Abstract
The development of biomaterials capable of driving dental pulp stem cell differentiation into odontoblast-like cells able to secrete reparative dentin is the goal of current conservative dentistry. In the present investigation, a biomembrane (BM) composed of a chitosan/collagen matrix embedded with calcium-aluminate microparticles was tested. The BM was produced by mixing collagen gel with a chitosan solution (2:1), and then adding bioactive calcium-aluminate cement as the mineral phase. An inert material (polystyrene) was used as the negative control. Human dental pulp cells were seeded onto the surface of certain materials, and the cytocompatibility was evaluated by cell proliferation and cell morphology, assessed after 1, 7, 14 and 28 days in culture. The odontoblastic differentiation was evaluated by measuring alkaline phosphatase (ALP) activity, total protein production, gene expression of DMP-1/DSPP and mineralized nodule deposition. The pulp cells were able to attach onto the BM surface and spread, displaying a faster proliferative rate at initial periods than that of the control cells. The BM also acted on the cells to induce more intense ALP activity, protein production at 14 days, and higher gene expression of DSPP and DMP-1 at 28 days, leading to the deposition of about five times more mineralized matrix than the cells in the control group. Therefore, the experimental biomembrane induced the differentiation of pulp cells into odontoblast-like cells featuring a highly secretory phenotype. This innovative bioactive material can drive other protocols for dental pulp exposure treatment by inducing the regeneration of dentin tissue mediated by resident cells.
Dental Pulp; Biocompatible Materials; Tissue Engineering; Stem Cells; Dentin
Introduction
The regeneration of dentin by the induction of odontoblastic differentiation from dental pulp stem cells (DPSCs) and the up-regulation of tertiary dentin deposition is the purpose of direct pulp-capping (DPC) therapy.11. Sangwan P, Sangwan A, Duhan J, Rohilla A. Tertiary dentinogenesis with calcium hydroxide: a review of proposed mechanisms. Int Endod J. 2013;46(1):3-19. doi:10.1111/j.1365-2591.2012.02101.x The materials clinically available for such purposes are based on calcium hydroxide (CH) and mineral trioxide aggregate (MTA). Both materials share the same pathway for dentin bridge deposition, by releasing high quantities of calcium ions (Ca++), thus creating an alkaline environment (pH 9.0–12.0).11. Sangwan P, Sangwan A, Duhan J, Rohilla A. Tertiary dentinogenesis with calcium hydroxide: a review of proposed mechanisms. Int Endod J. 2013;46(1):3-19. doi:10.1111/j.1365-2591.2012.02101.x,22. Briso AL, Rahal V, Mestrener SR, Dezan Junior E. Biological response of pulps submitted to different capping materials. Braz Oral Res. 2006;20(3):219-25. doi:10.1590/S1806-83242006000300007 However, this high pH causes a superficial burn on pulp tissue, leading to an area of coagulation necrosis associated with subjacent inflammation, tissue disorganization, and hemorrhage on surrounding pulp tissue.33. Costa CAS, Oliveira MF, Giro EM, Hebling J. Biocompatibility of resin-based materials used as pulp-capping agents. Int Endod J. 2003;36(12):831-9. doi:10.1111/j.1365-2591.2003.00702.x,44. Albuquerque DS, Gominho LF, Santos RA. Histologic evaluation of pulpotomy performed with ethyl-cyanoacrylate and calcium hydroxide. Braz Oral Res. 2006;20(3):226-30. doi:10.1590/S1806-83242006000300008,55. Cavalcanti BN, Rode SM, França CM, Marques MM. Pulp capping materials exert an effect on the secretion of IL-1β and IL-8 by migrating human neutrophils. Braz Oral Res. 2011;25(1):13-8. doi:10.1590/S1806-83242011000100003 Only after this inflammation is resolved, one to two weeks thereafter, can migration of DPSCs to the necrotic area, followed by cytodifferentiation into odontoblast-like cells, be observed. Dentin-like tissue with some tubular structure is observed after 4 weeks.11. Sangwan P, Sangwan A, Duhan J, Rohilla A. Tertiary dentinogenesis with calcium hydroxide: a review of proposed mechanisms. Int Endod J. 2013;46(1):3-19. doi:10.1111/j.1365-2591.2012.02101.x,22. Briso AL, Rahal V, Mestrener SR, Dezan Junior E. Biological response of pulps submitted to different capping materials. Braz Oral Res. 2006;20(3):219-25. doi:10.1590/S1806-83242006000300007,44. Albuquerque DS, Gominho LF, Santos RA. Histologic evaluation of pulpotomy performed with ethyl-cyanoacrylate and calcium hydroxide. Braz Oral Res. 2006;20(3):226-30. doi:10.1590/S1806-83242006000300008,55. Cavalcanti BN, Rode SM, França CM, Marques MM. Pulp capping materials exert an effect on the secretion of IL-1β and IL-8 by migrating human neutrophils. Braz Oral Res. 2011;25(1):13-8. doi:10.1590/S1806-83242011000100003 Therefore, this therapy cannot be considered biocompatible with the pulp tissue, since it causes the death of numerous cells, including stem cells, which are essential for the regenerative potential of pulp tissue.66. Costa CAS, Hebling J, Scheffel DLS, Soares DGS, Basso FG, Ribeiro APD. Methods to evaluate and strategies to improve the biocompatibility of dental materials and operative techniques. Dent Mater. 2014;30(7):769-84. doi:10.1016/j.dental.2014.04.010 Although some authors claim that Ca++ ions released from these materials play a major role in the regenerative events that occur after direct pulp-capping,11. Sangwan P, Sangwan A, Duhan J, Rohilla A. Tertiary dentinogenesis with calcium hydroxide: a review of proposed mechanisms. Int Endod J. 2013;46(1):3-19. doi:10.1111/j.1365-2591.2012.02101.x this effect has been mainly attributed to the inherent regenerative potential of pulp tissue.66. Costa CAS, Hebling J, Scheffel DLS, Soares DGS, Basso FG, Ribeiro APD. Methods to evaluate and strategies to improve the biocompatibility of dental materials and operative techniques. Dent Mater. 2014;30(7):769-84. doi:10.1016/j.dental.2014.04.010,77. Schmalz G, Smith AJ. Pulp development, repair, and regeneration: challenges of the transition from traditional dentistry to biologically based therapies. J Endod. 2014;40(4 Suppl):S2-5. doi:10.1016/j.joen.2014.01.018
