have ever been found (Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.). However, potential miswritten on previous literature (Costa, 2006Costa, W. J. E. M. 2006. Redescription of Kryptolebias ocellatus (Hensel) and K. caudomarginatus (Seegers) (Teleostei: Cyprinodontiformes: Rivulidae), two killifishes from mangroves of south-eastern Brazil. Aqua Journal of Ichthyology and Aquatic Biology, 11: 5-12.; Lira et al., 2015Lira, M. G. S., R. E. C. Paiva, T. P. A. Ramos & S. M. Q. Lima. 2015. First record of Kryptolebias hermaphroditus Costa, 2011 (Cyprinodontiformes: Rivulidae) in the extreme north Atlantic Forest mangroves, Rio Grande do Norte state, Brazil. Check List, 11: 1656.) needs to be clarified for a better understanding of the issue. Both studies used the subjective term "rare" while discussing the existence of males in K . hermaphroditus.Costa (2006Costa, W. J. E. M. 2006. Redescription of Kryptolebias ocellatus (Hensel) and K. caudomarginatus (Seegers) (Teleostei: Cyprinodontiformes: Rivulidae), two killifishes from mangroves of south-eastern Brazil. Aqua Journal of Ichthyology and Aquatic Biology, 11: 5-12.) had already mentioned two reports of potential K. hermaphroditus males based on pictures and brief descriptions of live specimens with light grey and dark orange spots on the flanks, grey humeral and caudal peduncle spots and dark grey to black distal zones of unpaired fins. However, this author recognized that this material was not preserved for study in any scientific collection and needed posterior confirmation. Posteriorly, Costa (2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.) described K. hermaphroditus as only composed by hermaphrodites, discarding the relevance of the two previous unconfirmed reports. In both cases male phenotypes were reported for Rio de Janeiro, where K. ocellatus (males and hermaphrodites, previously known as K. caudomarginatus Seegers) occurs in syntopy with K. hermaphroditus making these records more doubtful. The potential records described above, the presence of males on populations of K . marmoratus (Avise & Tatarenkov, 2015Avise, J. C. & A. Tatarenkov. 2015. Population genetics and evolution of the mangrove rivulus Kryptolebias marmoratus , the world's only self-fertilizing hermaphroditic vertebrate. Journal of Fish Biology, 87: 519-538.), as well as the cryptic and complex mangrove microhabitats inhabited by K . hermaphroditus , may have led Costa (2006Costa, W. J. E. M. 2006. Redescription of Kryptolebias ocellatus (Hensel) and K. caudomarginatus (Seegers) (Teleostei: Cyprinodontiformes: Rivulidae), two killifishes from mangroves of south-eastern Brazil. Aqua Journal of Ichthyology and Aquatic Biology, 11: 5-12.) and Lira et al . (2015Lira, M. G. S., R. E. C. Paiva, T. P. A. Ramos & S. M. Q. Lima. 2015. First record of Kryptolebias hermaphroditus Costa, 2011 (Cyprinodontiformes: Rivulidae) in the extreme north Atlantic Forest mangroves, Rio Grande do Norte state, Brazil. Check List, 11: 1656.) to not fully discard the presence of males of K . hermaphroditus , despite extensive sampling and no reliable scientific evidence of males have been made available up to that moment.
The recently collected specimen of Kryptolebias hermaphroditus with male appearance, described herein, matches the description of the putative K. hermaphroditus males mentioned by Costa (2006Costa, W. J. E. M. 2006. Redescription of Kryptolebias ocellatus (Hensel) and K. caudomarginatus (Seegers) (Teleostei: Cyprinodontiformes: Rivulidae), two killifishes from mangroves of south-eastern Brazil. Aqua Journal of Ichthyology and Aquatic Biology, 11: 5-12.) and K. marmoratus males (Davis et al., 1990Davis, W. P., D. S. Taylor & B. J. Turner. 1990. Field observations of the ecology and habits of mangrove rivulus (Rivulus marmoratus ) in Belize and Florida (Teleostei: Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 1: 123-134.; Soto & Noakes, 1994Soto, C. G. & D. L. G. Noakes. 1994. Coloration and gender in the hermaphroditic fish Rivulus marmoratus Poey (Teleostei: Rivulidae). Ichthyological Exploration of Freshwaters, 5: 79-90.; Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.). Although gonadal histological evidence was not surveyed in order to maintain the unique specimen integrity, its morphological characters support this first documentation of a male K. hermaphroditus . This evidence includes orange body and fins, faded ocellus, black margins of anal and caudal fins and cylindrical urogenital papillae (Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.). Once the color pattern of males is the most conspicuous character for differentiating cynolebiid species (Costa, 2003Costa, W. J. E. M. 2003. Family Rivulidae. Pp. 526-548. In: Reis, R. E., S. O. Kullander & C. J. Ferraris Jr. (Eds.). Check list of freshwater fishes of South and Central America. Porto Alegre, Edipucrs.), the discovery of a K. hermaphroditus male may reveal important morphological differences between this species and K. marmoratus .
