Abstract
The brachioradialis is an important muscle that acts in the external rotation of the forearm (supination). However, its occurrence is controversial and little studied in the order Carnivora. Thus, this study investigates the occurrence and anatomo-functional arrangement of this muscle in wild carnivorans species. Fifty-eight thoracic limbs of specimens from species of Canidae, Procyonidae, Mustelidae and Felidae were dissected. Measurements of the length of the muscle (ML), the length of the forearm (FL), latero-medial width of the muscle (MW) and the lateral-medial diameter of the forearm (FD) were obtained to establish the ratios MW/FD and ML/FL in order to investigate the relative proportion of the muscle in relation to the forearm of each species. The brachioradialis muscle was identified in all species, although it was unilaterally or bilaterally absent in some canid individuals. The ratios demonstrated significant differences in the anatomical proportions among the families, with greater functional importance in the mustelids, procyonids, and felids because of a set of elaborate movements in the thoracic limb of representatives of these families when compared to canids.
Comparative anatomy; forelimb; myology
Since thoracic limbs are not only used in locomotion, but also in prey capture and grooming
and mating behavior, their morphology can be a good predictor of numerous ecological
variables, such as the size and kind of prey, the variety of movements, the role in
supporting body mass (Andersson 2004Andersson K (2004a) Predicting carnivoran body mass from a
weight-bearing joint. Journal of Zoology 262(2): 161-172. doi:
10.1017/S0952836903004564
https://doi.org/10.1017/S095283690300456...
a, Fabre et al. 2013Fabre AC, Cornette R, Peigné S, Goswami A (2013a) Influence of body mass
on the shape of forelimb in musteloid carnivorans. Biological Journal of the Linnean
Society 110: 91-103. doi: 10.1111/bij.12103
https://doi.org/10.1111/bij.12103...
a) and the habitat (Davis 1964Davis DD (1964) The giant panda: a morphological study of evolutionary
mechanisms. Fieldiana: Zoology Memors 3: 1-339., Ewer et al.
1973Ewer RF (1973) The Carnivores. New York, Cornell University Press,
494p., Taylor 1989Taylor ME (1989) Locomotor adaptations by carnivores, p. 382-409. In:
Gittleman JL (Ed.) Carnivore Behavior, Ecology, and Evolution. Cornell, Comstosck
Publiching Associates, 620p., Polly 2007Polly PD (2007) Limbs in mammalian evolution, p. 245-268. In: Hall BK
(Ed.) Fins into Limbs: Evolution, Development, and Transformation. Chicago,
University of Chicago Press, 344p., Meachen-Samuels &
Van-Valkenburgh 2009Meachen-Samuels JA, Van-Valkenburgh B (2009) Forelimb indicators of
prey-size preference in the Felidae. Journal of Morphology 270(6): 729-744. doi:
10.1002/jmor.10712
https://doi.org/10.1002/jmor.10712...
, Fabre et al. 2013Fabre AC, Cornette R, Peigné S, Goswami A (2013a) Influence of body mass
on the shape of forelimb in musteloid carnivorans. Biological Journal of the Linnean
Society 110: 91-103. doi: 10.1111/bij.12103
https://doi.org/10.1111/bij.12103...
a,
et al. 2013Fabre AC, Cornette R, Slater G, Argot C, Peigné S, Goswami A, Pouydebat
E (2013b) Getting a grip on the evolution of grasping in musteloid carnivorans: a
three-dimensional analysis of forelimb shape. Journal of Evolutionary Biology 26:
1521-1535. doi: 10.1111/jeb.12161
https://doi.org/10.1111/jeb.12161...
b, Meloro et al. 2013Meloro C, Elton C, Louys J, Bishop LC, Ditchfield P (2013) Cats in the
forest: predicting habitat adaptations from humerus morphometry in extant and fossil
Felidae (Carnivora). Paleobiology 39(3): 323-244. doi: 10.1666/12001
https://doi.org/10.1666/12001...
, Martín-Serra et al.
2014Martín-Serra A, Figueirido B, Palmqvist P (2014) A Three-Dimen sional
Analysis of Morphological Evolution and Locomotor Performance of the Carnivoran
Forelimb. PLoS ONE 9: e85574. doi: 10.1371/journal.pone.0085574
https://doi.org/10.1371/journal.pone.008...
). Together with cranio-dental data, data on thoracic limbs are also used to
extrapolate the predatory behavior of extinct species (Iwaniuk et al. 1999Iwaniuk AN, Pellis SM, Whishaw IQ (1999) The relationship between
forelimb morphology and behaviour in North American carnivores (Carnivora). Canadian
Journal of Zoology 77(7): 1064-1074. doi: 10.1139/z99-082
https://doi.org/10.1139/z99-082...
, Andersson & Werdelin
2003Andersson K, Werdelin L (2003) The evolution of cursorial carnivores in
the Tertiary: implications of elbow joint morphology. Proceeding of the Biological
Society 270(Suppl.): S163-S165. doi: 10.1098/rsbl.2003.0070
https://doi.org/10.1098/rsbl.2003.0070...
). Most ecomorphology studies have prioritized the osteological
characteristics of the humerus, whereas the shape and other features of the radio-ulnar
joint remain largely unstudied (Fabre et al. 2013Fabre AC, Cornette R, Peigné S, Goswami A (2013a) Influence of body mass
on the shape of forelimb in musteloid carnivorans. Biological Journal of the Linnean
Society 110: 91-103. doi: 10.1111/bij.12103
https://doi.org/10.1111/bij.12103...
a,
b, 2014Fabre AC, Goswami A, Peigné S, Cornette R (2014) Morphological
integration in the forelimb of musteloid carnivorans. Journal of Anatomy 225: 19-30.
doi: 10.1111/joa.12194
https://doi.org/10.1111/joa.12194...
). Moreover, the muscular arrangement is
rarely taken into account. Knowledge about muscular disposition associated with some
biomechanical findings can better elucidate the function of some important bone structures
that would otherwise be neglected (Julik et al.
2012Julik E, Zack S, Adrian B, Maredia S, Parsa A, Poole M, Starbuck A,
Fisher RE (2012) Functional Anatomy of the Forelimb Muscles of the Ocelot (Leopardus
pardalis). Journal of Mammalian Evolution 19(4): 277-304. doi:
10.1007/s10914-012-9191-4
https://doi.org/10.1007/s10914-012-9191-...
). Several studies in this field have biases because of misunderstanding of the
muscular topography. Thus, the addition of quantitative and qualitative data on muscular
anatomy would contribute to improve the capacity to characterize forelimb morphology in the
context of locomotion, grasping ability and dexterity of the species (Iwaniuk et al. 2001Iwaniuk AN, Pellis SM, Whishaw IQ (2001) Are long digits correlated with
high forepaw dexterity? A comparative test in terrestrial carnivores (Carnivora).
Canadian Journal of Zoology 79(5): 900-906. doi: 10.1139/z01-058
https://doi.org/10.1139/z01-058...
, Fabre et al.
2013Fabre AC, Cornette R, Slater G, Argot C, Peigné S, Goswami A, Pouydebat
E (2013b) Getting a grip on the evolution of grasping in musteloid carnivorans: a
three-dimensional analysis of forelimb shape. Journal of Evolutionary Biology 26:
1521-1535. doi: 10.1111/jeb.12161
https://doi.org/10.1111/jeb.12161...
b). By homology, soft tissue information from extant species can help making
well-founded or even speculative inferences about extinct species (Witmer 1995Witmer LM (1995) The extant phylogenetic bracket and the importance of
reconstructing soft tissues in fossils, p. 19-33. In: Thomason JJ (Ed.) Functional
morphology in vertebrate paleontology. Cambridge, Cambridge University Press,
277p.).
The musculoskeletal system forms an arrangement based on levers in which the joints act as fulcra (Hermanson 2013Hermanson JW (2013) The Muscular System, p. 185-280. In: Evans HE, DeLahunta A (Eds) Miller's Anatomy of the Dog. Missouri, Elsevier, 872p.). The mechanical benefits of its configuration depend on the positions of the muscle attachments (relative to the fulcrum) and the usage of the load. A muscle attached close to a fulcrum is less powerful than a comparable muscle inserted at a greater distance, although the former produces its effects faster. This reflects a conflict between the requirements of speed and power (Dyce et al. 2010Dyce KM, Sack WO, Wensing CJG (2010) Tratado de Anatomia Veterinária. São Paulo, Elsevier, 856p.).