Biomembranes for guided tissue regeneration have emerged as an interesting alternative for directing the regeneration of mineralized tissues, such as bone and dentin, by inducing the following events: (1) migration and adhesion of target cells to the surface of the membrane; (2) differentiation/maturation into secretive cells; and (3) induction of new tissue deposition.88. Zheng Z, Wei Y, Wang G, Gong Y, Zhang X. Surface characterization and cytocompatibility of three chitosan/polycation composite membranes for guided bone regeneration. J Biomater Appl. 2009;24(3):209-29. doi:10.1177/0885328208095825,99. Li F, Liu X, Zhao S, Wu H, Xu HH. Porous chitosan bilayer membrane containing TGF-β1 loaded microspheres for pulp capping and reparative dentin formation in a dog model. Dent Mater. 2014;30(2):172-81. doi:10.1016/j.dental.2013.11.005 Calcium-rich chitosan/collagen composite has been extensively studied regarding its tissue engineering applications, since this material provides a substrate similar to the bone/dentin extracellular matrix, promoting cell adhesion and proliferation, and supporting mesenchymal stem cell function, thus increasing osteogenic/odontogenic differentiation and mineralized matrix deposition capabilities of precursor bone.1010. Kim IY, Seo SJ, Moon HS, Yoo MK, Park IY, Kim BC, et al. Chitosan and its derivatives for tissue engineering applications. Biotechnol Adv. 2008;26(1):1-21. doi:10.1016/j.biotechadv.2007.07.009,1111. Venkatesan J, Pallela R, Bhatnagar I, Kim SK. Chitosan-amylopectin/hydroxyapatite and chitosan-chondroitin sulphate/hydroxyapatite composite scaffolds for bone tissue engineering. Int J Biol Macromol. 2012;51(5):1033-42. doi:10.1016/j.ijbiomac.2012.08.020,1212. Liu H, Peng H, Wu Y, Zhang C, Cai Y, Xu G, et al. The promotion of bone regeneration by nanofibrous hydroxyapatite/chitosan scaffolds by effects on integrin-BMP/Smad signaling pathway in BMSCs. Biomaterials. 2013;34(18):4404-17. doi:10.1016/j.biomaterials.2013.02.048,1313. Qiao P, Wang J, Xie Q, Li F, Dong L, Xu T. Injectable calcium phosphate-alginate-chitosan microencapsulated MC3T3-E1 cell paste for bone tissue engineering in vivo. Mater Sci Eng C. 2013;33(8):4633-9. doi:10.1016/j.msec.2013.07.022,1414. Shen S, Fu D, Xu F, Long T, Hong F, Wang J. The design and features of apatite-coated chitosan microspheres as injectable scaffold for bone tissue engineering. Biomed Mater. 2013;8(2):025007. doi:10.1088/1748-6041/8/2/025007 Recently, researchers have found calcium-aluminate to be a promising mineral phase for both dentin and bone regeneration, since several levels of Ca++ release and neutral pH may be achieved, depending on the production route, which has a direct effect on cell differentiation.1515. Castro-Raucci LMS, Oliveira IR, Teixeira LN, Rosa AL, Oliveira PT, Jacobovitz M. Effects of a novel calcium aluminate cement on the early events of the progression of osteogenic cell cultures. Braz Dent J. 2011;22(2):99-104. doi:10.1590/S0103-64402011000200002,1616. Aguilar FG, Garcia LFR, Pires-de-Souza FCP. Biocompatibility of new calcium aluminate cement (EndoBinder). J Endod. 2012;38(3):367-71. doi:10.1016/j.joen.2011.11.002,1717. Pires-de-Souza FCP, Morães PC, Garcia LFR, Aguilar FG, Watanabe E. Evaluation of pH, calcium ion release and antimicrobial activity of a new calcium aluminate cement. Braz Oral Res. 2013;27(4):324-30. doi:10.1590/S1806-83242013000400006,1818. Garcia LFR, Huck C, Oliveira LM, Souza PPC, Costa CAS. Biocompatibility of new calcium aluminate cement: tissue reaction and expression of inflammatory mediators and cytokines. J Endod. 2014;40(12):2024-9. doi:10.1016/j.joen.2014.08.015,1919. Silva EJNL, Herrera DR, Rosa TP, Duque TM, Jacinto RC, Gomes BPFA, et al. Evaluation of cytotoxicity, antimicrobial activity and physicochemical properties of a calcium aluminate-based endodontic material. J Appl Oral Sci. 2014;22(1):61-7. doi: 10.1590/1678-775720130031 Therefore, human dental pulp cells were used in the present investigation to assess the dentinogenic potential of a novel biomembrane composed of chitosan and collagen containing calcium-aluminate as a mineral phase.
Methodology
Biomembrane production: A solution of chitosan (75–85% deacetylated, animal origin; Sigma-Aldrich, St. Louis, USA) in acetic acid 2% w/v (Sigma-Aldrich) was obtained and mixed with a collagen gel suspension from bovine tendon, in a 1:2 ratio. Then, the calcium-aluminate phase was added, composed of 65% CaO.Al2O3, 25% CaO.2Al2O3 and 10% 12CaO.7Al2O3. The mixture was poured onto a polytetrafluoroethylene surface and dried in a desiccator under vacuum. The material was then compacted and dried uniformly, yielding a flat biomembrane. The superficial morphology, as well as the presence of calcium-aluminate, was detected by scanning electron microscopy (SEM; 10.0 kV) and by energy-dispersive x-ray spectroscopy (EDS; 25 kV; take-off angle 3.7) (JEOL-JMS-6610V Scanning Microscope, Tokyo, Japan). The sizes of the mineral particles were measured with ImageJ software (National Institutes of Health, Bethesda, USA). Prior to in vitro testing, the BM was sterilized by gamma radiation (CBE, Cotia, Brazil) with the irradiation dose fixed at 20 kilogray.
Cell culture: A primary culture of human dental pulp cells (HDPCs) was obtained by enzymatic digestion of the pulp tissue from third molars, as previously described.2020. Soares DG, Basso FG, Scheffel DS, Hebling J, Costa CAS. Responses of human dental pulp cells after application of a low-concentration bleaching gel to enamel. Arch Oral Biol. 2015;60(9):1428-36. doi:10.1016/j.archoralbio.2015.06.014 The teeth were obtained from volunteers who signed informed consent documents (Proc. no. 30939314.5.0000.5416; Research Ethics Committee, Araraquara School of Dentistry, Brazil). The cells were cultured in DMEM (Dulbecco’s Modified Eagle’s Medium; Sigma-Aldrich) containing 10% Fetal Bovine Serum (FBS; GIBCO, Grand Island, USA), 100 IU/mL and 100 µg/mL of penicillin and streptomycin, respectively, and 2 mmol/L of glutamine (GIBCO). The biomembrane was adapted to a metallic device between two silicon rings, to keep it in position and distant from the bottom of the culture plate. A 3 x 104 quantity of cells were seeded onto the biomembrane (BM). After 24 h, the culture medium was replaced by osteogenic medium (complete DMEM + 10% FBS + 10 nmol/L β-glycerophosphate + 50 µg/mL sodium ascorbate; Sigma), and the cells were cultivated for up to 28 days (the medium was changed every 48 h). Cells seeded on an inert surface (polystyrene) were used as a control (NC group).