Regardless the presence of an ocellus on the caudal peduncle, it appeared faded in comparison to that of hermaphrodite individuals. According to Soto & Noakes (1994Soto, C. G. & D. L. G. Noakes. 1994. Coloration and gender in the hermaphroditic fish Rivulus marmoratus Poey (Teleostei: Rivulidae). Ichthyological Exploration of Freshwaters, 5: 79-90.), the most distinctive characters attributed to Kryptolebias marmoratus males are: a bright orange overall coloration and the absence of a distinct caudal ocellus. However, they also considered that individuals with intermediate phenotypes (with orange color and a faded ocellus) could also be males. Davis et al. (1990Davis, W. P., D. S. Taylor & B. J. Turner. 1990. Field observations of the ecology and habits of mangrove rivulus (Rivulus marmoratus ) in Belize and Florida (Teleostei: Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 1: 123-134.) had previously mentioned that it is not possible yet to distinguish primary and secondary males, the latter of which result from sex change of functional hermaphrodites into males due to environmental conditions (Harrington, 1971Harrington, R. W. 1971. How ecological and genetic factors interact to determine when self-fertilizing hermaphrodites of Rivulus marmoratus change into functional secondary males, with a reappraisal of the mode of intersexuality among fishes. Copeia, 1971: 389-432.). Huber (1992Huber, J. H. 1992. Review of Rivulus: ecobiogeography- relationships. Paris, Cybium, Société Française d'Ichtyologie, 572p.) cited the presence of ocellus in secondary males of K . ocellatus , which agrees with our specimen description. However, as discussed above, this report was not afterwards confirmed.
Primary males of Kryptolebias marmoratus can be readily produced in laboratories by inducing eggs to temperatures lower than those usually found in mangrove areas (<20°C), while secondary males can be induced by maintaining individuals at higher temperatures (>30°C), which are similar to those found on populations with higher rate of males (Turner et al., 2006Turner, B. J., M. T. Fisher, D. S. Taylor, W. P. Davis & B. L. Jarrett. 2006. Evolution of 'maleness' and outcrossing in a population of the self-fertilizing killifish, Kryptolebias marmoratus . Evolutionary Ecology Research, 8: 1475-1486.; Earley et al., 2012Earley, R. L., A. F. Hannineb, A. Fuller, M. J. Garcia & E. A. Lee. 2012. Phenotypic plasticity and integration in the mangrove rivulus (Kryptolebias marmoratus ): a prospectus. Integrative and Comparative Biology, 52: 814-827.). Their induction in the lab at naturally occurring temperatures suggests that, in the wild, secondary males may be the most ecologically relevant type of male (Turner et al., 2006Turner, B. J., M. T. Fisher, D. S. Taylor, W. P. Davis & B. L. Jarrett. 2006. Evolution of 'maleness' and outcrossing in a population of the self-fertilizing killifish, Kryptolebias marmoratus . Evolutionary Ecology Research, 8: 1475-1486.). This may also be the case for K. hermaphroditus in northeastern Brazil, where higher temperatures are predominant due to the Equator's line proximity.
Davis et al. (1990Davis, W. P., D. S. Taylor & B. J. Turner. 1990. Field observations of the ecology and habits of mangrove rivulus (Rivulus marmoratus ) in Belize and Florida (Teleostei: Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 1: 123-134.) demonstrated distinct male phenotypes within Kryptolebias marmoratus . One, from Twin Cays, Belize, exhibited a dark orange color pattern, an inconspicuous caudal ocellus, and caudal black margins along the dorsal, anal and caudal fins. The other one, from Florida, USA, exhibited a bright orange coloration almost devoid of dark chromatophores, except the head. Ellison et al. (2012Ellison, A., P. Wright, D. S. Taylor, C. Cooper, K. Regan, S. Currie & S. Consuegra. 2012. Environmental diel variation, parasite loads, and local population structuring of a mixed-mating mangrove fish. Ecology and Evolution, 2: 1682-1695.) also found a male of K. marmoratus with orange coloration, faded ocellus and black borders on the anal and caudal fins in Belize; a pattern similar to that of the K. hermaphroditus male herein described.