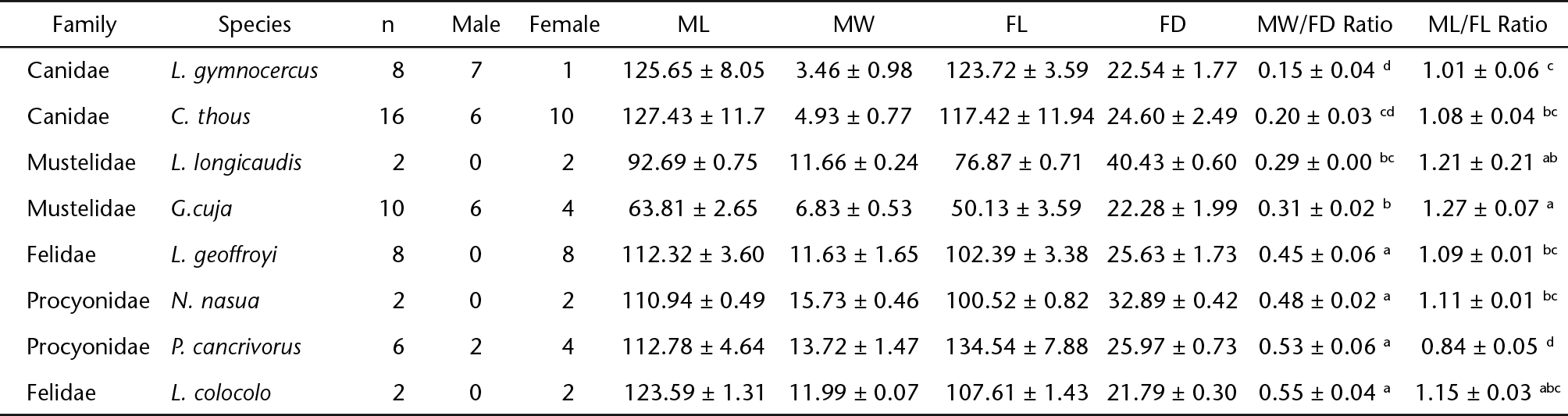
The brachioradialis muscle (formerly called the supinator longus) usually consists of a narrow muscular band situated at the flexor angle of the humerus-radius-ulna joint (Fig. 1) (Budras et al. 2012Budras KD, McCarthy PH, Fricke W, Richter R, Horowitz A, Berg R (2012) Anatomia do Cão, Texto e Atlas. Barueri, Manole, 219p., Hermanson 2013Hermanson JW (2013) The Muscular System, p. 185-280. In: Evans HE, DeLahunta A (Eds) Miller's Anatomy of the Dog. Missouri, Elsevier, 872p.). It is positioned between the superficial and deep layers of antebrachial fascia and adheres to the surface of the deep fascia's leaflet (Mills 2003Mills P (2003) Comparative Animal Anatomy. Queensland, Gatton Desktop Publishing, 316p., Dyce et al. 2010Dyce KM, Sack WO, Wensing CJG (2010) Tratado de Anatomia Veterinária. São Paulo, Elsevier, 856p., Hermanson 2013Hermanson JW (2013) The Muscular System, p. 185-280. In: Evans HE, DeLahunta A (Eds) Miller's Anatomy of the Dog. Missouri, Elsevier, 872p.), together with the cephalic vein and the superficial branch of the radial nerve (Saint Clair 1986Saint Clair LE (1986) Músculos do carnívoro, p. 1416-1444. In: Getty R (Ed.) Anatomia dos Animais Domésticos. Rio de Janeiro, Guanabara Koogan, 2048p., Sebastiani & Fishbeck 2005Sebastiani AM, Fishbeck DW (2005) Mammalian anatomy: the cat. Colorado, Morton Publishing Company, 184p.), therefore being the most cranial and superficial muscle of the craniolateral group of the forearm (Bohensky 2002Bohensky F (2002) Photo manual and dissection Guide of the Cat. New York, Square One Publishers, 167p., Sebastiani & Fishbeck 2005Sebastiani AM, Fishbeck DW (2005) Mammalian anatomy: the cat. Colorado, Morton Publishing Company, 184p.).
(1) Schematic representation of the basic arrangement of brachioradialis muscle in the domestic dog. (2) Schematic representations of the measurement points. ML: muscle length; FL: forearm length; MW: muscle width; FD: forearm diameter.
In domestic carnivorans, this muscle has its origin at the proximal extremity of the
humeral lateral supracondylar crest, immediately proximal and superficial to the extensor
carpi radialis muscle (Schwarze 1984Schwarze E (1984) Compendio de Anatomia Veterinaria. Zaragoza, Editora
Acribia, 318p., Mills 2003Mills P (2003) Comparative Animal Anatomy. Queensland, Gatton Desktop
Publishing, 316p., Liebich et
al. 2011Liebich HG, Maierl J, König HE (2011) Membros Torácicos ou Anteriores
(Membra Thoracica), p. 165-234. In:, König HE Liebich HG (Eds) Anatomia dos Animais
Domésticos: Texto e Atlas Colorido. Porto Alegre, Artmed, 787p., Hermanson 2013Hermanson JW (2013) The Muscular System, p. 185-280. In: Evans HE,
DeLahunta A (Eds) Miller's Anatomy of the Dog. Missouri, Elsevier,
872p.). The muscle
extends cranially over the proximal part of the extensor carpi radialis muscle, crosses the
forearm medially, and extends distally in the groove between the extensor carpi radialis
muscle and the radius (Schwarze 1984Schwarze E (1984) Compendio de Anatomia Veterinaria. Zaragoza, Editora
Acribia, 318p., Liebich et al. 2011Liebich HG, Maierl J, König HE (2011) Membros Torácicos ou Anteriores
(Membra Thoracica), p. 165-234. In:, König HE Liebich HG (Eds) Anatomia dos Animais
Domésticos: Texto e Atlas Colorido. Porto Alegre, Artmed, 787p., Hermanson 2013Hermanson JW (2013) The Muscular System, p. 185-280. In: Evans HE,
DeLahunta A (Eds) Miller's Anatomy of the Dog. Missouri, Elsevier,
872p.). It ends in the periosteum of the radius at the level of the
third or fourth distal parts, by a thin aponeurosis (Bohensky 2002Bohensky F (2002) Photo manual and dissection Guide of the Cat. New
York, Square One Publishers, 167p., Mills 2003Mills P (2003) Comparative Animal Anatomy. Queensland, Gatton Desktop
Publishing, 316p., Budras et al. 2012Budras KD, McCarthy PH, Fricke W, Richter R, Horowitz A, Berg R (2012)
Anatomia do Cão, Texto e Atlas. Barueri, Manole, 219p., Hermanson 2013Hermanson JW (2013) The Muscular System, p. 185-280. In: Evans HE,
DeLahunta A (Eds) Miller's Anatomy of the Dog. Missouri, Elsevier,
872p.). Some authors describe its insertion into the styloid process of
the radius (Leach 1976Leach D (1976) The forelimb musculature of marten (Martes americana
Turton) and fisher (Martes pennanti Erxleben). Canadian Journal of Zoology 55(1):
31-41. doi: 10.1139/z77-00
https://doi.org/10.1139/z77-00...
, Sebastian & Fishbeck 2005Sebastiani AM, Fishbeck DW (2005) Mammalian anatomy: the cat. Colorado,
Morton Publishing Company, 184p., Liebich
et al. 2011Liebich HG, Maierl J, König HE (2011) Membros Torácicos ou Anteriores
(Membra Thoracica), p. 165-234. In:, König HE Liebich HG (Eds) Anatomia dos Animais
Domésticos: Texto e Atlas Colorido. Porto Alegre, Artmed, 787p., Moore et al. 2013Moore AL, Budny JE, Russel AP, Butcher MT (2013) Architectural
specialization of the intrinsic thoracic limb musculature of the American badger
(Taxidea taxus). Journal of Morphology 274(1): 35-48. doi:
10.1002/jmor.20074
https://doi.org/10.1002/jmor.20074...
, Ercoli et al. 2014Ercoli MD, Álvarez A, Stefanini MI, Busker F, Morales MM (2014) Muscular
Anatomy of the Forelimbs of the Lesser Grison (Galictis cuja), and a Functional and
Phylogenetic Overview of Mustelidae and Other Caniformia. Journal of Mammalian
Evolution 22(1): 57-91. doi: 10.1007/s10914-014-9257-6
https://doi.org/10.1007/s10914-014-9257-...
).
The function of the brachioradialis muscle is to perform the craniolateral rotation of the radius (supination) (Bohensky 2002Bohensky F (2002) Photo manual and dissection Guide of the Cat. New York, Square One Publishers, 167p., Sebastiani & Fishbeck 2005Sebastiani AM, Fishbeck DW (2005) Mammalian anatomy: the cat. Colorado, Morton Publishing Company, 184p., Hermanson 2013Hermanson JW (2013) The Muscular System, p. 185-280. In: Evans HE, DeLahunta A (Eds) Miller's Anatomy of the Dog. Missouri, Elsevier, 872p.).
Supination is a movement of flipping the distal radius over the distal ulna, rotating the
radius craniolaterally around its long axis (Andersson
2004Andersson K (2004b) Elbow-joint morphology as a guide to forearm
function and foraging behaviour in mammalian carnivores. Zoological Journal of the
Linnean Society 142: 91-104. doi: 10.1111/j.1096-3642.2004.00129.x
https://doi.org/10.1111/j.1096-3642.2004...
b). The movement starts with the contraction of forearm muscles (supinator
and brachioradialis), transmitting external rotation also to the manus. Cursorial mammals
often have restricted pronation-supination, whereas scansorial mammals can usually
completely supinate the manus (Polly 2007Polly PD (2007) Limbs in mammalian evolution, p. 245-268. In: Hall BK
(Ed.) Fins into Limbs: Evolution, Development, and Transformation. Chicago,
University of Chicago Press, 344p.). Thus,
the brachioradialis functionally belongs to the group of muscles that act on the
radio-ulnar joint, and is expected to be well developed only in carnivorans. In domestic
ungulates, this muscle is vestigial or absent due to the reduced or lost capacity of
movement between these two bones (Polly 2007Polly PD (2007) Limbs in mammalian evolution, p. 245-268. In: Hall BK
(Ed.) Fins into Limbs: Evolution, Development, and Transformation. Chicago,
University of Chicago Press, 344p., Liebich et al. 2011Liebich HG, Maierl J, König HE (2011) Membros Torácicos ou Anteriores
(Membra Thoracica), p. 165-234. In:, König HE Liebich HG (Eds) Anatomia dos Animais
Domésticos: Texto e Atlas Colorido. Porto Alegre, Artmed, 787p.).
In domestic dogs, the occurrence and antimeric distribution of the brachioradialis muscle
has been well documented by Wakuri & Kano
(1966Wakuri H, Kano Y (1966) Anatomical studies on the brachioradial muscle
in dogs. Acta Anatomica Nipponica 41: 222-231.), Santos Junior et al. (2002Santos Junior I, Rodrigues CA, Campos A, Santos D (2002) Presença do
músculo braquiorradial em cães. Bioscience Journal 18(1): 79-83.), and
Pestana et al. (2009Pestana FM, Silva BX, Chagas MA, Babinski MA, Abidu-Figueiredo M (2009)
Distribuição antimérica do músculo braquiorradial em cães sem raça definida. Revista
de Ciências da Vida 29(1): 55-59.). There are no reports in
the literature, as far as we know, about the occurrence of the brachioradialis muscle in
wild carnivorans, although there are studies with variable contexts containing references
to the presence of the muscle in Carnivora (Davis
1964Davis DD (1964) The giant panda: a morphological study of evolutionary
mechanisms. Fieldiana: Zoology Memors 3: 1-339., Arlamowska-Palider 1970Arlamowska-Palider A (1970) Morphological Studies on the Main Branches
of the Radial Nerve in Mammals. Acta Theriologica 15(2): 185-197., Julik et al. 2012Julik E, Zack S, Adrian B, Maredia S, Parsa A, Poole M, Starbuck A,
Fisher RE (2012) Functional Anatomy of the Forelimb Muscles of the Ocelot (Leopardus
pardalis). Journal of Mammalian Evolution 19(4): 277-304. doi:
10.1007/s10914-012-9191-4
https://doi.org/10.1007/s10914-012-9191-...
