Abstract
It is well known that the culture conditions of microorganisms may affect their surface properties, zeta potential and hydrophobicity via the modification of the cell wall functional groups or metabolic products. The R. opacus bacteria strain was separately adapted to the presence of apatite and quartz, after which a cellular adaptation procedure was developed by repeated sub-culturing with a successive increase in the mineral content. Zeta potential, surface tension, FTIR and microflotation studies were used to evaluate the behavior of the cells that were developed under defined culture conditions. The cellular adaptation induced a modification of the bacterial surface charge. The FTIR results showed a modification of its functional groups. The surface tension results suggested that longer growing time promoted a higher production of metabolites. The use of mineral-adapted cells promoted an improvement in the flotability of both minerals, but it was more significant for apatite flotation. Additionally, the mineral flotability remained unchanged when the cells developed under a longer culture time. Nevertheless, there was a reduction in the surface tension.
Keywords:
Rhodococcus opacus; bioflotation; cellular adaptation; apatite; quartz
1. Introduction
Studies on the application of biotechnology in mineral processing have intensified in recent years. One of the principal motivations is to reduce the environmental impacts caused by the use of conventional flotation reagents (Rao and Subramanian, 2007RAO, K.H., SUBRAMANIAN, S. Bioflotation and Bioflocculation of relevance to minerals bioprocessing. IN: Donati, R. E., SAND, W. Microbial processing of metal sulfides. 2007. p. 267-286.). The bioflotation technique is in the fundamental stage, and considerable research is still necessary before its industrial application. A fundamental bioflotation study should determine the response of each mineral that is present in an ore. Thus, considering that phosphate ores are associated with several gangue minerals, especially quartz, carbonates and silicates, then a detailed bioflotation study of each component is necessary to establish the bioflotation of phosphate ores. One bacteria, quite recently used in mineral bioflotation, is Rhodococcus opacus (Botero et al., 2007BOTERO, A.E.C., TOREM, M.L., MESQUITA, L.M.S. Fundamental studies of Rhodococcus opacus as a biocollector of calcite and magnesite. Minerals Engineering, v. 20, n. 10, p. 1026-1032, 2007.; Mesquita et al., 2003MESQUITA, L.M.S., LINS, F.A.F., TOREM, M.L. Interaction of a hydrophobic bacterium strain in a hematite-quartz flotation system. International Journal of Mineral Processing, v. 71, p. 31-44, 2003.; Botero et al., 2008BOTERO, A.E.C., TOREM, M.L., MESQUITA, L.M.S. Surface chemistry fundamentals of biosorption of Rhodococcus opacus and its effect in calcite and magnesite flotation. Minerals Engineering, v. 21, n. 1, p. 83-92, 2008.; Merma et al., 2013MERMA, A.G., TOREM, M.L., MORAN, J.V., MONTE, M.B.M. On the fundamental aspects of apatite and quartz flotation using a gran positive strain as a bioreagent. Minerals Engineering, v., 48, p. 61-67, 2013.). It is a Gram positive bacterium with different types of components on their cell-wall, which are primarily polysaccharides, carboxylic acids, lipid groups and mycolic acids, that result in amphoteric behavior (hydrophobic and/or hydrophilic properties) of the bacteria (Mesquita et al., 2003MESQUITA, L.M.S., LINS, F.A.F., TOREM, M.L. Interaction of a hydrophobic bacterium strain in a hematite-quartz flotation system. International Journal of Mineral Processing, v. 71, p. 31-44, 2003.; Natarajan, 2006NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006.; Vilinska and Rao., 2008VILINSKA, A., RAO, K.H. Leptosririllum ferrooxidans-sulfide mineral interactions with reference to bioflotation and bioflocculation. Trans. Nonferrous Met. Soc. China, v. 18, p. 1403-409, 2008.). According to Natarajan (2006)NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006., the