Abstract
OBJECTIVES:
We explored whether high blood pressure is associated with metabolic, inflammatory and prothrombotic dysregulation in patients with metabolic syndrome.
METHODS:
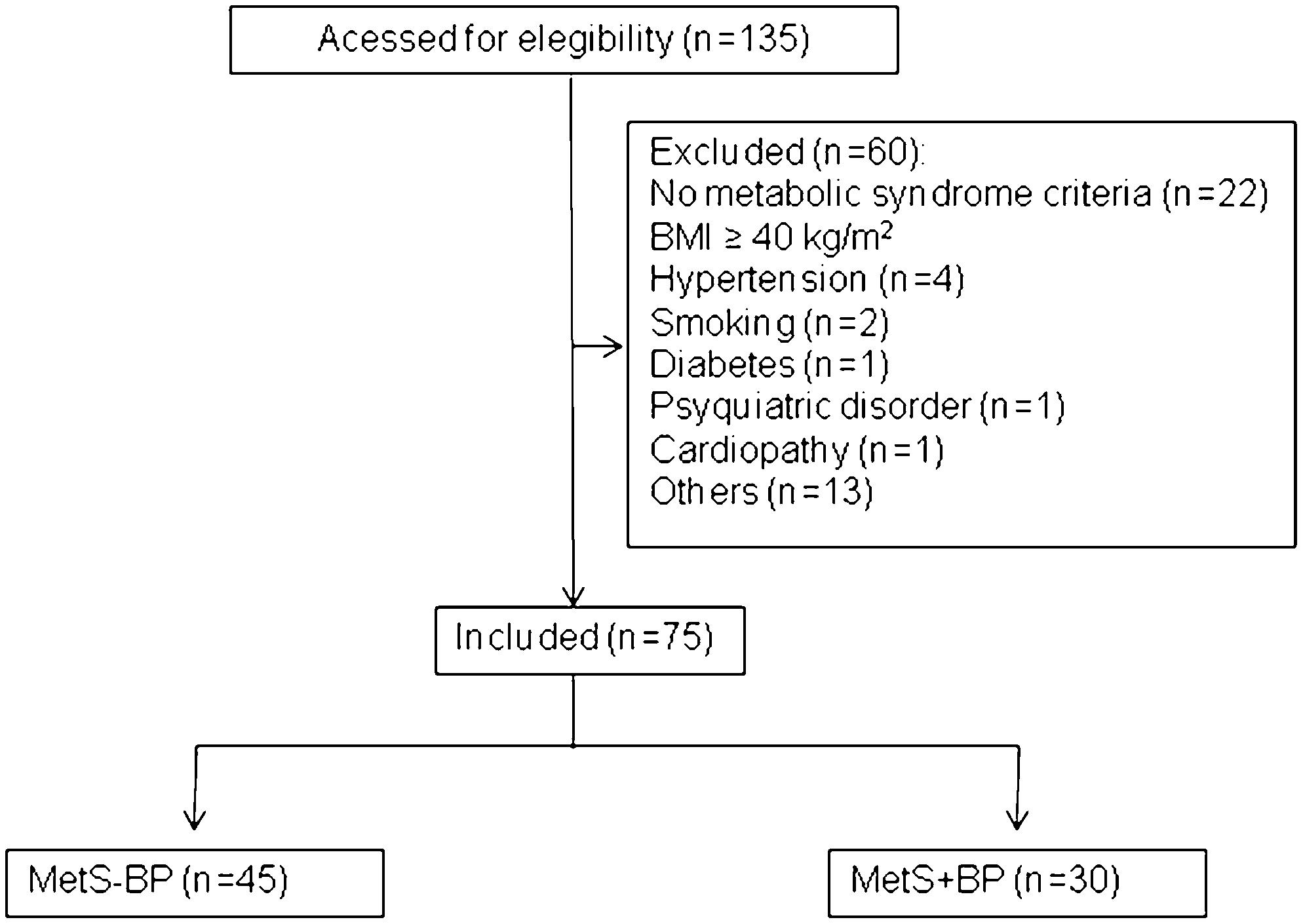
We evaluated 135 consecutive overweight/obese patients. From this group, we selected 75 patients who were not under the regular use of medications for metabolic syndrome as defined by the current Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults criteria. The patients were divided into metabolic syndrome with and without high blood pressure criteria (≥130/≥85 mmHg).
RESULTS:
Compared to the 45 metabolic syndrome patients without high blood pressure, the 30 patients with metabolic syndrome and high blood pressure had significantly higher glucose, insulin, homeostasis model assessment insulin resistance index, total cholesterol, low-density lipoprotein-cholesterol, triglycerides, uric acid and creatinine values; in contrast, these patients had significantly lower high-density lipoprotein-cholesterol values. Metabolic syndrome patients with high blood pressure also had significantly higher levels of retinol-binding protein 4, plasminogen activator inhibitor 1, interleukin 6 and monocyte chemoattractant protein 1 and lower levels of adiponectin. Moreover, patients with metabolic syndrome and high blood pressure had increased surrogate markers of sympathetic activity and decreased baroreflex sensitivity. Logistic regression analysis showed that high-density lipoprotein, retinol-binding protein 4 and plasminogen activator inhibitor-1 levels were independently associated with metabolic syndrome patients with high blood pressure. There is a strong trend for an independent association between metabolic syndrome patients with high blood pressure and glucose levels.
CONCLUSIONS:
High blood pressure, which may be related to the autonomic dysfunction, is associated with metabolic, inflammatory and prothrombotic dysregulation in patients with metabolic syndrome.
Hypertension; Sympathetic Activity; Insulin Resistance; Inflammation; Prothrombosis; Metabolic Syndrome
INTRODUCTION
In 1988, the cardiovascular risk factor cluster, which includes obesity, increased blood
pressure, high triglycerides and glucose and low HDL (high-density lipoprotein)-cholesterol, was
given the name “metabolic syndrome” (MetS) (11. Reaven GM. Banting lecture 1988. Role of insulin resistance in human disease.
Diabetes. 1988;37(12):1595-607, http://dx.doi.org/10.2337/diab.37.12.1595.
http://dx.doi.org/10.2337/diab.37.12.159...
).
Since then, the scientific community has tried to better define whether MetS predicts cardiovascular
morbidity and mortality better than the sum of the individual components and whether one component
is pivotal over the others. Most evidence points to visceral obesity and insulin resistance as
central features of MetS. Little attention has been given to exploring other components of MetS,
such as blood pressure and the potential factors that can influence not only blood pressure but also
the metabolic dysregulation observed in MetS.
Obesity-related sympathetic activation is an attractive explanation for several components of
MetS. In particular, in 1994, the aggregation of cardiovascular risk factors and signs of a hyper
sympathetic state was demonstrated in the Tecumseh population (22. Smith S, Julius S, Jamerson K, Amerena J, Schork N. Hematocrit levels and
physiologic factors in relationship to cardiovascular risk in Tecumseh, Michigan. J Hypertens.