, Sánchez et al. 2013Sánchez HL, Silva LB, Rafasquino ME, Mateo AG, Zuccolilli GO, Portiansky
EL, Alonso CR (2013) Anatomical study of the forearm and hand nerves of the domestic
cat (Felis catus), puma (Puma concolor) and jaguar (Panthera onca). Anatomia
Histologia Embryologia 42(2): 99-104. doi:
10.1111/j.1439-0264.2012.01170.x
https://doi.org/10.1111/j.1439-0264.2012...
, Ercoli et al.
2014Ercoli MD, Álvarez A, Stefanini MI, Busker F, Morales MM (2014) Muscular
Anatomy of the Forelimbs of the Lesser Grison (Galictis cuja), and a Functional and
Phylogenetic Overview of Mustelidae and Other Caniformia. Journal of Mammalian
Evolution 22(1): 57-91. doi: 10.1007/s10914-014-9257-6
https://doi.org/10.1007/s10914-014-9257-...
).
Carnivorans form a successful and functionally diverse clade, with close to 300 living
species (Ewer 1973Ewer RF (1973) The Carnivores. New York, Cornell University Press,
494p., Wilson & Mittermeier 2009Wilson DE, Mittermeier RA (2009) Handbook of the Mammals of the World.
Barcelona, Lynx Edicions, vol. 1, 727p., Hunter
2011Hunter L (2011) Carnivores of the world. Princeton, Princeton University
Press, 240p.). Despite this diversity, the accumulated knowledge of myological variation
within the order is still incomplete (Macalister
1873Macalister A (1873a) The muscular anatomy of the civet and tayra.
Proceedings of the Royal Irish Academy Academy Series 2: 506-513.a, Mackintosh 1875Mackintosh BA (1875) Notes on the myology of the coati mondth (Nasua
narica and N. fusca) and common marten (Martes foina). Proceedings of the Royal lrish
Academy Series 2: 48-55., Windle & Parsons 1897Windle BCA, Parsons FG (1897) On the myology of the terrestrial
Carnivora. Part I: muscles of the head, neck, and fore-limb. Proceedings of
Zoological Society of London 65: 370-409., Hall 1926Hall ER (1926). The muscular anatomy of three mustelid mammals,
Mephitis, Spilogale, and Martes. University of California Publications in Zoology
30(2): 7-39., 1927Hall ER (1927) The muscular anatomy of the American badger (Taxidea
taxus). University of California Publications in Zoology 30(8):
205-219., Howard 1973Howard LD (1973) Muscular anatomy of the fore-limb of the sea otter
(Enhydra lutris). Proceedings of the California Academia of Science 39(4):
411-500., Leach
1976Leach D (1976) The forelimb musculature of marten (Martes americana
Turton) and fisher (Martes pennanti Erxleben). Canadian Journal of Zoology 55(1):
31-41. doi: 10.1139/z77-00
https://doi.org/10.1139/z77-00...
, Fischer et al. 2009Fisher RE, Adrian B, Barton M, Holmgren J, Tang SY (2009) The phylogeny
of the red panda (Ailurus fulgens): evidence from the forelimb. Journal of Anatomy
215(6): 611-635. doi: 10.1111/j.1469-7580.2009.01156.x
https://doi.org/10.1111/j.1469-7580.2009...
, Moore et al. 2013Moore AL, Budny JE, Russel AP, Butcher MT (2013) Architectural
specialization of the intrinsic thoracic limb musculature of the American badger
(Taxidea taxus). Journal of Morphology 274(1): 35-48. doi:
10.1002/jmor.20074
https://doi.org/10.1002/jmor.20074...
, Ercoli et al. 2014Ercoli MD, Álvarez A, Stefanini MI, Busker F, Morales MM (2014) Muscular
Anatomy of the Forelimbs of the Lesser Grison (Galictis cuja), and a Functional and
Phylogenetic Overview of Mustelidae and Other Caniformia. Journal of Mammalian
Evolution 22(1): 57-91. doi: 10.1007/s10914-014-9257-6
https://doi.org/10.1007/s10914-014-9257-...
). Hence, these animals provide a good model for this study,
as they represent one of the most successful cases of repeated and independent evolution of
similar morphologies in a great range of ecologies (Andersson 2004Andersson K (2004a) Predicting carnivoran body mass from a
weight-bearing joint. Journal of Zoology 262(2): 161-172. doi:
10.1017/S0952836903004564
https://doi.org/10.1017/S095283690300456...
a, b, 2005Andersson K (2005) Were there pack-hunting canids in the Tertiary, and
how can we know? Paleobiology 31(1): 56-72. doi:
10.1666/0094-8373(2005)031<0056:WTPCIT>2.0.CO;2
https://doi.org/10.1666/0094-8373(2005)0...
, Nowak 2005Nowak RM (2005) Walker's Carnivores of the World. Baltimore, The Johns
Hopkins University Press, 328p., Sato et al.
2009Sato JJ, Wolsan M, Minami S, Hosoda T, Sinaga MH, Hiyama K, Yamaguchi Y,
Suzuki H (2009) Deciphering and dating the red panda's ancestry and early adaptive
radiation of Musteloidea. Molecular Phylogenetics and Evolution 53(3): 907-922. doi:
10.1016/j.ympev.2009.08.019
https://doi.org/10.1016/j.ympev.2009.08....
, 2012Sato JJ, Wolsan M, Prevosti FJF, D'Elía G, Begg C, Begg K, Hosoda T,
Campbell KL, Suzuki H (2012) Evolutionary and biogeographic history of Weasellike
carnivorans (Musteloidea). Molecular Phylogenetics and Evolution 63(3): 745-757. doi:
10.1016/j.ympev.2012.02.025
https://doi.org/10.1016/j.ympev.2012.02....
, Slater et al. 2012Slater GJ, Harmon LJ, Alfaro ME (2012) Integrating fossils with
molecular phylogenies improves inference of trait evolution. Evolution 66(12):
3931-3944. doi: 10.1111/j.1558-5646.2012.01723.x
https://doi.org/10.1111/j.1558-5646.2012...
, Fabre et al.
2013Fabre AC, Cornette R, Peigné S, Goswami A (2013a) Influence of body mass
on the shape of forelimb in musteloid carnivorans. Biological Journal of the Linnean
Society 110: 91-103. doi: 10.1111/bij.12103
https://doi.org/10.1111/bij.12103...
a, 2014Fabre AC, Goswami A, Peigné S, Cornette R (2014) Morphological
integration in the forelimb of musteloid carnivorans. Journal of Anatomy 225: 19-30.
doi: 10.1111/joa.12194
https://doi.org/10.1111/joa.12194...
, Samuels et al. 2013Samuels JX, Meachen JA, Sakai SA (2013) Postcranial morphology and the
locomotor habits of living and extinct carnivorans. Journal of Morphology 274(2):
121-146. doi: 10.1002/jmor.20077
https://doi.org/10.1002/jmor.20077...
, Martín-Serra et
al. 2014Martín-Serra A, Figueirido B, Palmqvist P (2014) A Three-Dimen sional
Analysis of Morphological Evolution and Locomotor Performance of the Carnivoran
Forelimb. PLoS ONE 9: e85574. doi: 10.1371/journal.pone.0085574
https://doi.org/10.1371/journal.pone.008...
). The locomotor range of movements of carnivorans includes, to varying
extents, climbing, digging, running and swimming (Andersson
& Werdelin 2003Andersson K, Werdelin L (2003) The evolution of cursorial carnivores in
the Tertiary: implications of elbow joint morphology. Proceeding of the Biological
Society 270(Suppl.): S163-S165. doi: 10.1098/rsbl.2003.0070
https://doi.org/10.1098/rsbl.2003.0070...
). Furthermore, carnivorans species show different degrees of
supination and some species cannot even use the forelimbs for grappling with or handling
prey (Ewer 1973Ewer RF (1973) The Carnivores. New York, Cornell University Press,
494p., Andersson & Werdelin 2003Andersson K, Werdelin L (2003) The evolution of cursorial carnivores in
the Tertiary: implications of elbow joint morphology. Proceeding of the Biological
Society 270(Suppl.): S163-S165. doi: 10.1098/rsbl.2003.0070
https://doi.org/10.1098/rsbl.2003.0070...
). We hypothesized that the life style of
carnivorans, including cursoriality and food procurement strategies, should be reflected in
changes in brachioradialis muscle arrangement. This muscle is expected to be frequently
found and to be relatively larger in species that need to rotate the forelimbs. Thereby,
the aim of this study is to verify the occurrence, anatomo-functional arrangement and
sexual dimorphism of the brachioradialis muscle in wild carnivoran species, thus
contributing to studies in ecomorphology.
MATERIAL AND METHODS
This study was carried out with 29 carnivorans cadavers of Canidae: Cerdocyon thous (Linnaeus, 1766) (three males and six females) and Lycalopex gymnocercus (G. Fischer, 1814) (four males and one female); Mustelidae: Galictis cuja (Molina, 1782) (three males and two females) and Lontra longicaudis (Olfers, 1818) (one female); Procyonidae: Procyon cancrivorus (G.[Baron] Cuvier, 1798) (one male and two females) and Nasua nasua (Linnaeus, 1766) (one female); and Felidae: Leopardus geoffroyi (d'Orbigny & Gervais, 1844) (four females) and Leopardus colocolo (Molina, 1782) (one female). These specimens were collected dead from highways in the southwest region of the state of Rio Grande do Sul (Pampa biome) between July 2012 and November 2013 (IBAMA/SISBIO authorization number 33667-1). Only adult individuals were included, based on inspection of permanent dentition.