Between 2011 and 2015, during extensive surveys at the Ceará-Mirim River estuary (Lira et al., 2015Lira, M. G. S., R. E. C. Paiva, T. P. A. Ramos & S. M. Q. Lima. 2015. First record of Kryptolebias hermaphroditus Costa, 2011 (Cyprinodontiformes: Rivulidae) in the extreme north Atlantic Forest mangroves, Rio Grande do Norte state, Brazil. Check List, 11: 1656.), we collected 107 hermaphrodites before finding this single male. This apparent rarity of males aligns with the extremely low degree of heterozygosis reported for populations of Kryptolebias hermaphroditus from southeastern Brazil (Tartarenkov et al. , 2011Tatarenkov, A., S. M. Q. Lima & J. C. Avise. 2011. Extreme homogeneity and low genetic diversity in Kryptolebias ocellatus from south-eastern Brazil suggest a recent foundation for this androdioecious fish population. Journal of Fish Biology, 79: 2095-2105.). Together with absence of male specimens in all known populations (Costa, 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.; Lira et al. , 2015Lira, M. G. S., R. E. C. Paiva, T. P. A. Ramos & S. M. Q. Lima. 2015. First record of Kryptolebias hermaphroditus Costa, 2011 (Cyprinodontiformes: Rivulidae) in the extreme north Atlantic Forest mangroves, Rio Grande do Norte state, Brazil. Check List, 11: 1656.), these findings suggest that self-fertilizing hermaphroditism is the major mode of reproduction in K . hermaphroditus . However, the persistent occurrence of male individuals in K. marmoratus , but at low densities (varying from 1% in Florida to 10-25% in Belize, Davis et al., 1990Davis, W. P., D. S. Taylor & B. J. Turner. 1990. Field observations of the ecology and habits of mangrove rivulus (Rivulus marmoratus ) in Belize and Florida (Teleostei: Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 1: 123-134.), suggests that sexual outcrossing serves an adaptive role within natural populations, possibly contributing to genetic diversity that helps the species to deal with new environmental pressures, such as parasites loads (Ellison et al., 2011Ellison, A., J. Cable & S. Consuegra. 2011. Best of both worlds? Association between outcrossing and parasite loads in a selfing fish. Evolution, 65: 3021-3026., 2013Ellison, A., J. Jones, C. Inchley & S. Consuegra. 2013. Choosy males could help explain androdioecy in a selfing fish. The American Naturalist, 181: 855-862.).
The occurrence of self-fertilization in Kryptolebias marmoratus has made that species a popular model for population genetics, development, evolutionary biology, behavior, among others (for reviews see Earley et al., 2012Earley, R. L., A. F. Hannineb, A. Fuller, M. J. Garcia & E. A. Lee. 2012. Phenotypic plasticity and integration in the mangrove rivulus (Kryptolebias marmoratus ): a prospectus. Integrative and Comparative Biology, 52: 814-827.; Taylor, 2012Taylor, D. S. 2012. Twenty-four years in the mud: What have we learned about the natural history and ecology of the mangrove rivulus, Kryptolebias marmoratus ? Integrative and Comparative Biology, 52: 724-736.; Avise & Tatarenkov, 2015Avise, J. C. & A. Tatarenkov. 2015. Population genetics and evolution of the mangrove rivulus Kryptolebias marmoratus , the world's only self-fertilizing hermaphroditic vertebrate. Journal of Fish Biology, 87: 519-538.; Turko & Wright, 2015Turko, A. J. & P. A. Wright. 2015. Evolution, ecology and physiology of amphibious killifishes (Cyprinodontiformes). Journal of Fish Biology, 87: 815-835.). Nonetheless, there have been relatively few studies on the South American lineage of K. hermaphroditus , possibly due to its misidentification as K. marmoratus (Seegers, 1984). Additionally, the difficulties of working in mangrove environments, which represent a complex, multi-dimensional, and cryptic habitat with variable environmental and physical features could also contributed to the scarcity of studies on the South American lineage (Taylor, 2012Taylor, D. S. 2012. Twenty-four years in the mud: What have we learned about the natural history and ecology of the mangrove rivulus, Kryptolebias marmoratus ? Integrative and Comparative Biology, 52: 724-736.).