presence of these components produces a selective behavior in the bacteria. Moreover, their production can be modified, rendering some bacteria more hydrophobic or even more selective. This modification can be performed through "cellular adaptation" or via genetic modifications. Thus, cellular adaptation in mineral bioprocessing has been the focus of some studies (Sharma et al., 2001SHARMA, P.K., RAO, K.H., FORSSBERG, K.S.E., NATARAJAN, K.A., Surface chemical characterisation of Paenibacillus polymyxa before and after adaptation to sulfide minerals. International Journal of Minerals Processing, v. 62, p. 3-25, 2001.; Subramanian et al., 2003SUBRAMANIAN, S., SANTHIYA, D., NATARAJAN, K.A. Surface modification studies on sulphide minerals using bioreagents. International Journal of Mineral Processing, v. 72, p. 175-188, 2003.; Sarvamangala and Natarajan, 2011SARVAMANGALA, H., NATARAJAN, K.A. Microbially induced flotation of alumina, silica/calcite from haematite. International Journal of Minerals processing, v. 99, p. 70-77, 2011.; Sarvamangala et al., 2012SARVAMANGALA, H., NATARAJAN, K.A., GIRISHA, S.T. Biobeneficiation of iron ores. International Journal of Mining Engineering and Mineral Processing, v. 1, v. 2, p. 21-30, 2012.; Vasanthakumar et al., 2013VASANTHAKUMAR, B., RAVISHANKAR, H., SUBRAMANIAN, S. Microbially induced selective flotation of sphalerite from galena using mineral-adapted strains of Bacillus megaterium. Colloids and Surfaces B: Biointerfaces, v. 112, p. 279-286, 2013.; Sarvamangala et al., 2013SARVAMANGALA, H., NATARAJAN, K.A., GIRISHA, S.T. Microbially-induced pyrite removal from galena using Bacillus subtilis. International Journal of Minerals Processing, v. 120, p. 15-21, 2013.). However, to our knowledge, there is no study on the cellular adaptation of R. opacus bacteria to a mineral substrate.
In mineral bioprocessing, the "cellular adaptation" of a microorganism refers to the growing of its cells in the presence of a mineral substrate. Some authors (Subramanian et al., 2003SUBRAMANIAN, S., SANTHIYA, D., NATARAJAN, K.A. Surface modification studies on sulphide minerals using bioreagents. International Journal of Mineral Processing, v. 72, p. 175-188, 2003.; Vasanthakumar et al., 2013VASANTHAKUMAR, B., RAVISHANKAR, H., SUBRAMANIAN, S. Microbially induced selective flotation of sphalerite from galena using mineral-adapted strains of Bacillus megaterium. Colloids and Surfaces B: Biointerfaces, v. 112, p. 279-286, 2013.; Sarvamangala et al., 2013SARVAMANGALA, H., NATARAJAN, K.A., GIRISHA, S.T. Microbially-induced pyrite removal from galena using Bacillus subtilis. International Journal of Minerals Processing, v. 120, p. 15-21, 2013.) have suggested that cellular adaptation can produce changes in bacterial morphology, in addition to quantitative and qualitative modifications during the secretion of biopolymers, which primarily constitute proteins and polysaccharides, during their growth. Therefore, the objective of this work is to study the effect of the different culture conditions of the Rhodococcus opacus in the presence of a mineral substrate on the bioflotation of apatite and quartz by analyzing their electrophoretic mobility, surface tension, FTIR signatures and flotation behaviors.
2. Material and methods
2.1 Sample preparation
This study used a pure quartz sample and a fluorapatite sample (42.33% P2O5 and 54.39% CaO) that were provided by a local supplier (Belo Horizonte, Minas Gerais State) to be used in this study. The samples were crushed and screened to -3 mm. Then, the samples were dry-ground in a porcelain mortar and wet-screened. Afterwards, the desired size fractions (+20-38; +75-106) in µm were separated. Then, the quartz sample rested in a KOH (0.1 M) solution for 24 hours to remove the impurities present on the surface. The samples were washed several times with double-distilled water and milli-Q water until the pH suspension achieved the initial pH. Finally, the quartz and apatite samples dried at room temperature and were stored in a desiccator.