Cell viability/proliferation and morphology: Cell viability was assessed by the Alamar Blue assay after 1, 7, 14, and 28 days in culture. The cells were incubated with DMEM plus Alamar Blue dye (10:1) at each time point (n = 6), for 4 h at 37°C, and the fluorescence intensity was monitored at 530 nm excitation and 590 nm emission (Synergy H1, BioTek, Winooski, USA). The cell viability was calculated considering the NC group as 100% of cell viability. The increase/decrease in the cell viability percentage was used for indirect determination of cell proliferation throughout the periods of analysis. The cell morphology was observed by SEM (n = 2), at the same time points, as previously described.2121. Couve E, Osorio R, Schmachtenberg O. The amazing odontoblast: activity, autophagy, and aging. J Dent Res. 2013;92(9):765-72. doi:10.1177/0022034513495874
Alkaline phosphatase (ALP) activity/total protein (TP) production: Analysis of ALP activity and Total Protein (TP) (n = 6) were performed on cells in culture, at the 14-day time point, by a colorimetric end-point assay (Labtest Diagnóstico S.A., Lagoa Santa, Brazil), with a thymolphthalein monophosphate substrate, and Read and North protocol, as described in detail by Soares et al.2020. Soares DG, Basso FG, Scheffel DS, Hebling J, Costa CAS. Responses of human dental pulp cells after application of a low-concentration bleaching gel to enamel. Arch Oral Biol. 2015;60(9):1428-36. doi:10.1016/j.archoralbio.2015.06.014 The ALP activity was normalized by the TP value (mg/mL), and the percentages of both parameters were calculated, considering the NC group as 100% of cell activity.
Gene expression of odontoblastic markers: The gene expression was assessed by real-time polymerase chain reaction (PCR) after 28 days of cell culture (n = 4). Total RNA was extracted with an RNAqueous®, Micro Kit (Ambion, Austin, USA), and 1 µg was then reverse-transcribed into single-stranded cDNA with a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, USA), in accordance with the manufacturer’s protocols. Taqman assays (Invitrogen, Grand Island, USA) were used for relative quantification of mRNA levels, as follows: DMP-1, Hs01009391_g1; DSPP, Hs00171962_m1; and β-actin, 4333762T. The CT values for each sample were normalized by the control (NC group).
Mineralized matrix deposition: After 28 days in culture, the cell/material constructs (n = 6) were fixed with cold 70% ethanol and stained with alizarin red dye (40 mM, pH 4.2; Sigma-Aldrich).2020. Soares DG, Basso FG, Scheffel DS, Hebling J, Costa CAS. Responses of human dental pulp cells after application of a low-concentration bleaching gel to enamel. Arch Oral Biol. 2015;60(9):1428-36. doi:10.1016/j.archoralbio.2015.06.014 A BM control group with no cells (n = 6) was used to normalize the values obtained from the BM group (with cells), since the material itself contains calcium. The materials were subjected to five 10-minute washes in deionized water under shaking for the removal of excess stain. Then, the materials were incubated with 10% cetylpyridinium chloride (Sigma-Aldrich) under shaking for 30 min, and the absorbance of the resulting solution was measured at 570 nm (Synergy H1). Average absorbance of the NC group was considered as 100% of alizarin red staining.
Statistical analysis: The experiments were performed twice to ensure reproducibility. The data were analyzed for cell viability, by two-way ANOVA, complemented by Tukey’s test. The data for ALP activity, TP production, gene expression, and the alizarin red assay were subjected to Student’s t-test for observation of significant differences between the study groups. A value of p < 0.05 was considered statistically significant.
Results
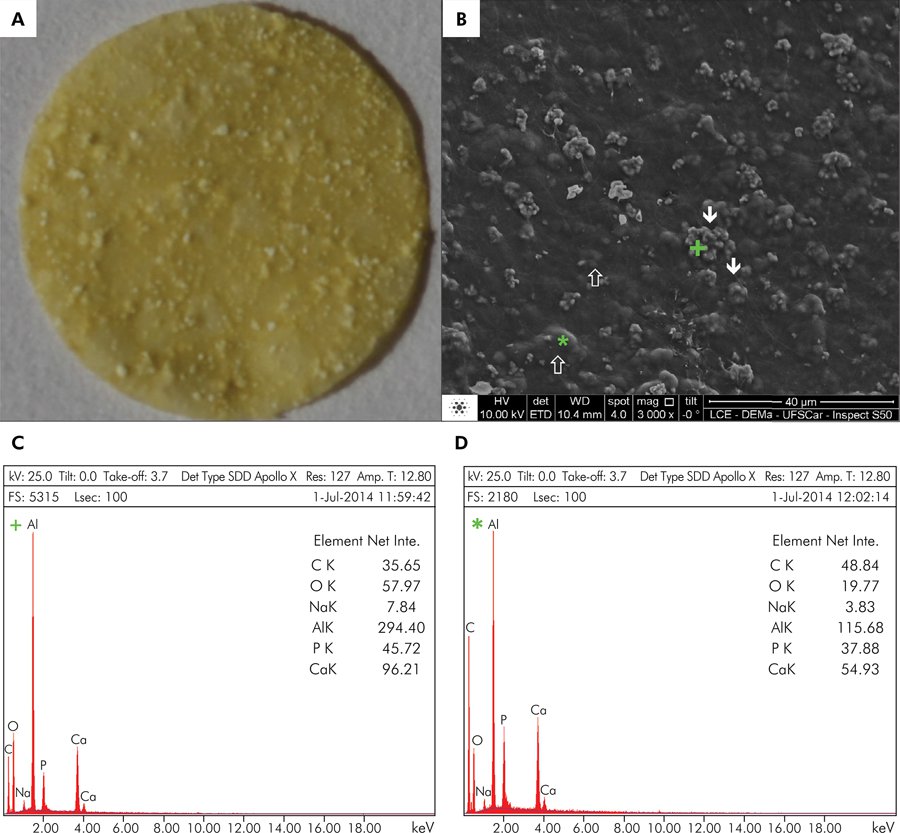
Biomembrane production: A 0.5-mm-thick membrane interlaced with mineral components was obtained (Figure 1A). In the SEM analysis, the mineral particles were observed on the material surface and sub-surface (Figure 1B). The EDS analysis of superficial mineral structures demonstrated the presence of Ca (calcium), Al (aluminum), and O (oxygen), the main components of the calcium-aluminate phase (Figure 1C). This mineral phase was organized into microparticle agglomerates, with single microparticles measuring from 0.48 to 1.47 µm (average = 0.95 µm ± 0.30) and agglomerates measuring from 2.53 to 9.71 µm (average = 4.98 µm ± 2.42). In the sub-superficial EDS analysis, the main components detected were C (carbon), Ca, Al, and O, which are present in the chitosan-collagen organic matrix, and in calcium-aluminate (Figure 1D). In both analyses, the authors detected the presence of Na (sodium) and P (phosphorus), components of the washing solutions used in BM production.
Biomembrane (BM) characterization. (A) Digital photograph of the BM. A flat membrane covered by mineralized components was obtained. (B) SEM 3,000x. The membrane was covered by mineralized microparticles (≈ 0.95 µm ± 0.30) organized into agglomerates (≈ 4.98 µm ± 2.42) randomly distributed onto its surface (↓) and sub-surface (⇑). (C) EDS analysis of superficial microparticles. Al, Ca, and O were the main components of the superficial (+) area. (D) EDS analysis of sub-superficial microparticles. C, Al, Ca, and O were the main components of the sub-superficial (*) area. Net Inte.: net intensity.
Cell viability/morphology: When each material was evaluated at the established time points, a significant increase in cell viability percentage was observed; this indirectly demonstrated significant cell proliferation through time in both materials (Figure 2). A significantly higher cell viability value was observed for the BM group at the 7-day period, as compared with the NC group. No significant difference between the groups was observed for the other time points. The SEM images showed cells covering the surface of both materials. A mineral-like structure was observed on the surface of the BM group cells at the 28-day period (Figure 3).