1994;12(4):455-62.). Indeed, increased sympathetic activity (faster heart rate, higher cardiac output and
plasma noradrenaline) correlated with higher levels of glucose, insulin, cholesterol, triglycerides,
body weight and hematocrit and lower levels of HDL-cholesterol. In the Framingham Heart Study (33. Kannel WB. Risk stratification in hypertension: new insights from the Framingham
Study. Am J Hypertens. 2000;13(1Pt 2):3S-10S.), more than 50% of hypertensive patients had 2 or more metabolic
abnormalities. Only 19% of males and 17% of females had isolated hypertension. In view of this
finding, the name hypertensive syndrome had been used in the past (44. Houston MC. New insights and approaches to reduce end-organ damage in the
treatment of hypertension: subsets of hypertension approach. Am Heart J. 1992;123(5):1337-67,
http://dx.doi.org/10.1016/0002-8703(92)91042-Y.
http://dx.doi.org/10.1016/0002-8703(92)9...
). However, it is not clear whether the increase in blood pressure is associated with
exacerbations of the metabolic, proinflammatory, prothrombotic, vascular and autonomic dysfunctions
in patients with MetS.
Evidence suggests that autonomic dysregulation also contributes to elevated blood pressure and metabolic abnormalities. Moreover, α1-adrenoceptor antagonists lower blood pressure, improve insulin sensitivity and ameliorate dyslipidemia (55. Chapman N, Chen CY, Fujita T, Hobbs FD, Kim SJ, Staessen JA, et al. Time to re-appraise the role of alpha-1 adrenoceptor antagonists in the management of hypertension? J Hypertens. 2010;28:1796-803. Erratum in: J Hypertens. 2010;28(11):2351.). However, it is unclear whether autonomic dysregulation in metabolic syndrome is associated not only with hemodynamic impairment but also with metabolic, inflammatory and other abnormalities associated with the syndrome. We hypothesized that high blood pressure, which may reflect increased sympathetic activity, is independently associated with metabolic dysregulation and inflammation in patients with MetS.
METHODS
Study population
This research was performed at the Heart Institute (InCor) at the University of São Paulo Medical School. Over a 1-year period, we initially evaluated 135 consecutive overweight or obese patients from the São Paulo metropolitan area. From this group, we selected 75 patients with MetS, diagnosed according to ATP III (Third Report of the Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults criteria, and with at least three of the following criteria: high blood pressure - arterial blood pressure ≥130 and/or ≥85 mmHg for systolic and diastolic blood pressure, respectively; high glucose - fasting glucose ≥100 mg/dL; increased waist circumference - ≥102 cm in men and ≥88 in women; increased triglycerides - ≥150 mg/dL; decreased HDL - <40 mg/dL in men and <50 mg/mg/dL in women) (66. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002;106(25):3143-421.). We excluded patients with morbid obesity (body mass index ≥40 kg/m2), severe hypertension (BP >180/110 mmHg), under regular physical activity, secondary forms of hypertension, diabetes, smokers, patients with any chronic disease and regularly using medications (including antihypertensives, see Figure 1). The patients were divided into 2 groups according to blood pressure status (patients with MetS without high blood pressure, i.e., <130/<85 mmHg: MetS-BP) and (patients with MetS with high blood pressure, i.e., ≥130 systolic and/or ≥85 mmHg diastolic: MetS+BP).
Clinical evaluation
Patients who fulfilled the criteria for the study underwent a medical history evaluation. Blood pressure was measured in triplicate after 5 minutes of seated rest using a calibrated sphygmomanometer by the same investigator (J.S.G). The average from 3 blood pressure values was used to define the presence or absence of the ATP III, the criterion used to define elevated blood pressure (≥130 or ≥85 mmHg for systolic and diastolic blood pressure levels, respectively). Fasting blood samples were drawn for biochemical tests and cytokine levels. Serum and plasma were obtained and then stored at -80°C. During the second visit, anthropometric measurements (body mass index - BMI; waist and hip circumferences; and bicipital, tricipital, subscapular and suprailiac skinfolds) and fat mass, lean mass, basal metabolism and total body water were measured using bioimpedance-measuring equipment (BIA 450, Bio dynamics, Seattle, USA) (77. Pimentel GD, Bernhard AB, Frezza MR, Rinaldi AE, Burini RC. Bioelectric impedance overestimates the body fat in overweight and underestimates in Brazilian obese women: a comparison with Segal equation 1. Nutr Hosp. 2010;25(5):741-5.).
Biochemistry measurements
Total cholesterol, LDL-cholesterol, HDL-cholesterol, triglycerides, uric acid, glucose and creatinine levels were measured using commercial kits.
Insulin resistance
Insulin resistance was measured using the following 2 methods: the traditional Homeostasis Model Assessment Insulin Resistance method (HOMA-IR) and the Retinol-Binding Protein 4 method (RBP4). The HOMA-IR index was calculated as [fasting serum glucose (mmol/L) x fasting serum insulin (UI/mL)]/22.5.
Cytokines
Cytokines, including total adiponectin, resistin, leptin, insulin, interleukin 6 (IL-6), tumoral
necrosis factor alpha (TNF-α), plasminogen activator inhibitor 1 (PAI-1) and monocyte
chemoattractant protein 1 (MCP-1), were measured using a multiplex sandwich enzyme-linked
immunosorbent assay (ELISA, Millipore, MA, USA) (88. Schipper HS, de Jager W, van Dijk ME, Meerding J, Zelissen PM, Adan RA, et al.
Clin Chem. 2010;56(8):1320-8, http://dx.doi.org/10.1373/clinchem.2010.146118.
http://dx.doi.org/10.1373/clinchem.2010....
).
Power spectral analysis
The data for spectral analysis were derived from a finger pulse contour recorded by beat-to-beat
blood pressure measurements (Finometer®, FMS, Finapres Medical System BV, Holland)
and from the electrocardiogram (ECG) for 10 minutes to characterize the RR interval series. Power
spectral density was obtained by the fast Fourier transformation, using Welch's method with a
Hanning window of 512 points and 50% overlap as previously described (99. Drager LF, Ueno LM, Lessa PS, Negrão CE, Lorenzi-Filho G, Krieger EM.
Sleep-related changes in hemodynamic and autonomic regulation in human hypertension.
J Hypertens. 2009;27(8):1655-63,
http://dx.doi.org/10.1097/HJH.0b013e32832c6982.
http://dx.doi.org/10.1097/HJH.0b013e3283...
). The entire time, series were previously re-sampled at 5.0 Hz to be equally
spaced in time by a special signal editor developed for Matlab (MATLAB 6.0; Mathworks, Natick,
Massachusetts, USA). Interpolated time series were decimated to be equally time-spaced. Two main
components were considered for the RR interval variability: low frequency (LF; 0.04 to 0.15 Hz) and
high frequency (HF; 0.15 to 0.4 Hz). The power density of each spectral component from the RR
interval was calculated in absolute values and normalized units. The normalized units were obtained
by calculating the percentages of LF variability (LFms2) and HF variability
(HFms2) with respect to the total power after subtracting the power of very low frequency
components (VLF; frequencies of <0.04 Hz). The LF/HF ratio was also calculated to evaluate the
sympathovagal balance. In addition, we also evaluated the power spectral analysis of systolic BP.
Systolic BP spectral powers were quantified in the LF component (0.04-0.15 Hz) and were reported in
absolute units. LF of systolic BP has been associated as a reliable marker of sympathetic activity
(1010. Pagani M, Lombardi F, Guzzetti S, Rimoldi O, Furlan R, Pizzinelli P, et al.