The discovery of a male specimen encourages more studies on Kryptolebias hermaphroditus . While researchers have addressed some aspects of systematics (Costa, 2006Costa, W. J. E. M. 2006. Redescription of Kryptolebias ocellatus (Hensel) and K. caudomarginatus (Seegers) (Teleostei: Cyprinodontiformes: Rivulidae), two killifishes from mangroves of south-eastern Brazil. Aqua Journal of Ichthyology and Aquatic Biology, 11: 5-12., 2011Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.), evolutionary history (Costa et al. , 2010Costa, W. J. E. M., S. M. Q. Lima & R. Bartolette. 2010. Androdioecy in Kryptolebias and the evolution of self-fertilizing hermaphroditism. Biological Journal of the Linnean Society, 99: 344-349.; Tatarenkov et al., 2009Tatarenkov, A., S. M. Q. Lima, D. S. Taylor & J. C. Avise. 2009. Long-term retention of self-fertilization in a fish clade. Proceedings of the National Academy of Sciences, 106: 14456-14459., 2011Tatarenkov, A., S. M. Q. Lima & J. C. Avise. 2011. Extreme homogeneity and low genetic diversity in Kryptolebias ocellatus from south-eastern Brazil suggest a recent foundation for this androdioecious fish population. Journal of Fish Biology, 79: 2095-2105.), and geographic distribution (Sarmento-Soares et al., 2014Sarmento-Soares, L. M., L. F. S. Ingenito, L. F. Duboc, R. F. Martins-Pinheiro, R. Borçato & J. P. Silva. 2014. Primeiros registros de Kryptolebias ocellatus (Hensel) (Cyprinodontiformes, Rivulidae) para riachos de Mata Atlântica no Espírito Santo. Boletim da Sociedade Brasileira de Ictiologia, 111: 15-19.; Lira et al., 2015Lira, M. G. S., R. E. C. Paiva, T. P. A. Ramos & S. M. Q. Lima. 2015. First record of Kryptolebias hermaphroditus Costa, 2011 (Cyprinodontiformes: Rivulidae) in the extreme north Atlantic Forest mangroves, Rio Grande do Norte state, Brazil. Check List, 11: 1656.), there is little available information regarding natural history, ecology and population dynamics. Despite the large gap in our knowledge of the biology of K. hermaphroditus , the recent studies above mentioned, including the present one, suggest some interesting perspectives for studies on evolution, morphology, behavior and physiology across the species' wide geographic distribution in the mangroves of the Brazilian coast.
Acknowledgements
We thank Lucas Medeiros, Mateus Lira and Roney Paiva for helping with fieldwork, and to Luciano Barros-Neto for providing the pictures of the live specimens. Carlos Eduardo Alencar provided information about mangrove crabs in the Ceará-Mirim estuary. Graco Viana and Centro de Treinamento em Aquicultura da Universidade Federal do Rio Grande do Norte provided logistical support. Waldir Berbel-Filho receives a PhD scholarship from Science without Borders Program/CNPq (process #233161/2014-7) and Helder Espírito-Santo received a Postdoctoral fellowship from PDJ Program/CNPq (process #168228/2014-9).
References
- Avise, J. C. & A. Tatarenkov. 2015. Population genetics and evolution of the mangrove rivulus Kryptolebias marmoratus , the world's only self-fertilizing hermaphroditic vertebrate. Journal of Fish Biology, 87: 519-538.
- Costa, W. J. E. M. 1995. Pearl killifishes: the Cynolebiatinae: systematics and biogeography of the Neotropical annual fish subfamily. Neptune city, TFH Publications, 128p.
- Costa, W. J. E. M. 2003. Family Rivulidae. Pp. 526-548. In: Reis, R. E., S. O. Kullander & C. J. Ferraris Jr. (Eds.). Check list of freshwater fishes of South and Central America. Porto Alegre, Edipucrs.
- Costa, W. J. E. M. 2006. Redescription of Kryptolebias ocellatus (Hensel) and K. caudomarginatus (Seegers) (Teleostei: Cyprinodontiformes: Rivulidae), two killifishes from mangroves of south-eastern Brazil. Aqua Journal of Ichthyology and Aquatic Biology, 11: 5-12.
- Costa, W. J. E. M. 2009. Peixes aploqueilóideos da Mata Atlântica brasileira: história, diversidade e conservação. Rio de Janeiro, Museu Nacional. 171p.