Cell proliferation and cell morphology. Percentage of cell viability for NC and BM groups at all the time points. ο Indicates a significant difference for the NC group among the time points (two-way ANOVA; Tukey’s test; p < 0.05).
Representative images of SEM analysis for the groups at 1 and 28 days, respectively (SEM 1,000x). *Indicates the material surface. ⇓ Indicates the presence of a mineral-like structure covering the cells.
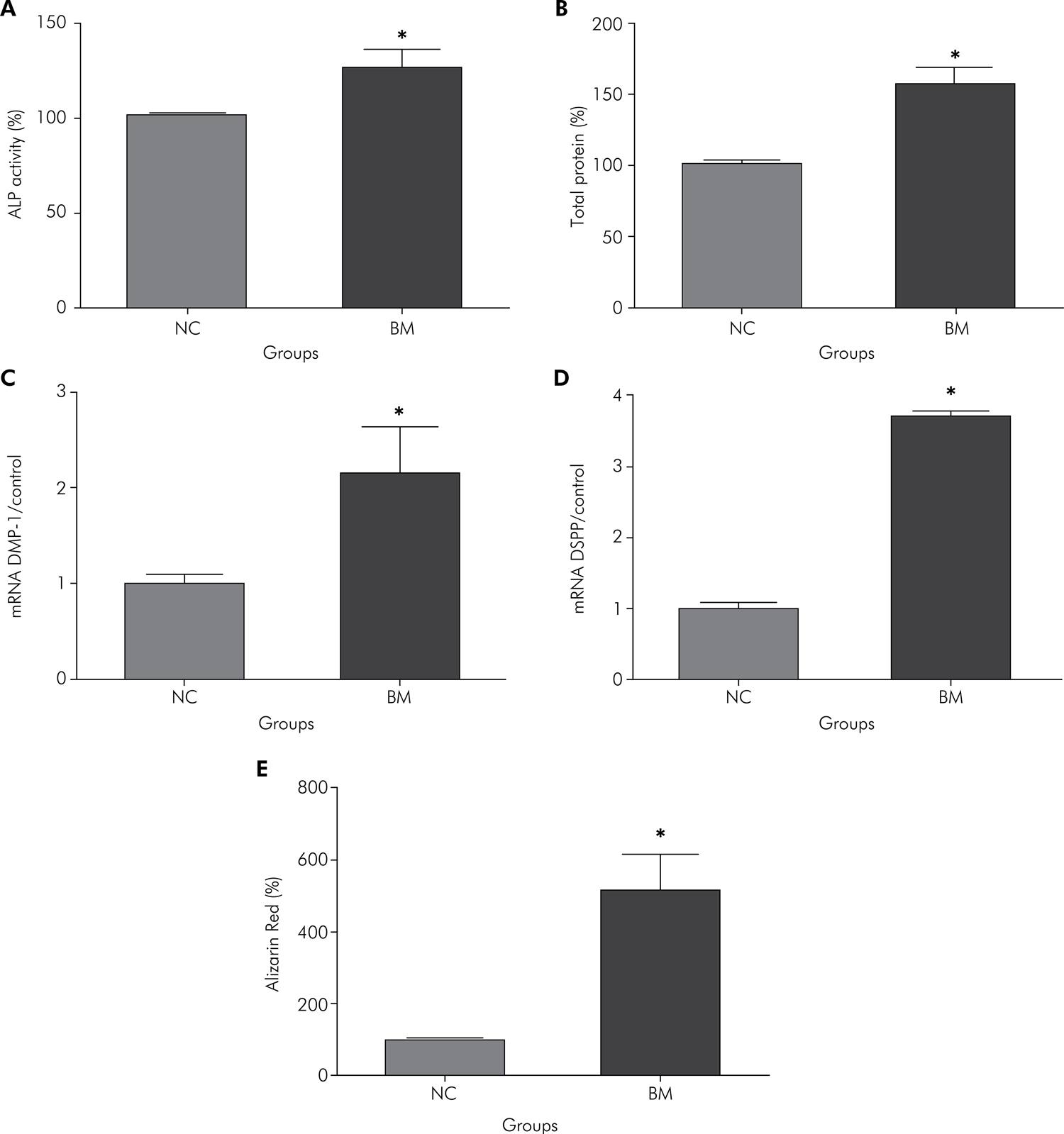
Odontoblastic marker expression: Considering the control group as 100% of ALP activity/TP production, the cells in the BM group showed a significant increase in both parameters in relation to those in the control group, namely about 26.4% for ALP activity and 57.2% for TP production. These data are displayed in Figures 4A and 4B. The cells of both groups presented DMP-1 and DSPP expression, which characterized their differentiation into odontoblast-like cells. However, significantly higher DMP-1 and DSPP mRNA were observed for the BM group, compared with the NC group (Figures 4C and 4D). The mineralized matrix deposition was also significantly higher for the BM group in comparison with the control group (Figure 4E).
Percentages of ALP activity (A) and TP production (B) at 14 days, and of mRNA gene expression of DMP-1 (C) and DSPP (D) at 28 days, and Alizarin Red Assay (E) for NC and BM groups. *Indicates statistically significant difference between the groups (Student’s t-test; p < 0.05).