After collection, the specimens were fixed in a formaldehyde solution (50%) and conserved in opaque polyethylene tanks with the same solution at 10% for at least 14 days, until they were dissected. The skin and fascia of the thoracic limbs were carefully removed and the superficial intrinsic muscles identified. The brachioradialis muscle, whenever present, was dissected until its origin and insertion were exposed. Then measurements were taken (Fig. 2) of the length of the brachioradialis muscle from its origin until its insertion (ML) and the length of the forearm from the olecranon tuberosity until the radiocarpian joint (FL). Also, lateral-medial width of the brachioradialis muscle (MW) and the lateral-medial diameter of the forearm (FD) were obtained at the level of their middle thirds. The measurements were performed by a single examiner using a digital pachymeter (resolution 0.01mm, accuracy ± 0.02 mm, ZAAS Precision Amatools(r)). Thereafter, two ratios were calculated: MW/FD and ML/FL. The MW/FD ratio was calculated to reflect the relative proportion of the functional participation of the brachioradialis muscle in the forearm region of the specimens. The ML/FL ratio represents the proportion of muscle length in relation to the forearm length and can be associated with added speed during contraction. The 29 specimens are deposited in the Laboratory of Animal Anatomy of the Universidade Federal do Pampa, Uruguaiana, RS, Brazil. The deposit numbers of the specimens analyzed are available in the appendix APPENDIX Appendix. The specimens analyzed are deposited in the Laboratory of Animal Anatomy of Universidade Federal do Pampa, Uruguaiana, RS, Brazil, with the following numbers: Cerdocyon thous: 5139, 5144, 5267, 5273, 5275, 5717, 8503, 8504, 8505 Lycalopex gymnocercus: 5134, 5261, 5269, 5274, 5603 Procyon cancrivorus: 5136, 5268, 8517 Galictis cuja: 5142, 5146, 5599, 5714, 5720 Leopardus geoffroyi: 5138, 5145, 5147, 5150 Nasua nasua: 8527 Lontra longicaudis: 8507 Leopardus colocolo: 5137 .
Descriptive statistical data (mean, standard deviation, variance and coefficient of variation) were calculated. The ratios were compared among species and families using analysis of variance (one-way ANOVA) and significant differences between the means were determined by using the Tukey test at 99% probability. In species with enough samples for comparison of the ratios bet ween genders (C. thous, G. cuja and P. cancrivorus), the t-test was performed at 99% probability. These tests were executed by the BioEstat 5.3(r) program. Photomacrographs were taken with a Sony Cybershot DSC-TF1(r) camera with 16.1 MP and the images were treated with the Photoscape(r) v.3.5 software.
RESULTS
Except for two specimens of L. gymnocercus, in which one male only had the muscle on the right antimere and a female only on the left, along with a female of C. thous that did not have the muscle in any antimere, all the other specimens (26) had the brachioradialis muscle in both antimeres. Therefore, among all 58 thoracic limbs analyzed, 54 presented the muscle (Table I).
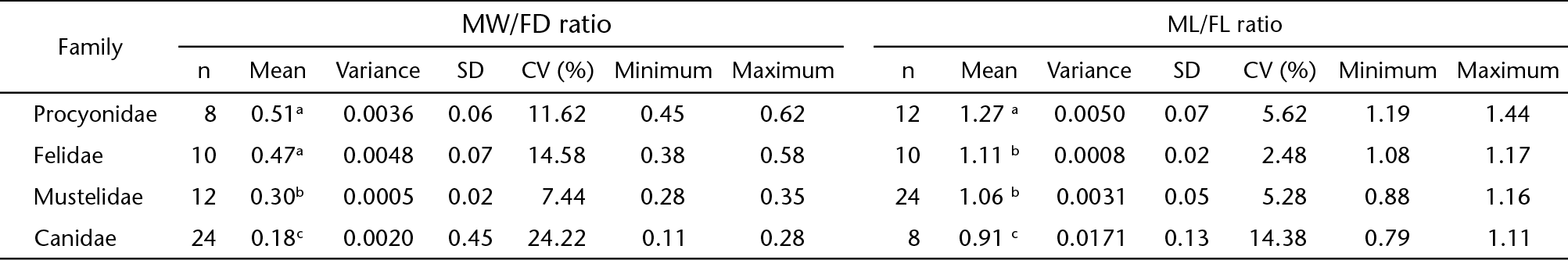
The results of the MW/FD and ML/FL ratios grouped by family (Canidae, Mustelidae, Procyonidae and Felidae) are presented in Table II and in Figs. 3 and 4.
Box-plots showing the mean ± SD of the MW/FD ratio (3) and ML/FL ratio (4) grouped by families of carnivorans analyzed.
In canids, felids and procyonids, the brachioradialis muscle originated on the lateral supracondylar crest of the humerus and its insertion was on the medial surface of the radial distal extremity (medial styloid process) in every specimen analyzed (Figs. 5-12), except one male specimen of L. gymnocercus, in which the right brachioradialis muscle was shortened and joined to the middle third of the extensor carpi radialis muscle. In mustelids, the muscle originated from the caudal surface of the humeral neck and caudomedially to the brachial muscle point of origin, and also was inserted in the styloid process of the radius, being very fleshy in its course. In procyonids, especially in P. cancrivorus, the muscular part was restricted until the middle third of the forearm, where it narrowed into a thin insertion tendon (Fig. 7). In every specimen it was the most superficial muscle in the forearm, transiting near the cephalic vein, and was innervated by branches of the radial nerve.
Photomacrographs showing the comparative anatomic arrangement of the brachioradialis muscle (white arrow) in the forearm region of: (5) Cerdocyon thous, right forearm; (6) Lycalopex gymnocercus, left forearm; (7) Procyon cancrivorus, right forearm; (8) Nasua nasua, right of forearm; (9) Leopardus geoffroyi, left forearm; (10) Leopardus colocolo, right forearm; (11) Lontra longicaudis, left forearm; (12) Galictis cuja, left forearm. Scale bars: 20 mm.
The t-test (p < 0.01) for comparison of means of the MW/FD ratios revealed similarity between genders in C. thous (p = 0.2219), G. cuja (p = 0.7273) and P. cancrivorus (p = 0.0986). The same test for comparison of means of the ML/FL ratios revealed similarity between genders in C. thous (p = 0.4026) and G. cuja (p = 0.1743) and difference in P. cancrivorus (p = 0.0024).
DISCUSSION
The presence in nearly all the specimens assessed in this study reflects the functional relevance of the brachioradialis muscle to the order Carnivora. Its occurrence is expected in species that require significant mobility in the radius-ulnar joint, especially in external rotation (supination) of the hand (paw) and forearm, which does not happen, for instance, in ungulates (Nickel et al. 1986Nickel R, Schummer A, Seiferle E, Frewein J, Wilkens H, Wille K (1986) The Locomotor System of Domestic Mammals. Berlin, Verlag Paul Parey, 515p., Liebich et al. 2011Liebich HG, Maierl J, König HE (2011) Membros Torácicos ou Anteriores (Membra Thoracica), p. 165-234. In:, König HE Liebich HG (Eds) Anatomia dos Animais Domésticos: Texto e Atlas Colorido. Porto Alegre, Artmed, 787p.). According to Saladin (2010Saladin KS (2010) Anatomy and Physiology: The Unity of Form and Function. New York, McGraw Hill, 1248p.), the brachioradialis muscle also acts as a synergist in the flexion of the humerus-radio-ulnar joint, but by itself it is not able to generate enough strength because its insertion is far from the fulcrum.
Only among canids were individuals identified that did not present the muscle
unilaterally or bilaterally (one C. thous and two L.
gymnocercus). Besides this, in one male specimen of L. gymnocercus
the brachioradialis muscle was not long enough to be inserted in the radius,
joining the extensor carpi radialis muscle in a clearly accessory position. The absence
or presence, unilaterally or bilaterally, of the brachioradialis muscle has also been
reported in C. familiaris by Wakuri
& Kano (1966Wakuri H, Kano Y (1966) Anatomical studies on the brachioradial muscle
in dogs. Acta Anatomica Nipponica 41: 222-231.), Santos Junior et al.
(2002Santos Junior I, Rodrigues CA, Campos A, Santos D (2002) Presença do
músculo braquiorradial em cães. Bioscience Journal 18(1): 79-83.) and Pestana et al. (2009Pestana FM, Silva BX, Chagas MA, Babinski MA, Abidu-Figueiredo M (2009)
Distribuição antimérica do músculo braquiorradial em cães sem raça definida. Revista
de Ciências da Vida 29(1): 55-59.).
Considered together, these three studies reveal that the brachioradialis muscle is
absent in 46 to 62% of dogs, appearing unilaterally in 15 to 20% of cases. In running
dogs of the greyhound breed, the muscle was not found in ten individuals dissected by
Williams et al. (2008Williams SB, Wilson AM, Rhodes L, Andrews J, Payne RC (2008) Functional
anatomy and muscle moment arms of the thoracic limb of an elite sprinting athlete:
the racing greyhound (Canis familiaris). Journal of Anatomy 213(4): 361-372. doi:
10.1111/j.1469-7580.2008.00962.x
https://doi.org/10.1111/j.1469-7580.2008...
). These findings
corroborate the well-recognized observation that canids have lost some of the ability to
supinate their manus (Ewer 1973Ewer RF (1973) The Carnivores. New York, Cornell University Press,
494p., Andersson & Werdelin 2003Andersson K, Werdelin L (2003) The evolution of cursorial carnivores in
the Tertiary: implications of elbow joint morphology. Proceeding of the Biological
Society 270(Suppl.): S163-S165. doi: 10.1098/rsbl.2003.0070
https://doi.org/10.1098/rsbl.2003.0070...