2.2 Microorganisms, media and growth
The Rhodococcus opacus strain (CBMAI 812) was supplied by The Chemical, Biological and Agricultural Pluridisciplinary Research Center (CPQBA) and was developed in a YMG solid medium containing the following: 10.0 g L-1 glucose, 5.0 g L-1 peptone, 3.0 g L-1 malt extract, 3.0 g L-1 yeast extract, and 12 g L-1 agar-agar. Stocks of the bacteria were prepared frequently using this medium in Petri plates and they were stored in a freezer at 12 ºC. Then, the cells were sub-cultured in an YMG liquid medium containing 10.0 g L-1 glucose, 5.0 g L-1 peptone, 3.0 g L-1 malt extract and 3.0 g L-1 yeast extract at pH 7.2. They were placed in a rotary shaker at 140 rpm and 28 ºC for 48 h. Afterwards, the bacterial cells were separated from the culture by centrifuging at 3500 rpm for 8 min, followed by re-suspending twice with deionized water. Then, the cells were re-suspended in a 10-3 M NaCl solution. Finally, the bacterial concentrate was inactivated in an autoclave for 20 minutes to avoid further development of the bacteria. The bacterial concentration of the suspension was measured using a spectrophotometer (UV-1800, Shimatzu UV-spectrophotometer) at a wavelength of 620 nm.
2.3 Cellular adaptation
The adaptation of R. opacus was performed via repeated sub-culturing, in which 10 mL of the strain was inoculated into 190 mL of YMG liquid medium. This was in the presence of incremental increases in the mineral content (quartz or apatite), which was prepared in a 250-mL Erlenmeyer flask and incubated at 28 ºC on a rotary shaker at 140 rpm. Initially, the sub-culturing started with only 1% pulp density; then, the pulp density was successfully increased during subsequent sub-culturing until achieving 5 wt. % of the mineral. The cellular adaptation was considered achieved when the growth curve of the adapted strain was identical to that of the control (grown curve of the unadapted bacteria). This adaptation procedure was performed in the presence of quartz and apatite separately. After this cellular adaptation, the adapted-cells were washed and concentrated as described in section 2.2.
2.4 Composition of bacterial cell wall
The bacterial cell wall components were extracted using the Cammarota method. The components associated with the cell wall were extracted with 0.01 M phosphate buffer solution at pH 7. A mixture of 50% bacterial suspension and 50% EDTA 2% was prepared. This solution was refrigerated for 4 h at 4ºC; then it was centrifuged and the supernatant was collected. This extraction method was applied in a previous work (Botero et al., 2008BOTERO, A.E.C., TOREM, M.L., MESQUITA, L.M.S. Surface chemistry fundamentals of biosorption of Rhodococcus opacus and its effect in calcite and magnesite flotation. Minerals Engineering, v. 21, n. 1, p. 83-92, 2008.); however, the complete method was described by Cammarota (1998)CAMMAROTA, M.C. Exopolysaccharides production and microbial adhesion. Rio de Janeiro: Departamento de Bioquímica, Univerdidade Federal do Rio de Janeiro, 1998. (Tese de Doutorado).. The cell wall-associated polysaccharides were measured using the Antrona method (Yemn and Willis, 1954YEMN, E.W. WILLIS, A.J. The estimation of carbohydrate in plant extracts by anthrone. The Biochemical Journal, v. 57, p. 508-514, 1954.), and the cell wall-associated proteins were measured using the Biureto method (Stickland, 1951STICKLAND, L. H. The determination of small quantities of bactéria by means of biuret reaction. J. Gen. Microbiol, v. 5, p. 698-703, 1951.).
2.5 Zeta potential studies
The Zeta potential measurements for the R. opacus cells were conducted in a micro-electrophoresis apparatus, "Zetasizer Nano" - Malvern. The concentration of the biomass suspension was 50 mg L-1, and a NaCl 10-3 M solution was used as an indifferent electrolyte. The pH was adjusted using the HCl and NaOH solutions. The evaluation of the zeta potential profiles for the R. opacus cells was conducted before and after cellular adaptation to the mineral substrate.