Power spectral analysis of heart rate and arterial pressure variabilities as a marker of
sympatho-vagal interaction in man and conscious dog. Circ Res. 1986;59(2):178-93,
http://dx.doi.org/10.1161/01.RES.59.2.178.
http://dx.doi.org/10.1161/01.RES.59.2.17...
).
Baroreflex sensitivity
Beat-to-beat values of systolic BP and RR interval were used to estimate the cardiac baroreflex
sensitivity (BRS) by spectral analysis, using the alpha index for the low-frequency band (0.04 to
0.15 Hz). The coherence between the RR interval and the systolic BP signal variability was assessed
by cross-spectral analysis. The alpha index was calculated only when the magnitude of the squared
coherence between the RR and systolic BP signals exceeded 0.5 (range 0-1) in the LF band. After
coherence calculation, the alpha index was obtained from the square root of the ratio between RR and
systolic BP variability in the 2 major LF bands (1010. Pagani M, Lombardi F, Guzzetti S, Rimoldi O, Furlan R, Pizzinelli P, et al.
Power spectral analysis of heart rate and arterial pressure variabilities as a marker of
sympatho-vagal interaction in man and conscious dog. Circ Res. 1986;59(2):178-93,
http://dx.doi.org/10.1161/01.RES.59.2.178.
http://dx.doi.org/10.1161/01.RES.59.2.17...
11. Heart rate variability: standards of measurement, physiological interpretation
and clinical use. Task Force of the European Society of Cardiology and the North American Society of
Pacing and Electrophysiology. Circulation. 1996;93(5):1043-65.
12. Lucini D, Cusumano G, Bellia A, Kozakova M, Difede G, Lauro R, et al. Is reduced
baroreflex gain a component of the metabolic syndrome? Insights from the LINOSA study.
J Hypertens. 2006;24(2):361-70,
http://dx.doi.org/10.1097/01.hjh.0000202817.02836.9c.
http://dx.doi.org/10.1097/01.hjh.0000202...
-1313. Pagani M, Somers V, Furlan R, Dell'Orto S, Conway J, Baselli G, et al.
Changes in autonomic regulation induced by physical training in mild hypertension. Hypertension.
1988;12(6):600-10, http://dx.doi.org/10.1161/01.HYP.12.6.600.
http://dx.doi.org/10.1161/01.HYP.12.6.60...
).
Anxiety and depression evaluation
We performed the Hamilton Scale to assess anxiety (1414. Hamilton M. The assessment of anxiety states by rating. Br J Med
Psychol. 1959;32(1):50-5.-1515. Maier W, Buller R, Philipp M, Heuser I. The Hamilton Anxiety Scale: reliability,
validity and sensitivity to change in anxiety and depressive disorders. J Affect Disord.
1988;14(1):61-8, http://dx.doi.org/10.1016/0165-0327(88)90072-9.
http://dx.doi.org/10.1016/0165-0327(88)9...
) and the Beck questionnaire to assess
depression (1616. Gomes-Oliveira MH, Gorenstein C, Lotufo Neto F, Andrade LH, Wang YP. Validation
of the Brazilian Portuguese version of the Beck Depression Inventory-II in a community sample. Rev.
Bras. Psiquiatr. 2012;34(4):389-94, http://dx.doi.org/10.1016/j.rbp.2012.03.005.
http://dx.doi.org/10.1016/j.rbp.2012.03....
).
Statistical analysis
The SPSS software (SPSS 10.0, Chicago, IL) was used for statistical calculations. Data were expressed as the means±standard deviations, medians or percentages, as indicated. The Kolmogorov-Smirnov test was used to assess the normality of distribution of each variable studied. Categorical variables (sex, race and percentage of MetS components) were compared using Fisher's test. Numerical variables were compared using the unpaired Student's t test. Logistic regression analysis (forward model) was used to identify the metabolic, pro-inflammatory and prothrombotic markers that were independently associated with the MetS+BP. The model was controlled for age and BMI. ANOVA was used to select variables for the logistic regression analysis. All variables with a p<0.1 in the univariate analysis were selected for the final model. P<0.05 was considered to be statistically significant.
Ethics statement
The local ethics committee (Institutional Review Board - Heart Institute) approved the protocol, which was in accordance with the ethical standards and with the Helsinki Declaration of 1975, and all participants gave written informed consent.
RESULTS
Figure 1 depicts derivation of the study sample of 75 subjects from the initial sample of 135 patients with MetS. The percentage of each MetS criterion is reported in the supplemental file.
Baseline descriptive data on the MetS patients without (N = 45) and with (N = 30) high blood pressure are provided in Table 1. We observed a significant trend for increasing age in the MetS+BP group. There was no significant difference in gender, % of Caucasians and anthropometric data, including body mass index, waist circumference, hip circumference, waist-to-hip ratio, fat percentage as calculated by skinfold, fat mass, lean mass, metabolic tax and total body water estimated by bioelectrical impedance analysis (BIA) (Table 1). As expected, there were significant differences in the systolic and diastolic blood pressure measurements between groups. Pulse intervals during spectral analysis were significantly lower (heart rate faster) in the MetS+BP compared to the MetS-BP group (Table 2). The LF components for heart rate and blood pressure and the LF/HF ratio were significantly higher in the MetS+BP group than in the MetS-BP group. Baroreflex sensitivity, as evaluated by the alpha index, was impaired in the MetS+BP group compared with the MetS-BP group. No differences were detected between groups with regards to anxiety and depression (supplemental file).
Biochemistry data (glucose, total cholesterol, HDL-cholesterol, LDL-cholesterol, triglycerides, uric acid and creatinine), along with insulin and the HOMA-IR index, were significantly higher in the MetS+BP group than in the MetS-BP group (Table 3).