). Therefore, in a
superficial analysis considering the canids, the brachioradialis muscle seems to be more
frequent in wild ones than in domestic ones, perhaps because the former animals retain a
greater need to perform supination movements to capture prey. Nevertheless, the
unilateral and even bilateral absence of this muscle in some individuals suggests that
it may no longer perform a useful function and instead is a rudimentary muscle that is
on its way out. The apparent absence of this muscle in greyhound dogs could reflect the
increased intensity of artificial selection to eliminate a useless muscle, since it can
interfere with a desirable running ability in wild canids.
After examining some specimens, W.J. Gonyea (unpubl. data) noted that the
brachioradialis muscle was present in arboreal and fossorial taxa and absent in
cursorial ones. In support of this hypothesis, the muscle was not identified in the
cursorial canids Canis latrans (Say, 1823) (n = 1), Urocyon
cinereoargenteus (Schreber, 1775) (n = 4) and Vulpes vulpes
(Linnaeus, 1758) (n = 5) (S.A. Feeney unpubl. data). The appearance of the
brachioradialis muscle in almost all cursorial canids C. thous (n =
8/9) and L. gymnocercus (n = 5/5) analyzed in this study contrasts with
previous observation (W.J. Gonyea, unpubl. data). Until there are more investigations,
we can propose that the presence of this muscle in canids may be a charac teristic
shared by Neotropical species with close phylogenetic relationship rather than just a
morphofunctional issue. Supporting this hypothesis, the presence of the brachioradialis
muscle was also mentioned by Vaz et al. (2011Vaz MGR, Lima AR, Souza ACB, Pereira LC, Branco E (2011) Estudo
morfológico dos músculos do antebraço de cachorro-do-mato-de-orelhas-curtas
(Atelocynus microtis) e cachorro-do-mato (Cerdocyon thous). Biotemas 24(4): 121-127.
doi: 10.5007/2175-7925.2011v24n4p121
https://doi.org/10.5007/2175-7925.2011v2...
) in
an adult female Atelocynus microtis (Sclater, 1883) and in an adult
male C. thous. Indeed, C. thous, L.
gymnocercus and A. microtis inhabit the Neotropics and
have the same number of chromosomes (74) (Pessutti et
al. 2001Pessutti C, Santiago MEB, Oliveira LTF (2001) Order Carnivora, Family
Canidae (Dogs, foxes and maned wolves), p. 279-290. In: Fowler ME, Cubas ZS (Eds)
Biology, Medicine and Surgery of South American Wild Animals. Ames, Iowa State
University Press, 550p.), unlike C. latrans, U.
cinereoargenteus and V. vulpes (S.A. Feeney, unpubl.
data).
Despite disagreement over the cursorial canids, observations of the presence of the
muscle in arboreal and fossorial carnivorans are compatible with the findings of this
study. In fact, the mustelids, procyonids and felids analyzed here have scansorial,
arboreal and fossorial habits (Reis et al. 2010Reis NR, Peracchi AL, Fregonezi MN, Rossaneis BK (2010) Mamíferos do
Brasil: guia de identificação. Rio de Janeiro, Technical Books,
560p.,
Hunter 2011Hunter L (2011) Carnivores of the world. Princeton, Princeton University
Press, 240p.). Mustelids and procyonids in
particular can display great ability to perform different kinds of movements such as
grasping, swimming and food manipulation (McClearn
1992McClearn D (1992) Locomotion, Posture, and feeding behavior of
kinkajous, coatis, and raccoons. Journal of Mammalogy 73(2):
245-261., Iwaniuk et al. 1999Iwaniuk AN, Pellis SM, Whishaw IQ (1999) The relationship between
forelimb morphology and behaviour in North American carnivores (Carnivora). Canadian
Journal of Zoology 77(7): 1064-1074. doi: 10.1139/z99-082
https://doi.org/10.1139/z99-082...
, Fabre et al. 2013Fabre AC, Cornette R, Slater G, Argot C, Peigné S, Goswami A, Pouydebat
E (2013b) Getting a grip on the evolution of grasping in musteloid carnivorans: a
three-dimensional analysis of forelimb shape. Journal of Evolutionary Biology 26:
1521-1535. doi: 10.1111/jeb.12161
https://doi.org/10.1111/jeb.12161...
b), and their lack of cursorial
adaptation means that none of them have lost the ability to supinate their paws, in
contrast to other carnivorans such as canids and some hyaenids (Iwaniuk et al. 1999Iwaniuk AN, Pellis SM, Whishaw IQ (1999) The relationship between
forelimb morphology and behaviour in North American carnivores (Carnivora). Canadian
Journal of Zoology 77(7): 1064-1074. doi: 10.1139/z99-082
https://doi.org/10.1139/z99-082...
, Polly
2007Polly PD (2007) Limbs in mammalian evolution, p. 245-268. In: Hall BK
(Ed.) Fins into Limbs: Evolution, Development, and Transformation. Chicago,
University of Chicago Press, 344p., Fabre et al. 2014Fabre AC, Goswami A, Peigné S, Cornette R (2014) Morphological
integration in the forelimb of musteloid carnivorans. Journal of Anatomy 225: 19-30.
doi: 10.1111/joa.12194
https://doi.org/10.1111/joa.12194...
). Although with
different purposes and small samples, some other studies have mentioned the occurrence
of this muscle in species of these families, for instance in the mustelids Aonyx
cinerea (Illiger, 1815) (Macalister
1873Macalister A (1873b) On the anatomy of Aonyx. Proceedings of the Royal
Irish Academy Series 2: 539-547.b), Lutra lutra (Linnaeus, 1758) (Windle & Parsons 1897Windle BCA, Parsons FG (1897) On the myology of the terrestrial
Carnivora. Part I: muscles of the head, neck, and fore-limb. Proceedings of
Zoological Society of London 65: 370-409.), Enhydra lutris
(Linnaeus, 1758) (Howard 1973Howard LD (1973) Muscular anatomy of the fore-limb of the sea otter
(Enhydra lutris). Proceedings of the California Academia of Science 39(4):
411-500.), Martes
pennanti (Erxleben, 1777) (n = 1) (S.A. Feeney, unpubl. data) and G.
cuja (n = 3) (Ercoli et al. 2014Ercoli MD, Álvarez A, Stefanini MI, Busker F, Morales MM (2014) Muscular
Anatomy of the Forelimbs of the Lesser Grison (Galictis cuja), and a Functional and
Phylogenetic Overview of Mustelidae and Other Caniformia. Journal of Mammalian
Evolution 22(1): 57-91. doi: 10.1007/s10914-014-9257-6
https://doi.org/10.1007/s10914-014-9257-...
); in
the procyonid Procyon lotor (Linnaeus, 1758) (n = 2) (S.A. Feeney,
unpubl. data); and in the felids Puma concolor (Linnaeus, 1771) (n = 2)
(Concha et al. 2004Concha I, Adaro L, Borroni C, Altamirano C (2004) Consideraciones
anatómicas sobre la musculatura intrínseca del miembro torácico del puma (Puma
concolor). International Journal of Morphology 22(2): 121-125. doi:
10.4067/S0717-95022004000200004
https://doi.org/10.4067/S0717-9502200400...
), Acinonyx
jubatus (Schreber, 1775) (n = 8) (Hudson et
al. 2011Hudson PE, Corr SA, Payne-Davis RC, Clancy SN, Lane E, Wilson AM (2011)
Functional anatomy of the cheetah (Acinonyx jubatus) forelimb. Journal of Anatomy
218(4): 375-385. doi: 10.1111/j.1469-7580.2011.01344.x
https://doi.org/10.1111/j.1469-7580.2011...
), Leopardus pardalis (Linnaeus, 1758) (n = 1) (Julik et al. 2012Julik E, Zack S, Adrian B, Maredia S, Parsa A, Poole M, Starbuck A,
Fisher RE (2012) Functional Anatomy of the Forelimb Muscles of the Ocelot (Leopardus
pardalis). Journal of Mammalian Evolution 19(4): 277-304. doi:
10.1007/s10914-012-9191-4
https://doi.org/10.1007/s10914-012-9191-...
) and Panthera
onca (Linnaeus, 1758) (n = 2) (Sánchez et
al. 2013Sánchez HL, Silva LB, Rafasquino ME, Mateo AG, Zuccolilli GO, Portiansky
EL, Alonso CR (2013) Anatomical study of the forearm and hand nerves of the domestic
cat (Felis catus), puma (Puma concolor) and jaguar (Panthera onca). Anatomia
Histologia Embryologia 42(2): 99-104. doi:
10.1111/j.1439-0264.2012.01170.x
https://doi.org/10.1111/j.1439-0264.2012...
).
The superficial location in the forearm, the proximity of the cephalic vein and innervation by branches of the radial nerve were common to all specimens analyzed, resembling the description of domestic carnivores (Sebastiani & Fishbeck 2005Sebastiani AM, Fishbeck DW (2005) Mammalian anatomy: the cat. Colorado, Morton Publishing Company, 184p., Budras et al. 2012Budras KD, McCarthy PH, Fricke W, Richter R, Horowitz A, Berg R (2012) Anatomia do Cão, Texto e Atlas. Barueri, Manole, 219p.).
The MW/FD ratio was calculated to reflect the relative proportion of its participation
in the forearm region of the specimens. This way, when comparing species and/or
families, those with the highest ratios should be the ones in which the brachioradialis
muscle has greater relative contribution through the group of antebrachium muscles.
Although calculation of physiological cross-section area (PCSA) has been used to
estimate the maximum isometric force of muscles (Williams et al. 2008Williams SB, Wilson AM, Rhodes L, Andrews J, Payne RC (2008) Functional
anatomy and muscle moment arms of the thoracic limb of an elite sprinting athlete:
the racing greyhound (Canis familiaris). Journal of Anatomy 213(4): 361-372. doi:
10.1111/j.1469-7580.2008.00962.x
https://doi.org/10.1111/j.1469-7580.2008...
), in this study we considered this determination to be
less important since it generates an absolute value which is highly influenced by the
body size of the individuals. For example, the brachioradialis of a crab-eating fox
(C. thous) would exhibit both higher absolute PCSA and maximum
isometric force values than the muscle in a lesser grison (G. cuja).