- Costa, W. J. E. M. 2011. Identity of Rivulus ocellatus and a new name for a hermaphroditic species of Kryptolebias from southeastern Brazil (Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 22: 185-192.
- Costa, W. J. E. M., S. M. Q. Lima & R. Bartolette. 2010. Androdioecy in Kryptolebias and the evolution of self-fertilizing hermaphroditism. Biological Journal of the Linnean Society, 99: 344-349.
- Davis, W. P., D. S. Taylor & B. J. Turner. 1990. Field observations of the ecology and habits of mangrove rivulus (Rivulus marmoratus ) in Belize and Florida (Teleostei: Cyprinodontiformes: Rivulidae). Ichthyological Exploration of Freshwaters, 1: 123-134.
- Earley, R. L., A. F. Hannineb, A. Fuller, M. J. Garcia & E. A. Lee. 2012. Phenotypic plasticity and integration in the mangrove rivulus (Kryptolebias marmoratus ): a prospectus. Integrative and Comparative Biology, 52: 814-827.
- Ellison, A., J. Cable & S. Consuegra. 2011. Best of both worlds? Association between outcrossing and parasite loads in a selfing fish. Evolution, 65: 3021-3026.
- Ellison, A., P. Wright, D. S. Taylor, C. Cooper, K. Regan, S. Currie & S. Consuegra. 2012. Environmental diel variation, parasite loads, and local population structuring of a mixed-mating mangrove fish. Ecology and Evolution, 2: 1682-1695.
- Ellison, A., J. Jones, C. Inchley & S. Consuegra. 2013. Choosy males could help explain androdioecy in a selfing fish. The American Naturalist, 181: 855-862.
- Harrington, R. W. 1971. How ecological and genetic factors interact to determine when self-fertilizing hermaphrodites of Rivulus marmoratus change into functional secondary males, with a reappraisal of the mode of intersexuality among fishes. Copeia, 1971: 389-432.
- Huber, J. H. 1992. Review of Rivulus: ecobiogeography- relationships. Paris, Cybium, Société Française d'Ichtyologie, 572p.
- Lira, M. G. S., R. E. C. Paiva, T. P. A. Ramos & S. M. Q. Lima. 2015. First record of Kryptolebias hermaphroditus Costa, 2011 (Cyprinodontiformes: Rivulidae) in the extreme north Atlantic Forest mangroves, Rio Grande do Norte state, Brazil. Check List, 11: 1656.
- Sarmento-Soares, L. M., L. F. S. Ingenito, L. F. Duboc, R. F. Martins-Pinheiro, R. Borçato & J. P. Silva. 2014. Primeiros registros de Kryptolebias ocellatus (Hensel) (Cyprinodontiformes, Rivulidae) para riachos de Mata Atlântica no Espírito Santo. Boletim da Sociedade Brasileira de Ictiologia, 111: 15-19.
- Soto, C. G. & D. L. G. Noakes. 1994. Coloration and gender in the hermaphroditic fish Rivulus marmoratus Poey (Teleostei: Rivulidae). Ichthyological Exploration of Freshwaters, 5: 79-90.
- Tatarenkov, A., S. M. Q. Lima, D. S. Taylor & J. C. Avise. 2009. Long-term retention of self-fertilization in a fish clade. Proceedings of the National Academy of Sciences, 106: 14456-14459.
- Tatarenkov, A., S. M. Q. Lima & J. C. Avise. 2011. Extreme homogeneity and low genetic diversity in Kryptolebias ocellatus from south-eastern Brazil suggest a recent foundation for this androdioecious fish population. Journal of Fish Biology, 79: 2095-2105.
- Taylor, D. S. 2012. Twenty-four years in the mud: What have we learned about the natural history and ecology of the mangrove rivulus, Kryptolebias marmoratus ? Integrative and Comparative Biology, 52: 724-736.
- Turko, A. J. & P. A. Wright. 2015. Evolution, ecology and physiology of amphibious killifishes (Cyprinodontiformes). Journal of Fish Biology, 87: 815-835.
- Turner, B. J., M. T. Fisher, D. S. Taylor, W. P. Davis & B. L. Jarrett. 2006. Evolution of 'maleness' and outcrossing in a population of the self-fertilizing killifish, Kryptolebias marmoratus . Evolutionary Ecology Research, 8: 1475-1486.
Publication Dates
-
Publication in this collection
2016
History
-
Received
06 Mar 2016 -
Accepted
18 Aug 2016