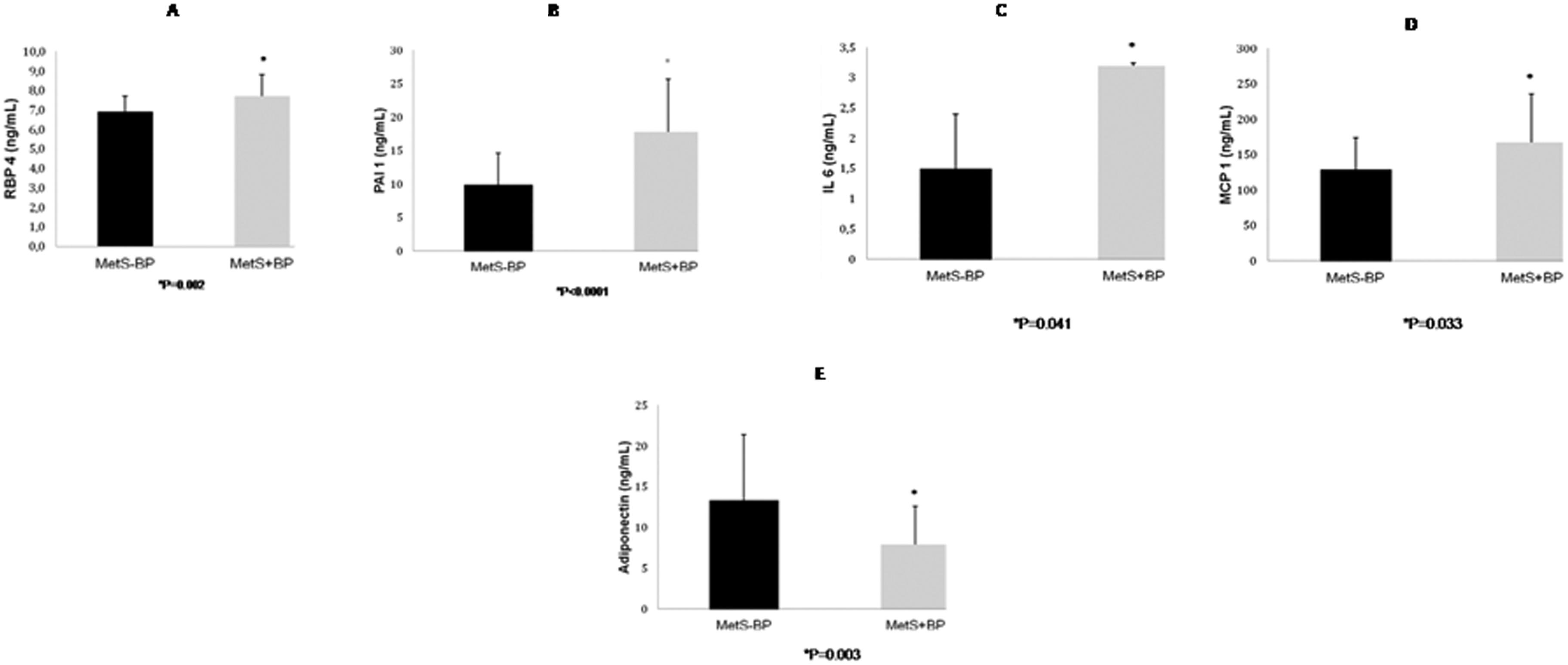
RBP4 was significantly higher in the MetS+BP group than the MetS-BP group (Figure 2A). Leptin, resistin and TNF-α levels did not differ between the MetS+BP group (49.4±41.5 pg/mL, 12.6±4.6 pg/mL and 4.8±1.7 ng/mL, respectively) and the MetS-BP group (41.4±20.0 pg/mL, 12.9±4.9 pg/mL and 4.4±1.4 ng/mL, respectively). IL-6, PAI-1 and MCP-1, a marker for inflammatory activity, were significantly higher in the MetS+BP group (Figures 2B-2D). Adiponectin was lower in the MetS+BP group compared to the MetS-BP group (Figure 2E). The differences in RBP4, PAI-1, IL-6, MCP-1 and adiponectin levels between groups remained significant after adjusting for age and sex.
Retinol-binding protein 4 levels (2A), plasminogen activator inhibitor 1 levels (2B), interleukin 6 levels (2C), monocyte chemoattractant protein 1 levels (2D) and adiponectin levels (2E) in metabolic syndrome - blood pressure and metabolic syndrome + blood pressure subjects. The results are presents as the means±SD.
Logistic regression analysis indicated that HDL, RBP-4 and PAI-1 levels were independently associated with MetS+BP. There was a strong trend for an independent association between MetS+BP and glucose levels (Table 4).
Logistic regression analysis for variables independently associated with metabolic syndrome with high blood pressure (metabolic syndrome + blood pressure)
DISCUSSION
Previous studies have shown that patients with MetS have multiple abnormalities that contribute
to increased cardiovascular risk, including metabolic, proinflammatory, prothrombotic and autonomic
variables (1717. Graham TE, Yang Q, Blüher M, Hammarstedt A, Ciaraldi TP, Henry RR, et al.
Retinol-binding protein 4 and insulin resistance in lean, obese, and diabetic subjects.
N Engl J Med. 2006;354(24):2552-63.
18. Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care.
2004;27(6):1487-95, http://dx.doi.org/10.2337/diacare.27.6.1487.
http://dx.doi.org/10.2337/diacare.27.6.1...
-1919. Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin RA, et al.
Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart,
Lung, and Blood Institute Scientific Statement. Circulation. 2005;112(17):2735-52,
http://dx.doi.org/10.1161/CIRCULATIONAHA.105.169404.
http://dx.doi.org/10.1161/CIRCULATIONAHA...
). We
hypothesized that MetS patients with elevated blood pressure may have greater abnormalities in these
same variables. By stratifying patients with similar obesity levels according to the presence or
absence of high blood pressure, we found that those with high blood pressure (MetS+BP) had
higher surrogate markers of sympathetic activity derived from spectral analysis and greater
impairment in several components of MetS, including higher glucose and triglycerides and lower
HDL-cholesterol. The MetS group with high blood pressure also had higher values for other risk
factors, including total cholesterol, LDL-cholesterol, uric acid and insulin, along with HOMA index
and RBP4, which are associated with insulin resistance (1717. Graham TE, Yang Q, Blüher M, Hammarstedt A, Ciaraldi TP, Henry RR, et al.
Retinol-binding protein 4 and insulin resistance in lean, obese, and diabetic subjects.
N Engl J Med. 2006;354(24):2552-63.,2020. Janssen I, Katzmarzyk PT, Ross R. Waist circumference and not body mass index
explains obesity-related health risk. Am J Clin Nutr. 2004;79(3):379-84.,2121. Lean ME, Han TS, Morrison CE. Waist circumference as a measure for indicating
need for weight management. BMJ. 1995;311(6998):158-61,
http://dx.doi.org/10.1136/bmj.311.6998.158.
http://dx.doi.org/10.1136/bmj.311.6998.1...
). In
addition, several markers of inflammatory and prothrombotic activity, including IL-6, PAI-1 and
MCP-1, were higher, while adiponectin levels were lower in patients with MetS and high blood
pressure. Finally, we found that HDL-cholesterol, RBP4 and PAI-1 were independently associated with
MetS+BP. Taken together, our results suggest a significant heterogeneity of the MetS in
promoting metabolic, pro-inflammatory, prothrombotic and autonomic impairments.
MetS was defined based on the observation that a number of interrelated characteristics and
diseases tended to cluster in the same individual and contributed to increased cardiovascular risk.
However, MetS is not a well-defined pathophysiological entity, and the diagnostic criteria are
variable and based on expert opinions (2222. Costa GB, Horta N, Resende ZF, Souza G, Barreto LM, Correia LH, et al. Body mass
index has a good correlation with proatherosclerotic profile in children and adolescents. Arq Bras
Cardiol. 2009;93(3):261-7, http://dx.doi.org/10.1590/S0066-782X2009000900010.
http://dx.doi.org/10.1590/S0066-782X2009...
). The literature
has debated the existence of a major trigger for MetS and its components or whether the whole is
really greater than the sum of the parts. Obesity emerges as an attractive candidate to explain all
manifestations of MetS, but the best way to measure this parameter in the MetS scenario is also
debatable. Some studies identified an association between abdominal obesity (waist circumference)
and components of MetS (2323. Katzmarzyk PT, Srinivasan SR, Chen W, Malina RM, Bouchard C, Berenson GS. Body
mass index, waist circumference, and clustering of cardiovascular disease risk factors in a biracial
sample of children and adolescents. Pediatrics. 2004;114(2):e198-205,
http://dx.doi.org/10.1542/peds.114.2.e198.
http://dx.doi.org/10.1542/peds.114.2.e19...