Howe ver, the muscle is proportionally weaker in the former. Since the brachioradialis
has parallel fibers, determination of the ratio between cross section widths (MW) to the
forearm diameter would give a more proportional estimate of functional rele vance, at
least in its topographic region (forearm). In fact, the results of MW/FD ratio reflected
observations from the usage of the forelimbs in each species.
The significantly lower MW/FD ratio in canids compared to the other three families used
in this study can be explained by the fact that canids are essentially terrestrial and
have developed a highly specialized lifestyle among carnivorans. In more cursorial taxa,
the functional adaptations prioritize, among other aspects, the movements of the limbs
in the sagittal plane, disfavoring supination or pronation (Ewer 1973Ewer RF (1973) The Carnivores. New York, Cornell University Press,
494p.). In contrast to felids, canids have a limited ability to
subdue and grapple with other animals by using their forelimbs. Instead they engage in
sustained pursuit predation, an activity whose success depends on the number of animals
participating in the hunt (Andersson 2005Andersson K (2005) Were there pack-hunting canids in the Tertiary, and
how can we know? Paleobiology 31(1): 56-72. doi:
10.1666/0094-8373(2005)031<0056:WTPCIT>2.0.CO;2
https://doi.org/10.1666/0094-8373(2005)0...
). In
addition, canids rarely climb or manipulate prey to a higher extent. These habits are
correlated to a less functional, or even absent, brachioradialis muscle.
In the mustelids G. cuja and L. longicaudis the MW/FD ratio, significantly higher only than in the canids, comparatively reflects greater recruitment of the muscle to help in specific swimming movements and fossorial habits. At times, they also use their thoracic limbs to drag prey out of the water (Reis et al. 2010Reis NR, Peracchi AL, Fregonezi MN, Rossaneis BK (2010) Mamíferos do Brasil: guia de identificação. Rio de Janeiro, Technical Books, 560p.).
The felids and procyonids of this study showed the highest MW/FD ratios, with no
differences among them. The reper toire of manual movements of these two families is
more complex because, besides being fast runners, they are also able to swim, climb
trees and remain balanced at tall heights (Reis et al.
2010Reis NR, Peracchi AL, Fregonezi MN, Rossaneis BK (2010) Mamíferos do
Brasil: guia de identificação. Rio de Janeiro, Technical Books,
560p., Hunter 2011Hunter L (2011) Carnivores of the world. Princeton, Princeton University
Press, 240p.). Undeniably,
scansorial habits and grasping requires accurate three-dimensional movements, which
demand morphofunctional adaptations and higher recruitment and precision in muscular
contraction (Ewer 1973Ewer RF (1973) The Carnivores. New York, Cornell University Press,
494p., Fabre et al. 2013Fabre AC, Cornette R, Slater G, Argot C, Peigné S, Goswami A, Pouydebat
E (2013b) Getting a grip on the evolution of grasping in musteloid carnivorans: a
three-dimensional analysis of forelimb shape. Journal of Evolutionary Biology 26:
1521-1535. doi: 10.1111/jeb.12161
https://doi.org/10.1111/jeb.12161...
b). Procyonids even use their hands to precisely
bring food into their mouths, requiring a greater capacity for supination (Paranaíba et al. 2012Paranaiba JF, Helrigle C, Araújo EG, Pereira KF (2012) Aspectos
morfológicos da mão e pé de Procyon cancrivorus. Natureza On line 10(4): 165-169.
Available online at:
http://www.naturezaonline.com.br/natureza/conteudo/pdf/03_ParanaibaJFetal_165_169.pdf.
[Accessed: 31 July 2014]
http://www.naturezaonline.com.br/naturez...
). Species showing
well-developed grasping ability potentially have a wide range of pronation-supination
movements, which can confer greater mobility to the forearm and the hand (Fabre et al. 2013Fabre AC, Cornette R, Slater G, Argot C, Peigné S, Goswami A, Pouydebat
E (2013b) Getting a grip on the evolution of grasping in musteloid carnivorans: a
three-dimensional analysis of forelimb shape. Journal of Evolutionary Biology 26:
1521-1535. doi: 10.1111/jeb.12161
https://doi.org/10.1111/jeb.12161...
b). This was the case of P.
cancrivorus, which exhibited a high MW/FD ratio. In felids, essentially
carnivores, rotation movements are even more necessary to capture, overwhelm and
manipulate prey extensively (Hudson et al.
2011Hudson PE, Corr SA, Payne-Davis RC, Clancy SN, Lane E, Wilson AM (2011)
Functional anatomy of the cheetah (Acinonyx jubatus) forelimb. Journal of Anatomy
218(4): 375-385. doi: 10.1111/j.1469-7580.2011.01344.x
https://doi.org/10.1111/j.1469-7580.2011...
).
These functional correlations are coherent with those previously proposed for domestic
carnivorans. According to Saint Clair (1986Saint Clair LE (1986) Músculos do carnívoro, p. 1416-1444. In: Getty R
(Ed.) Anatomia dos Animais Domésticos. Rio de Janeiro, Guanabara Koogan,
2048p.), the
distal muscles of the thoracic limbs are more developed in cats than in dogs to assure
the greater rotation of the distal portion of the limb. The brachioradialis muscle
presents a correlation not only with the forelimb usage in carnivorans. Andersson (2004Andersson K (2004b) Elbow-joint morphology as a guide to forearm
function and foraging behaviour in mammalian carnivores. Zoological Journal of the
Linnean Society 142: 91-104. doi: 10.1111/j.1096-3642.2004.00129.x
https://doi.org/10.1111/j.1096-3642.2004...
b) stated that manipulation and
locomotion are conflicting functions, since elbow-joint morphology supports a division
between grapplers (i.e., ambushers) and nongrapplers (i.e., pursuers). Joints of the
former are relatively wide, while in the latter they are relatively narrow and box-like
with pronounced stabilizing features. Concerning forepaw dexterity, Iwaniuk et al. (2001Iwaniuk AN, Pellis SM, Whishaw IQ (2001) Are long digits correlated with
high forepaw dexterity? A comparative test in terrestrial carnivores (Carnivora).
Canadian Journal of Zoology 79(5): 900-906. doi: 10.1139/z01-058
https://doi.org/10.1139/z01-058...
) considered that manus and
carpal shape and myology may play a more critical role than manus proportions. According
to them, behavioral observations also suggest that manus proportions correlated more
closely with locomotion than non-locomotory forepaw usage.
The ML/FL ratio represents the proportion of muscle length in relation to the forearm length. This ratio was significantly higher in mustelids. Longer muscles, especially with parallel fibers (which is the case of the brachioradialis), have more sarcomeres in series, which means added speed during contraction (Kardong 2011Kardong KV (2011) Vertebrados: anatomia comparada, função e evolução. São Paulo, Roca, 913p.). Therefore, in the mustelids one can assume that the brachioradialis muscle has a higher shortening speed than in the other families, an aspect that may be functionally important during swimming. This relatively longer length is a reflection of a more proximal level of origin in the humerus than in the other families, in other words, on the caudal surface of the humeral neck instead of the lateral supracondylar crest. Strengthening this hypothesis, in procyonids the ratio was significantly lower, because these animals have the slowest movements among the analyzed families. This shorter length was determined by the fact that its muscular part extends only until the forearm's middle third. This trait was also verified in two specimens of P. lotor (S.A. Feeney, unpubl. data).
From a mechanical point of view, it can be supposed that the arrangement of the mustelid
brachioradialis muscle raises the power leverage, creating a low power ratio, which
increases the strength and becomes important in fossorial habits. In felids, in
contrast, the origin at a more distal level makes the power leverage lower, creating a
higher power ratio, which increases speed, an important aspect for chasing prey. This
confirms the findings of Hudson et al. (2011Hudson PE, Corr SA, Payne-Davis RC, Clancy SN, Lane E, Wilson AM (2011)
Functional anatomy of the cheetah (Acinonyx jubatus) forelimb. Journal of Anatomy
218(4): 375-385. doi: 10.1111/j.1469-7580.2011.01344.x
https://doi.org/10.1111/j.1469-7580.2011...
) for
A. jubatus, that the internal architecture with long fibers of the
brachioradialis muscle is an adaptation that allows the muscle to contract at high
speeds and extensively rotate the radio-ulnar joint.
In the mustelids of the subfamily Lutrinae A. cinerea, L. lutra
and E. lutris (Macalister
1873Macalister A (1873b) On the anatomy of Aonyx. Proceedings of the Royal
Irish Academy Series 2: 539-547.b, Windle & Parsons 1897Windle BCA, Parsons FG (1897) On the myology of the terrestrial
Carnivora. Part I: muscles of the head, neck, and fore-limb. Proceedings of
Zoological Society of London 65: 370-409., Howard 1973Howard LD (1973) Muscular anatomy of the fore-limb of the sea otter
(Enhydra lutris). Proceedings of the California Academia of Science 39(4):
411-500.), the brachioradialis muscle has its
origin proximal to the humeral diaphysis, which also happened with L.
longicaudis, a member of the same subfamily analyzed in this study. The
origin at a proximal level could be a synapomorphy of Lutrinae. In the Lontra
canadensis (Schreber, 1777), a representative species of the most basal
lineage of otters, Fisher (1942Fisher EM (1942) The Osteology and Myology of the California River
Otter. Stanford, Stanford University Press, 66p.) described the
brachioradialis muscle as originating from the proximal region of the humerus and
running to the lateral supracondylar crest. From an evolutionary perspective, this
condition may reflect an intermediate position between the mustelids of the ferret type
(Mustelinae) and lineages like otters (Lutrinae). Based on Bayesian inference methods,
Koepfli et al. (2008Koepfli KP, Deere KA, Slater GJ, Begg C, Begg K, Grassman L, Lucherini
M, Veron G, Wayne RK (2008) Multigene phylogeny of the Mustelidae: resolving
relationships, tempo and biogeographic history of a mammalian adaptive radiation. BMC
Biology 6(10): 122. doi: 10.1186/1741-7007-6-10
https://doi.org/10.1186/1741-7007-6-10...