2.6 Fourier Transform Infrared Spectroscopy (FTIR)
Infrared absorption spectra were recorded on a FTIR Thermo-Nicolet 6700 spectrophotometer. The data acquisition was conducted in transmission mode. The spectra of the R. opacus bacteria before and after cellular adaptation were evaluated. The cell suspension samples were washed and lyophilized. Then, the lyophilized sample was properly mixed with spectroscopic grade KBr (1:100) and pressed into pellets to record the spectra, which was collected after 120 scans at 4 cm-1 in the frequency range of 4000 cm-1 - 400 cm-1.
2.7 Surface tension measurements
The surface tension measurements of the bacterial suspension were performed using the ring method in a Kruss K10 digital tensiometer with an accuracy of ± 0.1 mN m-1. The surface tension measurements were conducted during the bacterial development and at different times and as a function of the pH suspension. All of the measurements were repeated five times, and their average values were plotted.
2.8 Microflotation experiments
The flotability of the minerals was evaluated in a modified Hallimond tube (Merma, 2012MERMA, A.G. Fundamental aspects in the apatite-quartz bioflotation system using the R. opacus bacteria as bioreagent. Rio de Janeiro: Departamento de Engenharia Química e de Materiais, Pontifícia Universidade Católica do Rio de Janeiro, 2012. (Tese de Doutorado).). Approximately 1.0 g of mineral was added to a 0.16 L total volume suspension of known bacterial concentration. The mineral was conditioned with the bacterial suspension at a desired pH value inside the Hallimond tube under constant stirring for 5 minutes. Finally, the microflotation tests were conducted using air at a flow rate of 15 ml min-1 for 2 min. The settled and floated fractions were carefully separated, washed, dried and weighed. The flotability was then calculated as the ratio of floated and non-floated mineral amounts to the total weighed sample.
3. Results and discussion
The microorganism culture conditions can affect the morphology, structure and composition of the bacterial cell wall and of metabolic products and therefore affect their surface characteristic (Natarajan, 2006NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006.; Natarajan and Deo, 2001NATARAJAN, K.A., DEO, N. Role of bacterial interaction and bioreagents in iron ore flotation. International Journal of Minerals Processing, v. 62, p. 62: 143-147, 2001.; Amézcua-Vega et al., 2007AMÉZCUA-VEGA, C., POGGI-VARALDO, H.M., ESPARZA-GARCÍA, F., RÍOS-LEAL, E., AND RODRÍGUEZ-VÁZQUEZ, R. Effect of culture conditions on fatty acids composition of a biosurfactant produced by Candida ingens and changes of surface tension of culture media. Bioresource Technology, v. 98, n. 1, p. 237-240, 2007.) as electrophoretic behavior, hydrophobic properties and, consequently, their response to mineral flotation. The cell shape is generally characteristic of each bacterial strain, but this can vary depending on the growth conditions. In previous studies (Mesquita et al., 2003MESQUITA, L.M.S., LINS, F.A.F., TOREM, M.L. Interaction of a hydrophobic bacterium strain in a hematite-quartz flotation system. International Journal of Mineral Processing, v. 71, p. 31-44, 2003.; Merma et al., 2013MERMA, A.G., TOREM, M.L., MORAN, J.V., MONTE, M.B.M. On the fundamental aspects of apatite and quartz flotation using a gran positive strain as a bioreagent. Minerals Engineering, v., 48, p. 61-67, 2013.), it was observed that the R. opacus cells had a "coccus" shape at the lag phase and at the beginning of the exponential phase; however, in the stationary phase, the cells presented a bacillus-shape. Although, the modification of the morphology of the cells occurs along the culture, it was observed that their hydrophobicity did not present any significant change, preserving a contact angle value of approximately 75º. This result could be an indication that the culture time does not affect the composition of the bacterial cell wall, even when their morphology is affected.