,2424. Schneider HJ, Glaesmer H, Klotsche J, Böhler S, Lehnert H, Zeiher AM, et
al. J Clin Endocrinol Metab. 2007;92(2):589-94.), while others showed an association with body mass index or waist-to-hip ratio (2525. Yusuf S, Hawken S, Ounpuu S, Bautista L, Franzosi MG, Commerford P, et al.
Obesity and the risk of myocardial infarction in 27,000 participants from 52 countries: a
case-control study. Lancet. 2005;366(9497):1640-9,
http://dx.doi.org/10.1016/S0140-6736(05)67663-5.
http://dx.doi.org/10.1016/S0140-6736(05)...
26. Lambert EA, Lambert GW. Stress and its role in sympathetic nervous system
activation in hypertension and the metabolic syndrome. Curr Hypertens Rep. 2011;13(3):244-8,
http://dx.doi.org/10.1007/s11906-011-0186-y.
http://dx.doi.org/10.1007/s11906-011-018...
27. Kotsis V, Stabouli S, Papakatsika S, Rizos Z, Parati G. Mechanisms of
obesity-induced hypertension. Hypertens Res. 2010;33(5):386-93,
http://dx.doi.org/10.1038/hr.2010.9.
http://dx.doi.org/10.1038/hr.2010.9...
-2828. Flaa A, Aksnes TA, Kjeldsen SE, Eide I, Rostrup M. Increased sympathetic
reactivity may predict insulin resistance: an 18-year follow-up study. Metabolism.
2008;57(10):1422-7, http://dx.doi.org/10.1016/j.metabol.2008.05.012.
http://dx.doi.org/10.1016/j.metabol.2008...
). Regardless of the
obesity parameter, the importance of obesity in inducing high blood pressure and metabolic and
inflammatory dysregulation is clear.
Elevated sympathetic nervous system activity seems to be crucial to the development of
obesity-related risk (2929. Jamerson KA, Nesbitt SD, Amerena JV, Grant E, Julius S. Angiotensin mediates
forearm glucose uptake by hemodynamic rather than direct effects. Hypertension. 1996;27(4):854-8,
http://dx.doi.org/10.1161/01.HYP.27.4.854.
http://dx.doi.org/10.1161/01.HYP.27.4.85...
). In our study, patients with MetS
and high blood pressure had higher surrogate markers of sympathetic activity. Long-term sympathetic
activation can lead to small artery vasoconstriction and remodeling, which increases the
wall-to-lumen ratio and may act synergistically with large artery damage to raise blood pressure
(3030. Jamerson KA, Smith SD, Amerena JV, Grant E, Julius S. Vasoconstriction with
norepinephrine causes less forearm insulin resistance than a reflex sympathetic vasoconstriction.
Hypertension. 1994;23(6 Pt 2):1006-11, http://dx.doi.org/10.1161/01.HYP.23.6.1006.
http://dx.doi.org/10.1161/01.HYP.23.6.10...
). Sympathetic activity is also related to baroreflex
impairment, as observed in this study. However, sympathetic activity may not be related only to
hemodynamic impairment but also to metabolic and inflammatory dysregulation.
In addition to the hemodynamic effects, sympathetically mediated vasoconstriction may also
antagonize insulin-mediated glucose uptake (2929. Jamerson KA, Nesbitt SD, Amerena JV, Grant E, Julius S. Angiotensin mediates
forearm glucose uptake by hemodynamic rather than direct effects. Hypertension. 1996;27(4):854-8,
http://dx.doi.org/10.1161/01.HYP.27.4.854.
http://dx.doi.org/10.1161/01.HYP.27.4.85...
). This theory
is supported by longitudinal studies demonstrating that elevated indices of sympathetic activation
precede the development of insulin resistance (3131. Pollare T, Lithell H, Selinus I, Berne C. Application of prazosin is associated
with an increase of insulin sensitivity in obese patients with hypertension. Diabetologia.
1988;31(7):415-20, http://dx.doi.org/10.1007/BF00271585.
http://dx.doi.org/10.1007/BF00271585...
). Jamerson
et al. showed that norepinephrine infusion, but not angiotensin, impaired insulin-stimulated glucose
uptake (2929. Jamerson KA, Nesbitt SD, Amerena JV, Grant E, Julius S. Angiotensin mediates
forearm glucose uptake by hemodynamic rather than direct effects. Hypertension. 1996;27(4):854-8,
http://dx.doi.org/10.1161/01.HYP.27.4.854.
http://dx.doi.org/10.1161/01.HYP.27.4.85...
30. Jamerson KA, Smith SD, Amerena JV, Grant E, Julius S. Vasoconstriction with
norepinephrine causes less forearm insulin resistance than a reflex sympathetic vasoconstriction.
Hypertension. 1994;23(6 Pt 2):1006-11, http://dx.doi.org/10.1161/01.HYP.23.6.1006.
http://dx.doi.org/10.1161/01.HYP.23.6.10...
-3131. Pollare T, Lithell H, Selinus I, Berne C. Application of prazosin is associated
with an increase of insulin sensitivity in obese patients with hypertension. Diabetologia.
1988;31(7):415-20, http://dx.doi.org/10.1007/BF00271585.
http://dx.doi.org/10.1007/BF00271585...
). In
line with the metabolic dysregulation observed in our study, Lichtand et al. found that autonomic
nervous system dysregulation was strongly associated with MetS and its components in a large cohort
of participants aged 18-65 years (3535. Wirtz PH, Ehlert U, Bartschi C, Redwine LS, von Kanel R. Changes in plasma
lipids with psychosocial stress are related to hypertension status and the norepinephrine stress
response. Metabolism. 2009;58(1):30-7,
http://dx.doi.org/10.1016/j.metabol.2008.08.003.
http://dx.doi.org/10.1016/j.metabol.2008...
). However, these authors
did not stratify patients with MetS according to blood pressure status and did not explore other
risk factors not included in the MetS definition, such as proinflammatory and prothrombotic
markers.
In our study, we also observed that adiponectin levels were lower and that markers of
inflammation were higher in MetS patients with high blood pressure than in MetS patients without
high blood pressure. Sympathetic activation reportedly reduces adiponectin release, whereas
central-sympathetic blockade may increase adiponectin levels (3636. Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr et al.
Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment
of High Blood Pressure. Hypertension. 2003;42(6):1206-52,
http://dx.doi.org/10.1161/01.HYP.0000107251.49515.c2.
http://dx.doi.org/10.1161/01.HYP.0000107...
). Plasma adiponectin is inversely related to insulin resistance (3535. Wirtz PH, Ehlert U, Bartschi C, Redwine LS, von Kanel R. Changes in plasma
lipids with psychosocial stress are related to hypertension status and the norepinephrine stress
response. Metabolism. 2009;58(1):30-7,
http://dx.doi.org/10.1016/j.metabol.2008.08.003.
http://dx.doi.org/10.1016/j.metabol.2008...