) stated these two
subfamilies diverged in the late Miocene (10 MYA) during the first burst of
diversification among Mustelidae.
Among the mustelids of Mustelinae, in the five specimens of ferrets (G.
cuja) dissected in this study, the muscle extended from the proximal part of
the humerus to the lateral supracondylar ridge. However, in six ferrets of the same
species dissected by Ercoli et al. (2014Ercoli MD, Álvarez A, Stefanini MI, Busker F, Morales MM (2014) Muscular
Anatomy of the Forelimbs of the Lesser Grison (Galictis cuja), and a Functional and
Phylogenetic Overview of Mustelidae and Other Caniformia. Journal of Mammalian
Evolution 22(1): 57-91. doi: 10.1007/s10914-014-9257-6
https://doi.org/10.1007/s10914-014-9257-...
), the
muscle had proximal origin in one-half (similar to the findings of this study) and in
the other half the origin was restricted to the supracondylar crest. In four individuals
of the species M. pennanti, the origin of the muscle occurred just
proximally to the lateral humeral supracondylar ridge (S.A. Feeney, unpubl. data).
In fact, a great number of morphologic characteristics shared between the subfamilies
Lutrinae and Mustelinae can be understood as favorable for both aquatic habits and
locomotion in tunnels (Ercoli et al. 2014Ercoli MD, Álvarez A, Stefanini MI, Busker F, Morales MM (2014) Muscular
Anatomy of the Forelimbs of the Lesser Grison (Galictis cuja), and a Functional and
Phylogenetic Overview of Mustelidae and Other Caniformia. Journal of Mammalian
Evolution 22(1): 57-91. doi: 10.1007/s10914-014-9257-6
https://doi.org/10.1007/s10914-014-9257-...
). This
peculiar arrangement, elongated and wide, of the brachioradialis muscle in this family
reinforces this observation. Furthermore, a more proximal origin means the levers are
more equilibrated. Thus it can be assumed that the muscle also acts as an important
flexor of the humerus radio-ulnar joint.
Schwarze (1984Schwarze E (1984) Compendio de Anatomia Veterinaria. Zaragoza, Editora Acribia, 318p.) and Dyce et al. (2010Dyce KM, Sack WO, Wensing CJG (2010) Tratado de Anatomia Veterinária. São Paulo, Elsevier, 856p.) reported that the muscle is small and almost never identified in dogs, especially in small ones. In L. gymnocercus, a canid slightly smaller than C. thous, the averages of the MW/FD and ML/FL ratios were also lower (significantly at 95% probability). However, the body size should not be a condition to predict the occurrence or functional relevance of the brachioradialis muscle in carnivorans, since small specimens such as G. cuja and felines exhibited a bilaterally well developed muscle.
In species with enough samples for comparison of the ratios between genders (C. thous, G. cuja and P. cancrivorus), only ML/FL ratio was significantly lower (p = 0.024) in female (0.81) than male (0.92) P. cancrivorus. Though it would be desirable to study a larger number of specimens, this may reflect the need for more developed skills for pray chasing in males.
Finally, the study allowed establishing that the brachiora dialis muscle occurs in individuals of the eight carnivorous species analyzed. This muscle originated on the lateral supracondylar crest of the humerus and was inserted in the medial surface of the radial distal extremity in canids, felids and procyonids, while its origin was on the caudal humeral neck in mustelids. Signs of sexual dimorphism were only detected in the relative length of the muscle in male P. cancrivorus. Overall, the mustelids, procyonids and felids have a proportionally more developed muscle than canids. As expected, these findings are consistent with the complexity and diversity of movements executed by the forelimbs of these species in the wild.
ACKNOWLEGMENTS
We acknowledge grants received from the Scholarships for Academic Development Program (PBDA) of Universidade Federal do Pampa and from the Young Talents for Science Program awarded to the second and third authors, respectively.
- Andersson K (2004a) Predicting carnivoran body mass from a weight-bearing joint. Journal of Zoology 262(2): 161-172. doi: 10.1017/S0952836903004564
» https://doi.org/10.1017/S0952836903004564 - Andersson K (2004b) Elbow-joint morphology as a guide to forearm function and foraging behaviour in mammalian carnivores. Zoological Journal of the Linnean Society 142: 91-104. doi: 10.1111/j.1096-3642.2004.00129.x
» https://doi.org/10.1111/j.1096-3642.2004.00129.x - Andersson K (2005) Were there pack-hunting canids in the Tertiary, and how can we know? Paleobiology 31(1): 56-72. doi: 10.1666/0094-8373(2005)031<0056:WTPCIT>2.0.CO;2
» https://doi.org/10.1666/0094-8373(2005)031<0056:WTPCIT>2.0.CO;2 - Andersson K, Werdelin L (2003) The evolution of cursorial carnivores in the Tertiary: implications of elbow joint morphology. Proceeding of the Biological Society 270(Suppl.): S163-S165. doi: 10.1098/rsbl.2003.0070
» https://doi.org/10.1098/rsbl.2003.0070 - Arlamowska-Palider A (1970) Morphological Studies on the Main Branches of the Radial Nerve in Mammals. Acta Theriologica 15(2): 185-197.
- Bohensky F (2002) Photo manual and dissection Guide of the Cat. New York, Square One Publishers, 167p.
- Budras KD, McCarthy PH, Fricke W, Richter R, Horowitz A, Berg R (2012) Anatomia do Cão, Texto e Atlas. Barueri, Manole, 219p.
- Concha I, Adaro L, Borroni C, Altamirano C (2004) Consideraciones anatómicas sobre la musculatura intrínseca del miembro torácico del puma (Puma concolor). International Journal of Morphology 22(2): 121-125. doi: 10.4067/S0717-95022004000200004
» https://doi.org/10.4067/S0717-95022004000200004 - Davis DD (1964) The giant panda: a morphological study of evolutionary mechanisms. Fieldiana: Zoology Memors 3: 1-339.
- Dyce KM, Sack WO, Wensing CJG (2010) Tratado de Anatomia Veterinária. São Paulo, Elsevier, 856p.
- Ercoli MD, Álvarez A, Stefanini MI, Busker F, Morales MM (2014) Muscular Anatomy of the Forelimbs of the Lesser Grison (Galictis cuja), and a Functional and Phylogenetic Overview of Mustelidae and Other Caniformia. Journal of Mammalian Evolution 22(1): 57-91. doi: 10.1007/s10914-014-9257-6
» https://doi.org/10.1007/s10914-014-9257-6 - Ewer RF (1973) The Carnivores. New York, Cornell University Press, 494p.
- Fabre AC, Cornette R, Peigné S, Goswami A (2013a) Influence of body mass on the shape of forelimb in musteloid carnivorans. Biological Journal of the Linnean Society 110: 91-103. doi: 10.1111/bij.12103
» https://doi.org/10.1111/bij.12103 - Fabre AC, Cornette R, Slater G, Argot C, Peigné S, Goswami A, Pouydebat E (2013b) Getting a grip on the evolution of grasping in musteloid carnivorans: a three-dimensional analysis of forelimb shape. Journal of Evolutionary Biology 26: 1521-1535. doi: 10.1111/jeb.12161
» https://doi.org/10.1111/jeb.12161 - Fabre AC, Goswami A, Peigné S, Cornette R (2014) Morphological integration in the forelimb of musteloid carnivorans. Journal of Anatomy 225: 19-30. doi: 10.1111/joa.12194
» https://doi.org/10.1111/joa.12194 - Fisher EM (1942) The Osteology and Myology of the California River Otter. Stanford, Stanford University Press, 66p.
- Fisher RE, Adrian B, Barton M, Holmgren J, Tang SY (2009) The phylogeny of the red panda (Ailurus fulgens): evidence from the forelimb. Journal of Anatomy 215(6): 611-635. doi: 10.1111/j.1469-7580.2009.01156.x
» https://doi.org/10.1111/j.1469-7580.2009.01156.x - Hall ER (1926). The muscular anatomy of three mustelid mammals, Mephitis, Spilogale, and Martes. University of California Publications in Zoology 30(2): 7-39.
- Hall ER (1927) The muscular anatomy of the American badger (Taxidea taxus). University of California Publications in Zoology 30(8): 205-219.
- Hermanson JW (2013) The Muscular System, p. 185-280. In: Evans HE, DeLahunta A (Eds) Miller's Anatomy of the Dog. Missouri, Elsevier, 872p.
- Howard LD (1973) Muscular anatomy of the fore-limb of the sea otter (Enhydra lutris). Proceedings of the California Academia of Science 39(4): 411-500.
- Hudson PE, Corr SA, Payne-Davis RC, Clancy SN, Lane E, Wilson AM (2011) Functional anatomy of the cheetah (Acinonyx jubatus) forelimb. Journal of Anatomy 218(4): 375-385. doi: 10.1111/j.1469-7580.2011.01344.x
» https://doi.org/10.1111/j.1469-7580.2011.01344.x - Hunter L (2011) Carnivores of the world. Princeton, Princeton University Press, 240p.
- Iwaniuk AN, Pellis SM, Whishaw IQ (1999) The relationship between forelimb morphology and behaviour in North American carnivores (Carnivora). Canadian Journal of Zoology 77(7): 1064-1074. doi: 10.1139/z99-082
» https://doi.org/10.1139/z99-082 - Iwaniuk AN, Pellis SM, Whishaw IQ (2001) Are long digits correlated with high forepaw dexterity? A comparative test in terrestrial carnivores (Carnivora). Canadian Journal of Zoology 79(5): 900-906. doi: 10.1139/z01-058
» https://doi.org/10.1139/z01-058 - Julik E, Zack S, Adrian B, Maredia S, Parsa A, Poole M, Starbuck A, Fisher RE (2012) Functional Anatomy of the Forelimb Muscles of the Ocelot (Leopardus pardalis). Journal of Mammalian Evolution 19(4): 277-304. doi: 10.1007/s10914-012-9191-4
» https://doi.org/10.1007/s10914-012-9191-4 - Kardong KV (2011) Vertebrados: anatomia comparada, função e evolução. São Paulo, Roca, 913p.