Discussion
The ideal dental material for use as direct pulp-capping therapy should be able to reproduce tooth developmental stages, ultimately with dentin deposition.11. Sangwan P, Sangwan A, Duhan J, Rohilla A. Tertiary dentinogenesis with calcium hydroxide: a review of proposed mechanisms. Int Endod J. 2013;46(1):3-19. doi:10.1111/j.1365-2591.2012.02101.x,66. Costa CAS, Hebling J, Scheffel DLS, Soares DGS, Basso FG, Ribeiro APD. Methods to evaluate and strategies to improve the biocompatibility of dental materials and operative techniques. Dent Mater. 2014;30(7):769-84. doi:10.1016/j.dental.2014.04.010,2222. Goldberg M, Smith AJ. Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med. 2004;15(1):13-27. doi:10.1177/154411130401500103 During this process, cells from the central areas of the dental papillae migrate to the basement membrane and undergo terminal differentiation into odontoblasts, followed by the deposition of a collagen-rich matrix, giving rise to pre-dentin.2121. Couve E, Osorio R, Schmachtenberg O. The amazing odontoblast: activity, autophagy, and aging. J Dent Res. 2013;92(9):765-72. doi:10.1177/0022034513495874,2323. Fouad AF, Verma P. Healing after regenerative procedures with and without pulpal infection. J Endod. 2014;40(4 Suppl):S58-64. doi:10.1016/j.joen.2014.01.022 The following steps include the expression of non-collagenous proteins and growth factors at the mineralization front, among other components, culminating in dentin mineralization.2222. Goldberg M, Smith AJ. Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med. 2004;15(1):13-27. doi:10.1177/154411130401500103 This process is constantly activated in dental pulp tissue, since odontoblasts are incapable of undergoing mitosis. When these cells are lost, a new generation of odontoblast-like cells capable of secreting a dentin matrix differentiates from progenitor cells within the pulp.77. Schmalz G, Smith AJ. Pulp development, repair, and regeneration: challenges of the transition from traditional dentistry to biologically based therapies. J Endod. 2014;40(4 Suppl):S2-5. doi:10.1016/j.joen.2014.01.018,2121. Couve E, Osorio R, Schmachtenberg O. The amazing odontoblast: activity, autophagy, and aging. J Dent Res. 2013;92(9):765-72. doi:10.1177/0022034513495874,2323. Fouad AF, Verma P. Healing after regenerative procedures with and without pulpal infection. J Endod. 2014;40(4 Suppl):S58-64. doi:10.1016/j.joen.2014.01.022,2424. Kotsanos N, Arapostathis KN, Arhakis A, Menexes G. Direct pulp capping of carious primary molars. A specialty practice based study. J Clin Pediatr Dent. 2014;38(4):307-12. doi:10.17796/jcpd.38.4.k651814611318430 However, the materials currently used for direct pulp-capping are known to cause a pulp scar tissue, leading to the death of several progenitor cells in Höhl’s layer, and triggering an inflammatory reaction in the surrounding tissue.22. Briso AL, Rahal V, Mestrener SR, Dezan Junior E. Biological response of pulps submitted to different capping materials. Braz Oral Res. 2006;20(3):219-25. doi:10.1590/S1806-83242006000300007,33. Costa CAS, Oliveira MF, Giro EM, Hebling J. Biocompatibility of resin-based materials used as pulp-capping agents. Int Endod J. 2003;36(12):831-9. doi:10.1111/j.1365-2591.2003.00702.x,44. Albuquerque DS, Gominho LF, Santos RA. Histologic evaluation of pulpotomy performed with ethyl-cyanoacrylate and calcium hydroxide. Braz Oral Res. 2006;20(3):226-30. doi:10.1590/S1806-83242006000300008,55. Cavalcanti BN, Rode SM, França CM, Marques MM. Pulp capping materials exert an effect on the secretion of IL-1β and IL-8 by migrating human neutrophils. Braz Oral Res. 2011;25(1):13-8. doi:10.1590/S1806-83242011000100003 This aggression may be enough to cause impaired regenerative potential and loss of pulp vitality, depending on the previous condition of the pulp tissue.2525. Cho SY, Seo DG, Lee SJ, Lee J, Lee SJ, Jung IY. Prognostic factors for clinical outcomes according to time after direct pulp capping. J Endod. 2013;39(3):327-31. doi:10.1016/j.joen.2012.11.034,2626. Mente J, Hufnagel S, Leo M, Michel A, Gehrig H, Panagidis D, et al. Treatment outcome of mineral trioxide aggregate or calcium hydroxide direct pulp capping: long-term results. J Endod. 2014;40(11):1746-51. doi:10.1016/j.joen.2014.07.019 Moreover, the loss of progenitor cells during this process has an aging effect that may reduce the regenerative capability of the pulp to withstand future challenges.66. Costa CAS, Hebling J, Scheffel DLS, Soares DGS, Basso FG, Ribeiro APD. Methods to evaluate and strategies to improve the biocompatibility of dental materials and operative techniques. Dent Mater. 2014;30(7):769-84. doi:10.1016/j.dental.2014.04.010 Therefore, the development of biomaterials that facilitate pulp regeneration of the remaining vital pulp tissues is desired to promote dental pulp self-repair, and regenerate dentin on exposed pulp.2727. Colombo JS, Moore AN, Hartgerink JD, D’Souza RN. Scaffolds to control inflammation and facilitate dental pulp regeneration. J Endod. 2014;40(4 Suppl):S6-12. doi:10.1016/j.joen.2014.01.019,2828. Ma PX. Biomimetic materials for tissue engineering. Adv Drug Deliv Rev. 2008;60(2):184-98. doi:10.1016/j.addr.2007.08.041
Ideally, the material for dentin regeneration should mimic the natural environment.2929. Goldberg M, Kulkarni AB, Young M, Boskey A. Dentin: structure, composition and mineralization. Front Biosci (Elite Ed.). 2011 Jan 1;3:711-35. doi:10.2741/e281 The pre-dentin extracellular matrix is composed of 90% collagen, with the other 10% representing non-collagenous proteins, enzymes, and growth factors. The glycosaminoglycans (GACs) and proteoglycans are the second major component of the pre-dentin matrix.2222. Goldberg M, Smith AJ. Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med. 2004;15(1):13-27. doi:10.1177/154411130401500103,3030. Goldberg M, Takagi M. Dentine proteoglycans: composition, ultrastructure and functions. Histochem J. 1993;25(11):781-806. doi:10.1007/BF02388111,3131. Embery G, Hall R, Waddington R, Septier D, Goldberg M. Proteoglycans in dentinogenesis. Crit Rev Oral Biol Med. 2001;12(4):331-49. doi:10.1177/10454411010120040401 Chitosan is a natural biopolymer composed of chitin, a long-chain polysaccharide of N-acetylglucosamine, displaying a structure very similar to that of GAGs.3232. Venkatesan J, Kim SK. Chitosan composites for bone tissue engineering – an overview. Mar Drugs. 2010;8(8):2252-66. doi:10.3390/md8082252 The addition of collagen to the chitosan matrix allows for a composition more similar to that of the pre-dentin extracellular matrix, and provides the Arg-Gly-Asp (RGD) sequence required to promote cell migration and adhesion,3333. Sayin E, Baran ET, Hasirci V. Protein-based materials in load-bearing tissue-engineering applications. Regen Med. 2014;9(5):687-701. doi:10.2217/rme.14.52 mimicking the natural basement membrane.2222. Goldberg M, Smith AJ. Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med. 2004;15(1):13-27. doi:10.1177/154411130401500103 In the present study, a chitosan-collagen calcium-aluminate biomembrane was developed to simulate dentin composition. Calcium-aluminate in contact with a wet environment leads to the formation of the AH3 (Al2O3.3H2O) and C3AH6 (3CaO.Al2O3.6H2O) phases, associated with the release of low amounts of Ca++, which maintains pH levels similar to those of human body tissues,1717. Pires-de-Souza FCP, Morães PC, Garcia LFR, Aguilar FG, Watanabe E. Evaluation of pH, calcium ion release and antimicrobial activity of a new calcium aluminate cement. Braz Oral Res. 2013;27(4):324-30. doi:10.1590/S1806-83242013000400006 and leads to the formation of a carbonated apatite phase.3434. Oliveira IR, Andrade TL, Jacobovitz M, Pandolfelli VC. Bioactivity of calcium aluminate endodontic cement. J Endod. 2013;39(6):774-8. doi:10.1016/j.joen.2013.01.013 The biocompatibility of this material, reported previously,1616. Aguilar FG, Garcia LFR, Pires-de-Souza FCP. Biocompatibility of new calcium aluminate cement (EndoBinder). J Endod. 2012;38(3):367-71. doi:10.1016/j.joen.2011.11.002,1818. Garcia LFR, Huck C, Oliveira LM, Souza PPC, Costa CAS. Biocompatibility of new calcium aluminate cement: tissue reaction and expression of inflammatory mediators and cytokines. J Endod. 2014;40(12):2024-9. doi:10.1016/j.joen.2014.08.015 seems to be related to the slightly alkaline pH level (about 8.0) achieved after its application to the connective tissues.1717. Pires-de-Souza FCP, Morães PC, Garcia LFR, Aguilar FG, Watanabe E. Evaluation of pH, calcium ion release and antimicrobial activity of a new calcium aluminate cement. Braz Oral Res. 2013;27(4):324-30. doi:10.1590/S1806-83242013000400006,1919. Silva EJNL, Herrera DR, Rosa TP, Duque TM, Jacinto RC, Gomes BPFA, et al. Evaluation of cytotoxicity, antimicrobial activity and physicochemical properties of a calcium aluminate-based endodontic material. J Appl Oral Sci. 2014;22(1):61-7. doi: 10.1590/1678-775720130031
According to the results, the pulp cells seeded onto the BM presented faster proliferative capability at the initial periods, in comparison with those of the NC group. This rapid cell proliferation may be attributed to the BM composition, since it has been found that calcium-aluminate1515. Castro-Raucci LMS, Oliveira IR, Teixeira LN, Rosa AL, Oliveira PT, Jacobovitz M. Effects of a novel calcium aluminate cement on the early events of the progression of osteogenic cell cultures. Braz Dent J. 2011;22(2):99-104. doi:10.1590/S0103-64402011000200002,1919. Silva EJNL, Herrera DR, Rosa TP, Duque TM, Jacinto RC, Gomes BPFA, et al. Evaluation of cytotoxicity, antimicrobial activity and physicochemical properties of a calcium aluminate-based endodontic material. J Appl Oral Sci. 2014;22(1):61-7. doi: 10.1590/1678-775720130031 and chitosan3535. Amir LR, Suniarti DF, Utami S, Abbas B. Chitosan as a potential osteogenic factor compared with dexamethasone in cultured macaque dental pulp stromal cells. Cell Tissue Res. 2014;358(2):407-15. doi:10.1007/s00441-014-1938-1 increase the proliferation of cultured cells. Additionally, the RGD sequences in the collagen fibrils may have played a role to promote faster DPSC proliferation. Galler et al.3636. Galler KM, Aulisa L, Regan KR, D’Souza RN, Hartgerink JD. Self-assembling multidomain peptide hydrogels: designed susceptibility to enzymatic cleavage allows enhanced cell migration and spreading. J Am Chem Soc. 2010;132(9):3217-23. doi:10.1021/ja910481t demonstrated that the addition of RGD sequences in hydrogels significantly increased DPSC proliferation. According to these authors, cell adhesion to tissue-engineered biomaterials is a prerequisite for cell proliferation. At later periods, such as 14 and 28 days, no significant difference was observed for cell proliferation between the BM and NC groups. This result may be a consequence of the inverse relationship between proliferation and differentiation. When cells are more proliferative, their differentiation declines, and vice versa.3737. Zhang Y, Wang Y, Shi B, Cheng X. A platelet-derived growth factor releasing chitosan/coral composite scaffold for periodontal tissue engineering. Biomaterials. 2007;28(8):1515-22. doi:10.1016/j.biomaterials.2006.11.040 At 14 days, the cells in the BM group showed significantly higher ALP activity and TP production, compared with those in the control group. These expressions indicate that cells in the BM group were undergoing odontoblastic differentiation, since ALP is involved in the initial phase of dentin matrix biomineralization, and promotes dephosphorylation of extracellular matrix proteins, providing inorganic phosphate. Therefore, this protein is considered an initial marker for odontoblastic differentiation.2222. Goldberg M, Smith AJ. Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med. 2004;15(1):13-27. doi:10.1177/154411130401500103 The greater TP content demonstrates a more intense protein expression on BM-seeded cells, and a direct relationship with cell differentiation.2121. Couve E, Osorio R, Schmachtenberg O. The amazing odontoblast: activity, autophagy, and aging. J Dent Res. 2013;92(9):765-72. doi:10.1177/0022034513495874,2222. Goldberg M, Smith AJ. Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med. 2004;15(1):13-27. doi:10.1177/154411130401500103 The odontoblastic differentiation was confirmed at the 28-day period, in which the cells in the BM group exhibited higher DSPP and DMP-1 gene expression, in comparison with those in the control group. These genes were chosen as markers for the odontogenic phenotype because they are the major dentinal non-collagenous proteins and play a crucial role during dentin mineralization, through their high calcium-ion-binding capacity.3030. Goldberg M, Takagi M. Dentine proteoglycans: composition, ultrastructure and functions. Histochem J. 1993;25(11):781-806. doi:10.1007/BF02388111 In the current study, the high expression of these odontogenesis-related genes by the pulp cells offered strong evidence that they differentiate into odontoblast-like cells. Furthermore, calcium content measurement showed that the BM acted on the cells to induce intense mineralized matrix deposition, which was about five times higher than that of the control group.