). Thus, the increased sympathetic activity in patients with MetS and high blood
pressure may decrease adiponectin levels that, in turn, can also contribute to insulin resistance.
Systemic inflammation may also be linked to sympathetic activity. Previous evidence found that
indirect markers of sympathetic activity (increased heart rate and reduced heart-rate variability)
were associated with subclinical inflammation in healthy middle-aged and elderly subjects. These
results suggest that an autonomic imbalance in favor of the sympathetic system may interact with
inflammatory processes to play a more important role in the process of vascular stiffness and
atherosclerosis.
A few limitations from this investigation should be addressed. First, due to the study design,
our findings suggest an association rather than a cause-effect relationship between high blood
pressure and metabolic, inflammatory, prothrombotic and autonomic impairments in patients with MetS.
Although consistent with the cited literature, we cannot prove that sympathetic activity is the main
obesity-related trigger underlying not only the increased blood pressure but also impairments in the
metabolic, inflammatory and prothrombotic profiles in this subgroup of patients with MetS. Second,
direct measurements of peripheral sympathetic activity using microneurography were not performed.
However, spectral analysis is an acceptable non-invasive method for estimating sympathovagal
activity (1313. Pagani M, Somers V, Furlan R, Dell'Orto S, Conway J, Baselli G, et al.
Changes in autonomic regulation induced by physical training in mild hypertension. Hypertension.
1988;12(6):600-10, http://dx.doi.org/10.1161/01.HYP.12.6.600.
http://dx.doi.org/10.1161/01.HYP.12.6.60...
). Our findings consistently suggest a decrease
in vagal activity and the activation of the sympathetic system in the spectral analyses of both
heart rate and blood pressure. Third, these results cannot be extrapolated to MetS patients with
diabetes. Finally, the precise reasons by which a subset of patients with MetS had higher blood
pressure and greater sympathetic activity despite similar indices of obesity are not clear. Recent
studies reported the potential role of psychosocial stress in inducing sympathetic activity (2929. Jamerson KA, Nesbitt SD, Amerena JV, Grant E, Julius S. Angiotensin mediates
forearm glucose uptake by hemodynamic rather than direct effects. Hypertension. 1996;27(4):854-8,
http://dx.doi.org/10.1161/01.HYP.27.4.854.
http://dx.doi.org/10.1161/01.HYP.27.4.85...
,3737. Drager LF, Bortolotto LA, Figueiredo AC, Krieger EM, Lorenzi GF. Effects of
continuous positive airway pressure on early signs of atherosclerosis in obstructive sleep apnea.
Am J Respir Crit Care Med. 2007;176(7):706-12,
http://dx.doi.org/10.1164/rccm.200703-500OC.
http://dx.doi.org/10.1164/rccm.200703-50...
,3838. Trombetta IC, Somers VK, Maki-Nunes C, Drager LF, Toschi-Dias E, Alves MJ, et
al. Consequences of comorbid sleep apnea in the metabolic syndrome-implications for cardiovascular
risk. Sleep. 2010;33(9):1193-9.). Another potential candidate is obstructive sleep apnea. This clinical
condition is characterized by sympathetic activation that is independent of obesity, and it is a
well-established cause of high blood pressure and increased arterial stiffness (3939. Drager LF, Lopes HF, Maki-Nunes C, Trombetta IC, Toschi-Dias E, Alves MJ, et al.
The impact of obstructive sleep apnea on metabolic and inflammatory markers in consecutive patients
with metabolic syndrome. PLoS One. 2010;5(8):e12065,
http://dx.doi.org/10.1371/journal.pone.0012065.
http://dx.doi.org/10.1371/journal.pone.0...
40. Drager LF, Bortolotto LA, Figueiredo AC, Silva BC, Krieger EM, Lorenzi-Filho G.
Obstructive sleep apnea, hypertension, and their interaction on arterial stiffness and heart
remodeling. Chest. 2007;131(5):1379-86, http://dx.doi.org/10.1378/chest.06-2703.
http://dx.doi.org/10.1378/chest.06-2703...
-4141. Wofford MR, Anderson DC, Jr., Brown CA, Jones DW, Miller ME, Hall JE.
Antihypertensive effect of alpha- and beta-adrenergic blockade in obese and lean hypertensive
subjects. Am J Hypertens. 2001;14(7 Pt 1):694-8.). Recent evidence in
consecutive patients with MetS suggests that obstructive sleep apnea is quite common and is
independently associated with increased sympathetic activity (4141. Wofford MR, Anderson DC, Jr., Brown CA, Jones DW, Miller ME, Hall JE.
Antihypertensive effect of alpha- and beta-adrenergic blockade in obese and lean hypertensive
subjects. Am J Hypertens. 2001;14(7 Pt 1):694-8.), metabolic and inflammatory dysregulation (4242. Witkowski A, Prejbisz A, Florczak E, KĄdziela J, Śliwiński
P, Bielen P, et al. Effects of renal sympathetic denervation on blood pressure, sleep apnea course,
and glycemic control in patients with resistant hypertension and sleep apnea. Hypertension.
2011;58(4):559-65.)
and increased markers of atherosclerosis (4343. Drager LF, Bortolotto LA, Maki-Nunes C,Trombetta IC, Alves MJ, Fraga RF, et al.
The incremental role of obstructive sleep apnoea on markers of atherosclerosis in patients with
metabolic syndrome. Atherosclerosis. 2010;208(2):490-5,
http://dx.doi.org/10.1016/j.atherosclerosis.2009.08.016.
http://dx.doi.org/10.1016/j.atherosclero...
). These
potential confounders should be addressed in future investigations.
In conclusion, this study provides several provocative new findings that suggest the importance
of characterizing patients with MetS according to the presence or absence of high blood pressure.
Although we cannot prove a cause-effect relationship, our results demonstrated that patients with
MetS and high blood pressure have higher surrogate markers of sympathetic activity and significant
metabolic, inflammatory and prothrombotic impairments. Thus, it is reasonable to speculate that
blocking sympathetic activity is an attractive strategy for treating patients with MetS. A previous
study showed that a combined α- and β-adrenergic blockade significantly reduced blood
pressure in obese patients compared to lean patients with essential hypertension (4444. Wofford MR, Anderson DC Jr., Brown CA, Jones DW, Miller ME, Hall JE.
Antihypertensive effect of alpha- and beta-adrenergic blockade in obese and lean hypertensive
subjects. Am J Hypertens. 2001;14(7 Pt 1):694-8.). Moreover, renal denervation, which reduces sympathetic
activity, lowers blood pressure and reduces insulin resistance, may improve obstructive sleep apnea
(4545. Witkowski A, Prejbisz A, Florczak E, Kadziela J, Sliwinski P, Bielen P, et al.
Effects of renal sympathetic denervation on blood pressure, sleep apnea course, and glycemic control
in patients with resistant hypertension and sleep apnea. Hypertension. 2011;58(4):559-65,
http://dx.doi.org/10.1161/HYPERTENSIONAHA.111.173799.
http://dx.doi.org/10.1161/HYPERTENSIONAH...
). Future studies will clarify whether pharmacological
suppression of sympathetic activity in patients with MetS will improve not only blood pressure but
also the metabolic, inflammatory and prothrombotic dysfunctions observed in these patients.