- Koepfli KP, Deere KA, Slater GJ, Begg C, Begg K, Grassman L, Lucherini M, Veron G, Wayne RK (2008) Multigene phylogeny of the Mustelidae: resolving relationships, tempo and biogeographic history of a mammalian adaptive radiation. BMC Biology 6(10): 122. doi: 10.1186/1741-7007-6-10
» https://doi.org/10.1186/1741-7007-6-10 - Leach D (1976) The forelimb musculature of marten (Martes americana Turton) and fisher (Martes pennanti Erxleben). Canadian Journal of Zoology 55(1): 31-41. doi: 10.1139/z77-00
» https://doi.org/10.1139/z77-00 - Liebich HG, Maierl J, König HE (2011) Membros Torácicos ou Anteriores (Membra Thoracica), p. 165-234. In:, König HE Liebich HG (Eds) Anatomia dos Animais Domésticos: Texto e Atlas Colorido. Porto Alegre, Artmed, 787p.
- Macalister A (1873a) The muscular anatomy of the civet and tayra. Proceedings of the Royal Irish Academy Academy Series 2: 506-513.
- Macalister A (1873b) On the anatomy of Aonyx. Proceedings of the Royal Irish Academy Series 2: 539-547.
- Mackintosh BA (1875) Notes on the myology of the coati mondth (Nasua narica and N. fusca) and common marten (Martes foina). Proceedings of the Royal lrish Academy Series 2: 48-55.
- Martín-Serra A, Figueirido B, Palmqvist P (2014) A Three-Dimen sional Analysis of Morphological Evolution and Locomotor Performance of the Carnivoran Forelimb. PLoS ONE 9: e85574. doi: 10.1371/journal.pone.0085574
» https://doi.org/10.1371/journal.pone.0085574 - McClearn D (1992) Locomotion, Posture, and feeding behavior of kinkajous, coatis, and raccoons. Journal of Mammalogy 73(2): 245-261.
- Meachen-Samuels JA, Van-Valkenburgh B (2009) Forelimb indicators of prey-size preference in the Felidae. Journal of Morphology 270(6): 729-744. doi: 10.1002/jmor.10712
» https://doi.org/10.1002/jmor.10712 - Meloro C, Elton C, Louys J, Bishop LC, Ditchfield P (2013) Cats in the forest: predicting habitat adaptations from humerus morphometry in extant and fossil Felidae (Carnivora). Paleobiology 39(3): 323-244. doi: 10.1666/12001
» https://doi.org/10.1666/12001 - Mills P (2003) Comparative Animal Anatomy. Queensland, Gatton Desktop Publishing, 316p.
- Moore AL, Budny JE, Russel AP, Butcher MT (2013) Architectural specialization of the intrinsic thoracic limb musculature of the American badger (Taxidea taxus). Journal of Morphology 274(1): 35-48. doi: 10.1002/jmor.20074
» https://doi.org/10.1002/jmor.20074 - Nickel R, Schummer A, Seiferle E, Frewein J, Wilkens H, Wille K (1986) The Locomotor System of Domestic Mammals. Berlin, Verlag Paul Parey, 515p.
- Nowak RM (2005) Walker's Carnivores of the World. Baltimore, The Johns Hopkins University Press, 328p.
- Paranaiba JF, Helrigle C, Araújo EG, Pereira KF (2012) Aspectos morfológicos da mão e pé de Procyon cancrivorus. Natureza On line 10(4): 165-169. Available online at: http://www.naturezaonline.com.br/natureza/conteudo/pdf/03_ParanaibaJFetal_165_169.pdf. [Accessed: 31 July 2014]
» http://www.naturezaonline.com.br/natureza/conteudo/pdf/03_ParanaibaJFetal_165_169.pdf - Pessutti C, Santiago MEB, Oliveira LTF (2001) Order Carnivora, Family Canidae (Dogs, foxes and maned wolves), p. 279-290. In: Fowler ME, Cubas ZS (Eds) Biology, Medicine and Surgery of South American Wild Animals. Ames, Iowa State University Press, 550p.
- Pestana FM, Silva BX, Chagas MA, Babinski MA, Abidu-Figueiredo M (2009) Distribuição antimérica do músculo braquiorradial em cães sem raça definida. Revista de Ciências da Vida 29(1): 55-59.
- Polly PD (2007) Limbs in mammalian evolution, p. 245-268. In: Hall BK (Ed.) Fins into Limbs: Evolution, Development, and Transformation. Chicago, University of Chicago Press, 344p.
- Reis NR, Peracchi AL, Fregonezi MN, Rossaneis BK (2010) Mamíferos do Brasil: guia de identificação. Rio de Janeiro, Technical Books, 560p.
- Saint Clair LE (1986) Músculos do carnívoro, p. 1416-1444. In: Getty R (Ed.) Anatomia dos Animais Domésticos. Rio de Janeiro, Guanabara Koogan, 2048p.
- Saladin KS (2010) Anatomy and Physiology: The Unity of Form and Function. New York, McGraw Hill, 1248p.
- Samuels JX, Meachen JA, Sakai SA (2013) Postcranial morphology and the locomotor habits of living and extinct carnivorans. Journal of Morphology 274(2): 121-146. doi: 10.1002/jmor.20077
» https://doi.org/10.1002/jmor.20077 - Sánchez HL, Silva LB, Rafasquino ME, Mateo AG, Zuccolilli GO, Portiansky EL, Alonso CR (2013) Anatomical study of the forearm and hand nerves of the domestic cat (Felis catus), puma (Puma concolor) and jaguar (Panthera onca). Anatomia Histologia Embryologia 42(2): 99-104. doi: 10.1111/j.1439-0264.2012.01170.x
» https://doi.org/10.1111/j.1439-0264.2012.01170.x - Santos Junior I, Rodrigues CA, Campos A, Santos D (2002) Presença do músculo braquiorradial em cães. Bioscience Journal 18(1): 79-83.
- Sato JJ, Wolsan M, Minami S, Hosoda T, Sinaga MH, Hiyama K, Yamaguchi Y, Suzuki H (2009) Deciphering and dating the red panda's ancestry and early adaptive radiation of Musteloidea. Molecular Phylogenetics and Evolution 53(3): 907-922. doi: 10.1016/j.ympev.2009.08.019
» https://doi.org/10.1016/j.ympev.2009.08.019 - Sato JJ, Wolsan M, Prevosti FJF, D'Elía G, Begg C, Begg K, Hosoda T, Campbell KL, Suzuki H (2012) Evolutionary and biogeographic history of Weasellike carnivorans (Musteloidea). Molecular Phylogenetics and Evolution 63(3): 745-757. doi: 10.1016/j.ympev.2012.02.025
» https://doi.org/10.1016/j.ympev.2012.02.025 - Schwarze E (1984) Compendio de Anatomia Veterinaria. Zaragoza, Editora Acribia, 318p.
- Slater GJ, Harmon LJ, Alfaro ME (2012) Integrating fossils with molecular phylogenies improves inference of trait evolution. Evolution 66(12): 3931-3944. doi: 10.1111/j.1558-5646.2012.01723.x
» https://doi.org/10.1111/j.1558-5646.2012.01723.x - Sebastiani AM, Fishbeck DW (2005) Mammalian anatomy: the cat. Colorado, Morton Publishing Company, 184p.
- Taylor ME (1989) Locomotor adaptations by carnivores, p. 382-409. In: Gittleman JL (Ed.) Carnivore Behavior, Ecology, and Evolution. Cornell, Comstosck Publiching Associates, 620p.
- Vaz MGR, Lima AR, Souza ACB, Pereira LC, Branco E (2011) Estudo morfológico dos músculos do antebraço de cachorro-do-mato-de-orelhas-curtas (Atelocynus microtis) e cachorro-do-mato (Cerdocyon thous). Biotemas 24(4): 121-127. doi: 10.5007/2175-7925.2011v24n4p121
» https://doi.org/10.5007/2175-7925.2011v24n4p121 - Wakuri H, Kano Y (1966) Anatomical studies on the brachioradial muscle in dogs. Acta Anatomica Nipponica 41: 222-231.
- Williams SB, Wilson AM, Rhodes L, Andrews J, Payne RC (2008) Functional anatomy and muscle moment arms of the thoracic limb of an elite sprinting athlete: the racing greyhound (Canis familiaris). Journal of Anatomy 213(4): 361-372. doi: 10.1111/j.1469-7580.2008.00962.x
» https://doi.org/10.1111/j.1469-7580.2008.00962.x - Wilson DE, Mittermeier RA (2009) Handbook of the Mammals of the World. Barcelona, Lynx Edicions, vol. 1, 727p.
- Windle BCA, Parsons FG (1897) On the myology of the terrestrial Carnivora. Part I: muscles of the head, neck, and fore-limb. Proceedings of Zoological Society of London 65: 370-409.
- Witmer LM (1995) The extant phylogenetic bracket and the importance of reconstructing soft tissues in fossils, p. 19-33. In: Thomason JJ (Ed.) Functional morphology in vertebrate paleontology. Cambridge, Cambridge University Press, 277p.
APPENDIX
Appendix. The specimens analyzed are deposited in the Laboratory of Animal Anatomy of Universidade Federal do Pampa, Uruguaiana, RS, Brazil, with the following numbers: Cerdocyon thous: 5139, 5144, 5267, 5273, 5275, 5717, 8503, 8504, 8505 Lycalopex gymnocercus: 5134, 5261, 5269, 5274, 5603 Procyon cancrivorus: 5136, 5268, 8517 Galictis cuja: 5142, 5146, 5599, 5714, 5720 Leopardus geoffroyi: 5138, 5145, 5147, 5150 Nasua nasua: 8527 Lontra longicaudis: 8507 Leopardus colocolo: 5137
Publication Dates
-
Publication in this collection
Jan-Feb 2015
History
-
Received
11 Aug 2014 -
Reviewed
25 Dec 2014 -
Accepted
05 Feb 2015