The effect of a chitosan-based biomembrane as a pulp-capping material was recently tested by Li et al.99. Li F, Liu X, Zhao S, Wu H, Xu HH. Porous chitosan bilayer membrane containing TGF-β1 loaded microspheres for pulp capping and reparative dentin formation in a dog model. Dent Mater. 2014;30(2):172-81. doi:10.1016/j.dental.2013.11.005 in dog teeth. These authors observed the presence of thin irregular reparative dentin formation along the pulpdentin border without the presence of dentinal tubules after 60 days, similar to that observed when CH was used. When the chitosan membrane was covered with transforming growth factor beta 1 (TGF-β1), the dentin bridge deposition was 3 – 6 times thicker. Therefore, the authors observed that the chitosan membrane itself has low bioactive potential on pulp tissue; however, the addition of growth factors increased dentin deposition. Similarly, when chitosan/collagen scaffolds were enriched with bone morphogenetic protein 7 (BMP-7), the pulp stem cells presented higher amounts of calcium deposition, ALP activity, and gene expression levels of DSPP and DMP-1, compared with control materials.3838. Yang X, Han G, Pang X, Fan M. Chitosan/collagen scaffold containing bone morphogenetic protein-7 DNA supports dental pulp stem cell differentiation in vitro and in vivo. J Biomed Mater Res A. 2012 Feb 18. doi: 10.1002/jbm.a.34064 The addition of a mineral phase to chitosan/collagen scaffolds seems to have an effect similar to that of growth factors.1010. Kim IY, Seo SJ, Moon HS, Yoo MK, Park IY, Kim BC, et al. Chitosan and its derivatives for tissue engineering applications. Biotechnol Adv. 2008;26(1):1-21. doi:10.1016/j.biotechadv.2007.07.009 The results of in vitro and in vivo studies have shown that chitosan/collagen scaffolds containing a Ca-based mineral phase were able to promote greater osteoblast phenotype induction and bone reconstruction.1111. Venkatesan J, Pallela R, Bhatnagar I, Kim SK. Chitosan-amylopectin/hydroxyapatite and chitosan-chondroitin sulphate/hydroxyapatite composite scaffolds for bone tissue engineering. Int J Biol Macromol. 2012;51(5):1033-42. doi:10.1016/j.ijbiomac.2012.08.020,1212. Liu H, Peng H, Wu Y, Zhang C, Cai Y, Xu G, et al. The promotion of bone regeneration by nanofibrous hydroxyapatite/chitosan scaffolds by effects on integrin-BMP/Smad signaling pathway in BMSCs. Biomaterials. 2013;34(18):4404-17. doi:10.1016/j.biomaterials.2013.02.048,1313. Qiao P, Wang J, Xie Q, Li F, Dong L, Xu T. Injectable calcium phosphate-alginate-chitosan microencapsulated MC3T3-E1 cell paste for bone tissue engineering in vivo. Mater Sci Eng C. 2013;33(8):4633-9. doi:10.1016/j.msec.2013.07.022,1414. Shen S, Fu D, Xu F, Long T, Hong F, Wang J. The design and features of apatite-coated chitosan microspheres as injectable scaffold for bone tissue engineering. Biomed Mater. 2013;8(2):025007. doi:10.1088/1748-6041/8/2/025007 The inclusion of a mineral phase provides an environment with a chemical composition similar to that of the inorganic components of bone, increasing the osteoblastic phenotype. In addition, these composites feature enhanced mechanical properties by matrix reinforcement.1010. Kim IY, Seo SJ, Moon HS, Yoo MK, Park IY, Kim BC, et al. Chitosan and its derivatives for tissue engineering applications. Biotechnol Adv. 2008;26(1):1-21. doi:10.1016/j.biotechadv.2007.07.009 It was previously demonstrated that calcium-aluminate is capable of inducing osteoblastic marker expression and mineralized matrix deposition in pre-osteoblast cells.1515. Castro-Raucci LMS, Oliveira IR, Teixeira LN, Rosa AL, Oliveira PT, Jacobovitz M. Effects of a novel calcium aluminate cement on the early events of the progression of osteogenic cell cultures. Braz Dent J. 2011;22(2):99-104. doi:10.1590/S0103-64402011000200002,1919. Silva EJNL, Herrera DR, Rosa TP, Duque TM, Jacinto RC, Gomes BPFA, et al. Evaluation of cytotoxicity, antimicrobial activity and physicochemical properties of a calcium aluminate-based endodontic material. J Appl Oral Sci. 2014;22(1):61-7. doi: 10.1590/1678-775720130031 Moreover, an in vivo study conducted on rat’s molars demonstrated that calcium-aluminate cement enhanced dentin bridge deposition after its application to mechanically exposed pulps.3939. Kramer PR, Woodmansey KF, White R, Primus CM, Opperman LA. Capping a pulpotomy with calcium aluminosilicate cement: comparison to mineral trioxide aggregates. J Endod. 2014;40(9):1429-34. doi:10.1016/j.joen.2014.02.001 One may conclude that the experimental chitosan/collagen biomembrane embedded with calcium-aluminate microparticles seems to be a very interesting candidate for direct pulp-capping therapy, since it was cytocompatible with human dental pulp cells, acting as a substrate for cell adhesion and proliferation, followed by high induction of odontoblastic phenotypes, and culminating in intense mineralized matrix deposition.
Conclusions
A biomembrane composed of a chitosan/collagen matrix, embedded with calcium-aluminate microparticles, enhanced human dental pulp cell proliferation, followed by a high expression of odontoblastic phenotypes, achieving intense deposition of a mineralized matrix. This innovative bioactive material can give new direction to and broaden the prospects for pulp exposure therapies by inducing a regenerative process without chemical damage to pulp cells.
Acknowledgements
The authors wish to acknowledge the support of the Fundação de Amparo à Pesquisa do Estado de São Paulo – FAPESP (grants no. 2013/23520-0 and no. 2014/13034-3) and the Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq (grant no. 303599/2014-6 and no. 443153/2014-0).
References
-
1Sangwan P, Sangwan A, Duhan J, Rohilla A. Tertiary dentinogenesis with calcium hydroxide: a review of proposed mechanisms. Int Endod J. 2013;46(1):3-19. doi:10.1111/j.1365-2591.2012.02101.x
-
2Briso AL, Rahal V, Mestrener SR, Dezan Junior E. Biological response of pulps submitted to different capping materials. Braz Oral Res. 2006;20(3):219-25. doi:10.1590/S1806-83242006000300007
-
3Costa CAS, Oliveira MF, Giro EM, Hebling J. Biocompatibility of resin-based materials used as pulp-capping agents. Int Endod J. 2003;36(12):831-9. doi:10.1111/j.1365-2591.2003.00702.x
-
4Albuquerque DS, Gominho LF, Santos RA. Histologic evaluation of pulpotomy performed with ethyl-cyanoacrylate and calcium hydroxide. Braz Oral Res. 2006;20(3):226-30. doi:10.1590/S1806-83242006000300008
-
5Cavalcanti BN, Rode SM, França CM, Marques MM. Pulp capping materials exert an effect on the secretion of IL-1β and IL-8 by migrating human neutrophils. Braz Oral Res. 2011;25(1):13-8. doi:10.1590/S1806-83242011000100003
-
6Costa CAS, Hebling J, Scheffel DLS, Soares DGS, Basso FG, Ribeiro APD. Methods to evaluate and strategies to improve the biocompatibility of dental materials and operative techniques. Dent Mater. 2014;30(7):769-84. doi:10.1016/j.dental.2014.04.010
-
7Schmalz G, Smith AJ. Pulp development, repair, and regeneration: challenges of the transition from traditional dentistry to biologically based therapies. J Endod. 2014;40(4 Suppl):S2-5. doi:10.1016/j.joen.2014.01.018
-
8Zheng Z, Wei Y, Wang G, Gong Y, Zhang X. Surface characterization and cytocompatibility of three chitosan/polycation composite membranes for guided bone regeneration. J Biomater Appl. 2009;24(3):209-29. doi:10.1177/0885328208095825
-