This work was supported by grants from FAPESP (Fundação de Amparo à Pesquisa de São Paulo) and Zerbini Foundation.
REFERENCES
-
1Reaven GM. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes. 1988;37(12):1595-607, http://dx.doi.org/10.2337/diab.37.12.1595.
» http://dx.doi.org/10.2337/diab.37.12.1595 -
2Smith S, Julius S, Jamerson K, Amerena J, Schork N. Hematocrit levels and physiologic factors in relationship to cardiovascular risk in Tecumseh, Michigan. J Hypertens. 1994;12(4):455-62.
-
3Kannel WB. Risk stratification in hypertension: new insights from the Framingham Study. Am J Hypertens. 2000;13(1Pt 2):3S-10S.
-
4Houston MC. New insights and approaches to reduce end-organ damage in the treatment of hypertension: subsets of hypertension approach. Am Heart J. 1992;123(5):1337-67, http://dx.doi.org/10.1016/0002-8703(92)91042-Y.
» http://dx.doi.org/10.1016/0002-8703(92)91042-Y -
5Chapman N, Chen CY, Fujita T, Hobbs FD, Kim SJ, Staessen JA, et al. Time to re-appraise the role of alpha-1 adrenoceptor antagonists in the management of hypertension? J Hypertens. 2010;28:1796-803. Erratum in: J Hypertens. 2010;28(11):2351.
-
6Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002;106(25):3143-421.
-
7Pimentel GD, Bernhard AB, Frezza MR, Rinaldi AE, Burini RC. Bioelectric impedance overestimates the body fat in overweight and underestimates in Brazilian obese women: a comparison with Segal equation 1. Nutr Hosp. 2010;25(5):741-5.
-
8Schipper HS, de Jager W, van Dijk ME, Meerding J, Zelissen PM, Adan RA, et al. Clin Chem. 2010;56(8):1320-8, http://dx.doi.org/10.1373/clinchem.2010.146118.
» http://dx.doi.org/10.1373/clinchem.2010.146118 -
9Drager LF, Ueno LM, Lessa PS, Negrão CE, Lorenzi-Filho G, Krieger EM. Sleep-related changes in hemodynamic and autonomic regulation in human hypertension. J Hypertens. 2009;27(8):1655-63, http://dx.doi.org/10.1097/HJH.0b013e32832c6982.
» http://dx.doi.org/10.1097/HJH.0b013e32832c6982 -
10Pagani M, Lombardi F, Guzzetti S, Rimoldi O, Furlan R, Pizzinelli P, et al. Power spectral analysis of heart rate and arterial pressure variabilities as a marker of sympatho-vagal interaction in man and conscious dog. Circ Res. 1986;59(2):178-93, http://dx.doi.org/10.1161/01.RES.59.2.178.
» http://dx.doi.org/10.1161/01.RES.59.2.178 -
11Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation. 1996;93(5):1043-65.
-
12Lucini D, Cusumano G, Bellia A, Kozakova M, Difede G, Lauro R, et al. Is reduced baroreflex gain a component of the metabolic syndrome? Insights from the LINOSA study. J Hypertens. 2006;24(2):361-70, http://dx.doi.org/10.1097/01.hjh.0000202817.02836.9c.
» http://dx.doi.org/10.1097/01.hjh.0000202817.02836.9c -
13Pagani M, Somers V, Furlan R, Dell'Orto S, Conway J, Baselli G, et al. Changes in autonomic regulation induced by physical training in mild hypertension. Hypertension. 1988;12(6):600-10, http://dx.doi.org/10.1161/01.HYP.12.6.600.
» http://dx.doi.org/10.1161/01.HYP.12.6.600 -
14Hamilton M. The assessment of anxiety states by rating. Br J Med Psychol. 1959;32(1):50-5.
-
15Maier W, Buller R, Philipp M, Heuser I. The Hamilton Anxiety Scale: reliability, validity and sensitivity to change in anxiety and depressive disorders. J Affect Disord. 1988;14(1):61-8, http://dx.doi.org/10.1016/0165-0327(88)90072-9.
» http://dx.doi.org/10.1016/0165-0327(88)90072-9 -
16Gomes-Oliveira MH, Gorenstein C, Lotufo Neto F, Andrade LH, Wang YP. Validation of the Brazilian Portuguese version of the Beck Depression Inventory-II in a community sample. Rev. Bras. Psiquiatr. 2012;34(4):389-94, http://dx.doi.org/10.1016/j.rbp.2012.03.005.
» http://dx.doi.org/10.1016/j.rbp.2012.03.005 -
17Graham TE, Yang Q, Blüher M, Hammarstedt A, Ciaraldi TP, Henry RR, et al. Retinol-binding protein 4 and insulin resistance in lean, obese, and diabetic subjects. N Engl J Med. 2006;354(24):2552-63.
-
18Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care. 2004;27(6):1487-95, http://dx.doi.org/10.2337/diacare.27.6.1487.
» http://dx.doi.org/10.2337/diacare.27.6.1487 -
19Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin RA, et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112(17):2735-52, http://dx.doi.org/10.1161/CIRCULATIONAHA.105.169404.
» http://dx.doi.org/10.1161/CIRCULATIONAHA.105.169404 -
20Janssen I, Katzmarzyk PT, Ross R. Waist circumference and not body mass index explains obesity-related health risk. Am J Clin Nutr. 2004;79(3):379-84.
-
21Lean ME, Han TS, Morrison CE. Waist circumference as a measure for indicating need for weight management. BMJ. 1995;311(6998):158-61, http://dx.doi.org/10.1136/bmj.311.6998.158.
» http://dx.doi.org/10.1136/bmj.311.6998.158 -
22Costa GB, Horta N, Resende ZF, Souza G, Barreto LM, Correia LH, et al. Body mass index has a good correlation with proatherosclerotic profile in children and adolescents. Arq Bras Cardiol. 2009;93(3):261-7, http://dx.doi.org/10.1590/S0066-782X2009000900010.
» http://dx.doi.org/10.1590/S0066-782X2009000900010 -
23Katzmarzyk PT, Srinivasan SR, Chen W, Malina RM, Bouchard C, Berenson GS. Body mass index, waist circumference, and clustering of cardiovascular disease risk factors in a biracial sample of children and adolescents. Pediatrics. 2004;114(2):e198-205, http://dx.doi.org/10.1542/peds.114.2.e198.
» http://dx.doi.org/10.1542/peds.114.2.e198 -
24Schneider HJ, Glaesmer H, Klotsche J, Böhler S, Lehnert H, Zeiher AM, et al. J Clin Endocrinol Metab. 2007;92(2):589-94.
-
25Yusuf S, Hawken S, Ounpuu S, Bautista L, Franzosi MG, Commerford P, et al. Obesity and the risk of myocardial infarction in 27,000 participants from 52 countries: a case-control study. Lancet. 2005;366(9497):1640-9, http://dx.doi.org/10.1016/S0140-6736(05)67663-5.
» http://dx.doi.org/10.1016/S0140-6736(05)67663-5 -
26Lambert EA, Lambert GW. Stress and its role in sympathetic nervous system activation in hypertension and the metabolic syndrome. Curr Hypertens Rep. 2011;13(3):244-8, http://dx.doi.org/10.1007/s11906-011-0186-y.