3.1 Zeta potential studies
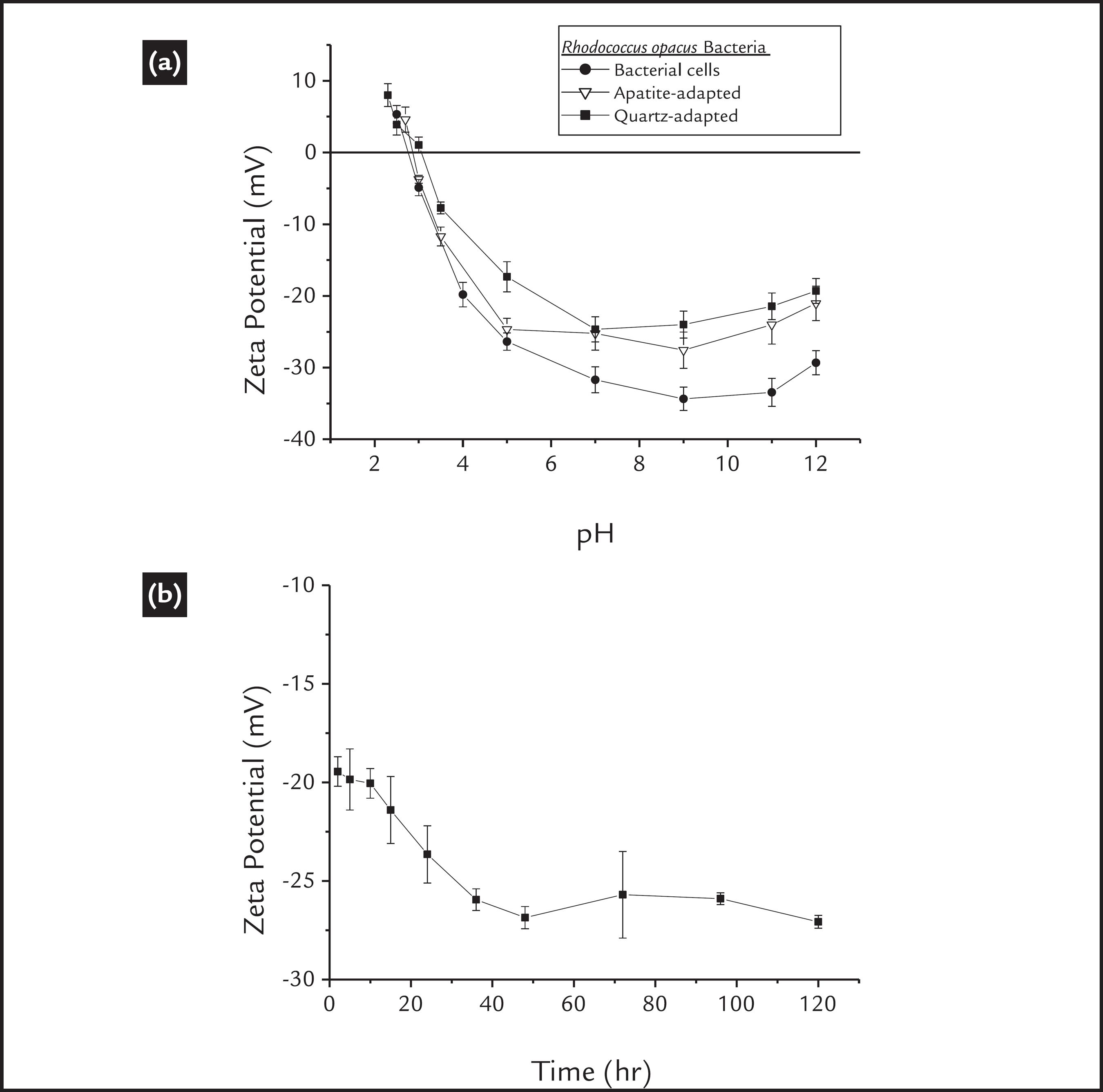
Any changes in the cell surface charge may be related to modifications in the bacterial cell wall functional groups. Fig. 1(a) shows the zeta potential of the microorganism at different pH values. Initially, we observed an isoelectric point (IEP) of approximately 2.8, as found in previous works (Botero et al., 2007BOTERO, A.E.C., TOREM, M.L., MESQUITA, L.M.S. Fundamental studies of Rhodococcus opacus as a biocollector of calcite and magnesite. Minerals Engineering, v. 20, n. 10, p. 1026-1032, 2007., 2008BOTERO, A.E.C., TOREM, M.L., MESQUITA, L.M.S. Surface chemistry fundamentals of biosorption of Rhodococcus opacus and its effect in calcite and magnesite flotation. Minerals Engineering, v. 21, n. 1, p. 83-92, 2008.; Mesquita et al., 2003MESQUITA, L.M.S., LINS, F.A.F., TOREM, M.L. Interaction of a hydrophobic bacterium strain in a hematite-quartz flotation system. International Journal of Mineral Processing, v. 71, p. 31-44, 2003.; Merma et al., 2013MERMA, A.G., TOREM, M.L., MORAN, J.V., MONTE, M.B.M. On the fundamental aspects of apatite and quartz flotation using a gran positive strain as a bioreagent. Minerals Engineering, v., 48, p. 61-67, 2013.). According to several authors (Rao and Subramanian, 2007RAO, K.H., SUBRAMANIAN, S. Bioflotation and Bioflocculation of relevance to minerals bioprocessing. IN: Donati, R. E., SAND, W. Microbial processing of metal sulfides. 2007. p. 267-286.; Natarajan, 2006NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006.; Vilinska and Rao., 2008VILINSKA, A., RAO, K.H. Leptosririllum ferrooxidans-sulfide mineral interactions with reference to bioflotation and bioflocculation. Trans. Nonferrous Met. Soc. China, v. 18, p. 1403-409, 2008.; Faharat et al., 2008FAHARAT, M., HIRAJIMA, T., SASAKI, K., AIBA, Y., DOI, K. Adsorption of SIP E. coli onto quartz and its applications in froth flotation. Minerals Engineering, v. 21, p. 389-395, 2008.; Van Der Wal et al., 1997VAN DER WAL, A., NORDE, A.A.J.B., ZEHNDER, B., LYKLEMA, J. A determination of the total charge in the cell walls of gram-positive bacteria. Colloids and Surfaces B: Biointerfaces, v. 9, p. 81-100, 1997.), this acidic value is related to the higher presence of anionic functional groups compared