9Li F, Liu X, Zhao S, Wu H, Xu HH. Porous chitosan bilayer membrane containing TGF-β1 loaded microspheres for pulp capping and reparative dentin formation in a dog model. Dent Mater. 2014;30(2):172-81. doi:10.1016/j.dental.2013.11.005
-
10Kim IY, Seo SJ, Moon HS, Yoo MK, Park IY, Kim BC, et al. Chitosan and its derivatives for tissue engineering applications. Biotechnol Adv. 2008;26(1):1-21. doi:10.1016/j.biotechadv.2007.07.009
-
11Venkatesan J, Pallela R, Bhatnagar I, Kim SK. Chitosan-amylopectin/hydroxyapatite and chitosan-chondroitin sulphate/hydroxyapatite composite scaffolds for bone tissue engineering. Int J Biol Macromol. 2012;51(5):1033-42. doi:10.1016/j.ijbiomac.2012.08.020
-
12Liu H, Peng H, Wu Y, Zhang C, Cai Y, Xu G, et al. The promotion of bone regeneration by nanofibrous hydroxyapatite/chitosan scaffolds by effects on integrin-BMP/Smad signaling pathway in BMSCs. Biomaterials. 2013;34(18):4404-17. doi:10.1016/j.biomaterials.2013.02.048
-
13Qiao P, Wang J, Xie Q, Li F, Dong L, Xu T. Injectable calcium phosphate-alginate-chitosan microencapsulated MC3T3-E1 cell paste for bone tissue engineering in vivo. Mater Sci Eng C. 2013;33(8):4633-9. doi:10.1016/j.msec.2013.07.022
-
14Shen S, Fu D, Xu F, Long T, Hong F, Wang J. The design and features of apatite-coated chitosan microspheres as injectable scaffold for bone tissue engineering. Biomed Mater. 2013;8(2):025007. doi:10.1088/1748-6041/8/2/025007
-
15Castro-Raucci LMS, Oliveira IR, Teixeira LN, Rosa AL, Oliveira PT, Jacobovitz M. Effects of a novel calcium aluminate cement on the early events of the progression of osteogenic cell cultures. Braz Dent J. 2011;22(2):99-104. doi:10.1590/S0103-64402011000200002
-
16Aguilar FG, Garcia LFR, Pires-de-Souza FCP. Biocompatibility of new calcium aluminate cement (EndoBinder). J Endod. 2012;38(3):367-71. doi:10.1016/j.joen.2011.11.002
-
17Pires-de-Souza FCP, Morães PC, Garcia LFR, Aguilar FG, Watanabe E. Evaluation of pH, calcium ion release and antimicrobial activity of a new calcium aluminate cement. Braz Oral Res. 2013;27(4):324-30. doi:10.1590/S1806-83242013000400006
-
18Garcia LFR, Huck C, Oliveira LM, Souza PPC, Costa CAS. Biocompatibility of new calcium aluminate cement: tissue reaction and expression of inflammatory mediators and cytokines. J Endod. 2014;40(12):2024-9. doi:10.1016/j.joen.2014.08.015
-
19Silva EJNL, Herrera DR, Rosa TP, Duque TM, Jacinto RC, Gomes BPFA, et al. Evaluation of cytotoxicity, antimicrobial activity and physicochemical properties of a calcium aluminate-based endodontic material. J Appl Oral Sci. 2014;22(1):61-7. doi: 10.1590/1678-775720130031
-
20Soares DG, Basso FG, Scheffel DS, Hebling J, Costa CAS. Responses of human dental pulp cells after application of a low-concentration bleaching gel to enamel. Arch Oral Biol. 2015;60(9):1428-36. doi:10.1016/j.archoralbio.2015.06.014
-
21Couve E, Osorio R, Schmachtenberg O. The amazing odontoblast: activity, autophagy, and aging. J Dent Res. 2013;92(9):765-72. doi:10.1177/0022034513495874
-
22Goldberg M, Smith AJ. Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med. 2004;15(1):13-27. doi:10.1177/154411130401500103
-
23Fouad AF, Verma P. Healing after regenerative procedures with and without pulpal infection. J Endod. 2014;40(4 Suppl):S58-64. doi:10.1016/j.joen.2014.01.022
-
24Kotsanos N, Arapostathis KN, Arhakis A, Menexes G. Direct pulp capping of carious primary molars. A specialty practice based study. J Clin Pediatr Dent. 2014;38(4):307-12. doi:10.17796/jcpd.38.4.k651814611318430
-
25Cho SY, Seo DG, Lee SJ, Lee J, Lee SJ, Jung IY. Prognostic factors for clinical outcomes according to time after direct pulp capping. J Endod. 2013;39(3):327-31. doi:10.1016/j.joen.2012.11.034
-
26Mente J, Hufnagel S, Leo M, Michel A, Gehrig H, Panagidis D, et al. Treatment outcome of mineral trioxide aggregate or calcium hydroxide direct pulp capping: long-term results. J Endod. 2014;40(11):1746-51. doi:10.1016/j.joen.2014.07.019
-
27Colombo JS, Moore AN, Hartgerink JD, D’Souza RN. Scaffolds to control inflammation and facilitate dental pulp regeneration. J Endod. 2014;40(4 Suppl):S6-12. doi:10.1016/j.joen.2014.01.019
-
28Ma PX. Biomimetic materials for tissue engineering. Adv Drug Deliv Rev. 2008;60(2):184-98. doi:10.1016/j.addr.2007.08.041
-
29Goldberg M, Kulkarni AB, Young M, Boskey A. Dentin: structure, composition and mineralization. Front Biosci (Elite Ed.). 2011 Jan 1;3:711-35. doi:10.2741/e281
-
30Goldberg M, Takagi M. Dentine proteoglycans: composition, ultrastructure and functions. Histochem J. 1993;25(11):781-806. doi:10.1007/BF02388111
-
31Embery G, Hall R, Waddington R, Septier D, Goldberg M. Proteoglycans in dentinogenesis. Crit Rev Oral Biol Med. 2001;12(4):331-49. doi:10.1177/10454411010120040401
-
32Venkatesan J, Kim SK. Chitosan composites for bone tissue engineering – an overview. Mar Drugs. 2010;8(8):2252-66. doi:10.3390/md8082252
-
33Sayin E, Baran ET, Hasirci V. Protein-based materials in load-bearing tissue-engineering applications. Regen Med. 2014;9(5):687-701. doi:10.2217/rme.14.52
-
34Oliveira IR, Andrade TL, Jacobovitz M, Pandolfelli VC. Bioactivity of calcium aluminate endodontic cement. J Endod. 2013;39(6):774-8. doi:10.1016/j.joen.2013.01.013
-
35Amir LR, Suniarti DF, Utami S, Abbas B. Chitosan as a potential osteogenic factor compared with dexamethasone in cultured macaque dental pulp stromal cells. Cell Tissue Res. 2014;358(2):407-15. doi:10.1007/s00441-014-1938-1
-
36Galler KM, Aulisa L, Regan KR, D’Souza RN, Hartgerink JD. Self-assembling multidomain peptide hydrogels: designed susceptibility to enzymatic cleavage allows enhanced cell migration and spreading. J Am Chem Soc. 2010;132(9):3217-23. doi:10.1021/ja910481t
-
37Zhang Y, Wang Y, Shi B, Cheng X. A platelet-derived growth factor releasing chitosan/coral composite scaffold for periodontal tissue engineering. Biomaterials. 2007;28(8):1515-22. doi:10.1016/j.biomaterials.2006.11.040
-
38Yang X, Han G, Pang X, Fan M. Chitosan/collagen scaffold containing bone morphogenetic protein-7 DNA supports dental pulp stem cell differentiation in vitro and in vivo. J Biomed Mater Res A. 2012 Feb 18. doi: 10.1002/jbm.a.34064
-
39Kramer PR, Woodmansey KF, White R, Primus CM, Opperman LA. Capping a pulpotomy with calcium aluminosilicate cement: comparison to mineral trioxide aggregates. J Endod. 2014;40(9):1429-34. doi:10.1016/j.joen.2014.02.001
Publication Dates
-
Publication in this collection
2016
History
-
Received
13 Oct 2015 -
Accepted
02 Dec 2015 -
Received
05 Mar 2016


 • Indicates a significant difference for the BM group among the time points (two-way ANOVA; Tukey’s test; p < 0.05).* Indicates a significant difference between the NC and BM groups at each time point (Student’s t-test; p < 0.05).
• Indicates a significant difference for the BM group among the time points (two-way ANOVA; Tukey’s test; p < 0.05).* Indicates a significant difference between the NC and BM groups at each time point (Student’s t-test; p < 0.05).