» http://dx.doi.org/10.1007/s11906-011-0186-y -
27Kotsis V, Stabouli S, Papakatsika S, Rizos Z, Parati G. Mechanisms of obesity-induced hypertension. Hypertens Res. 2010;33(5):386-93, http://dx.doi.org/10.1038/hr.2010.9.
» http://dx.doi.org/10.1038/hr.2010.9 -
28Flaa A, Aksnes TA, Kjeldsen SE, Eide I, Rostrup M. Increased sympathetic reactivity may predict insulin resistance: an 18-year follow-up study. Metabolism. 2008;57(10):1422-7, http://dx.doi.org/10.1016/j.metabol.2008.05.012.
» http://dx.doi.org/10.1016/j.metabol.2008.05.012 -
29Jamerson KA, Nesbitt SD, Amerena JV, Grant E, Julius S. Angiotensin mediates forearm glucose uptake by hemodynamic rather than direct effects. Hypertension. 1996;27(4):854-8, http://dx.doi.org/10.1161/01.HYP.27.4.854.
» http://dx.doi.org/10.1161/01.HYP.27.4.854 -
30Jamerson KA, Smith SD, Amerena JV, Grant E, Julius S. Vasoconstriction with norepinephrine causes less forearm insulin resistance than a reflex sympathetic vasoconstriction. Hypertension. 1994;23(6 Pt 2):1006-11, http://dx.doi.org/10.1161/01.HYP.23.6.1006.
» http://dx.doi.org/10.1161/01.HYP.23.6.1006 -
31Pollare T, Lithell H, Selinus I, Berne C. Application of prazosin is associated with an increase of insulin sensitivity in obese patients with hypertension. Diabetologia. 1988;31(7):415-20, http://dx.doi.org/10.1007/BF00271585.
» http://dx.doi.org/10.1007/BF00271585 -
32Licht CM, Vreeburg SA, van Reedt Dortland AK, Giltay EJ, Hoogendijk WJ, DeRijk RH, et al. Increased sympathetic and decreased parasympathetic activity rather than changes in hypothalamic-pituitary-adrenal axis activity is associated with metabolic abnormalities. J Clin Endocrinol Metab. 2010;95(5):2458-66, http://dx.doi.org/10.1210/jc.2009-2801.
» http://dx.doi.org/10.1210/jc.2009-2801 -
33Weyer C, Funahashi T, Tanaka S, Hotta K, Matsuzawa Y, Pratley RE, et al. Hypoadiponectinemia in obesity and type 2 diabetes: close association with insulin resistance and hyperinsulinemia. J Clin Endocrinol Metab. 2001;86(5):1930-5, http://dx.doi.org/10.1210/jc.86.5.1930.
» http://dx.doi.org/10.1210/jc.86.5.1930 -
34Lambert E, Dawood T, Straznicky N, Sari C, Schlaich M, Esler M, et al. Association between the sympathetic firing pattern and anxiety level in patients with the metabolic syndrome and elevated blood pressure. J Hypertens. 2010;28(3):543-50, http://dx.doi.org/10.1097/HJH.0b013e3283350ea4.
» http://dx.doi.org/10.1097/HJH.0b013e3283350ea4 -
35Wirtz PH, Ehlert U, Bartschi C, Redwine LS, von Kanel R. Changes in plasma lipids with psychosocial stress are related to hypertension status and the norepinephrine stress response. Metabolism. 2009;58(1):30-7, http://dx.doi.org/10.1016/j.metabol.2008.08.003.
» http://dx.doi.org/10.1016/j.metabol.2008.08.003 -
36Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension. 2003;42(6):1206-52, http://dx.doi.org/10.1161/01.HYP.0000107251.49515.c2.
» http://dx.doi.org/10.1161/01.HYP.0000107251.49515.c2 -
37Drager LF, Bortolotto LA, Figueiredo AC, Krieger EM, Lorenzi GF. Effects of continuous positive airway pressure on early signs of atherosclerosis in obstructive sleep apnea. Am J Respir Crit Care Med. 2007;176(7):706-12, http://dx.doi.org/10.1164/rccm.200703-500OC.
» http://dx.doi.org/10.1164/rccm.200703-500OC -
38Trombetta IC, Somers VK, Maki-Nunes C, Drager LF, Toschi-Dias E, Alves MJ, et al. Consequences of comorbid sleep apnea in the metabolic syndrome-implications for cardiovascular risk. Sleep. 2010;33(9):1193-9.
-
39Drager LF, Lopes HF, Maki-Nunes C, Trombetta IC, Toschi-Dias E, Alves MJ, et al. The impact of obstructive sleep apnea on metabolic and inflammatory markers in consecutive patients with metabolic syndrome. PLoS One. 2010;5(8):e12065, http://dx.doi.org/10.1371/journal.pone.0012065.
» http://dx.doi.org/10.1371/journal.pone.0012065 -
40Drager LF, Bortolotto LA, Figueiredo AC, Silva BC, Krieger EM, Lorenzi-Filho G. Obstructive sleep apnea, hypertension, and their interaction on arterial stiffness and heart remodeling. Chest. 2007;131(5):1379-86, http://dx.doi.org/10.1378/chest.06-2703.
» http://dx.doi.org/10.1378/chest.06-2703 -
41Wofford MR, Anderson DC, Jr., Brown CA, Jones DW, Miller ME, Hall JE. Antihypertensive effect of alpha- and beta-adrenergic blockade in obese and lean hypertensive subjects. Am J Hypertens. 2001;14(7 Pt 1):694-8.
-
42Witkowski A, Prejbisz A, Florczak E, KĄdziela J, Śliwiński P, Bielen P, et al. Effects of renal sympathetic denervation on blood pressure, sleep apnea course, and glycemic control in patients with resistant hypertension and sleep apnea. Hypertension. 2011;58(4):559-65.
-
43Drager LF, Bortolotto LA, Maki-Nunes C,Trombetta IC, Alves MJ, Fraga RF, et al. The incremental role of obstructive sleep apnoea on markers of atherosclerosis in patients with metabolic syndrome. Atherosclerosis. 2010;208(2):490-5, http://dx.doi.org/10.1016/j.atherosclerosis.2009.08.016.
» http://dx.doi.org/10.1016/j.atherosclerosis.2009.08.016 -
44Wofford MR, Anderson DC Jr., Brown CA, Jones DW, Miller ME, Hall JE. Antihypertensive effect of alpha- and beta-adrenergic blockade in obese and lean hypertensive subjects. Am J Hypertens. 2001;14(7 Pt 1):694-8.
-
45Witkowski A, Prejbisz A, Florczak E, Kadziela J, Sliwinski P, Bielen P, et al. Effects of renal sympathetic denervation on blood pressure, sleep apnea course, and glycemic control in patients with resistant hypertension and sleep apnea. Hypertension. 2011;58(4):559-65, http://dx.doi.org/10.1161/HYPERTENSIONAHA.111.173799.
» http://dx.doi.org/10.1161/HYPERTENSIONAHA.111.173799
-
No potential conflict of interest was reported.
Publication Dates
-
Publication in this collection
Dec 2013
History
-
Received
27 Apr 2013 -
Reviewed
7 May 2013 -
Accepted
11 June 2013