with cationic groups. We also observed a singular effect: above pH 9, the zeta potential values of the cells systematically shifts to more positive values (as the pH shifts to more basic values). This particularity is attributed to an increase in ionic strength due to the addition of NaOH, which was done to raise the pH above 9. That would cause a compression of the electrical double layer, and thus, a reduction of the absolute value of the zeta potential (Mozes et al., 1998MOZES, N., LÉONARD, A.J., ROUXHET, P.G. On the relations between the elemental surface compositions of yeasts and bacteria and their charge and hydrophobicity. Biochimica et Biophysica Acta, v. 945, p. 324-334, 1988).
Zeta potential of cells (a) before and (b) after adaptation as a function of the culture time.
As mentioned before, the microorganism culture conditions affect their surface properties, and according to Natarajan (2006)NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006. the cellular adaptation may affect the surface properties of the bacterial cells, due to a modification in the balance of anionic and cationic groups present in the cell wall components or in their metabolic products. In this work, no significant change in the bacterial IEP after cellular adaptation was observed (Fig.1a). However, a small change in the zeta potential profile of the cells was observed; both mineral-adapted cells presented less negative zeta potential values. These modifications may be attributed to a reduction of anionic groups or to an increment of cationic groups, which could be a consequence of a lower polysaccharides production or a higher proteins production, during cellular adaptation.
Additionally, the zeta potential profile of the bacteria as a function of culture time is shown in Fig. 1 (b). We observed that the net charge exhibited a small decrease from approximately -20 mV to -27 mV during the first 40 h. of culture, which corresponds to the beginning of the exponential phase of growth (See Fig. 2a), and then the bacterial net charge remained practically unchanged. These results support the idea that the culture time may affect the morphology of the cells, but may not affect the surface properties of the cells under the conditions used in this work (see section 2.2).
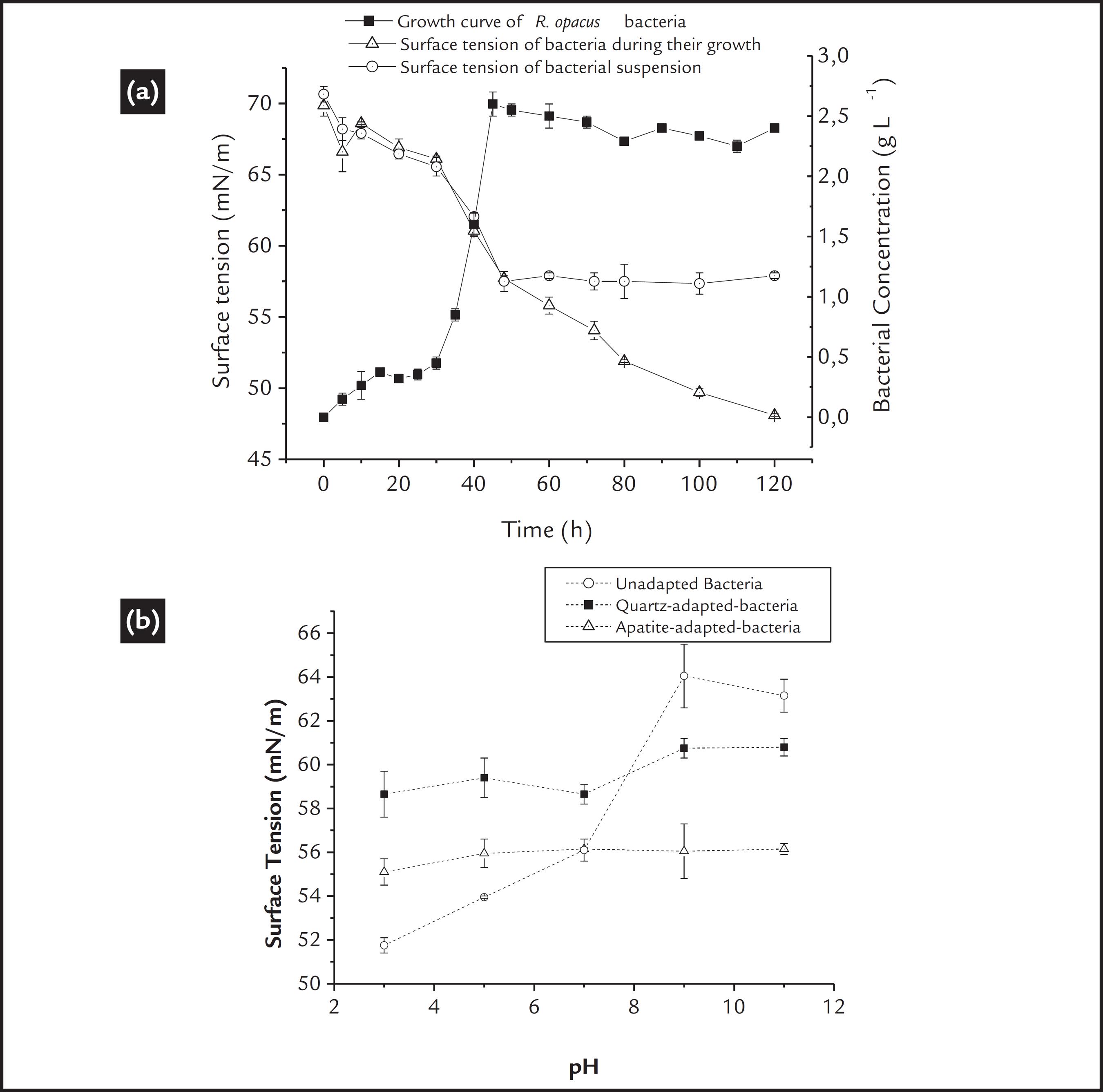
a) Growth curve and surface tension of the R. opacus bacteria as a function of time. b) Surface tension measurements of the R. opacus suspensions before and after adaptation to the mineral substrate.
3.2 Surface tension of R. opacus suspensions
Surface tension measurements that are associated with the bacterial growth curve can help elucidate any production of biosurfactants (Didyk and Sadowski, 2013DIDYK, A.M., SADOWSKI, Z. Flotation of serpentinite and quartz using biosurfactans. Physicochemical Problems of Mineral Processing, v. 48, n. 2, p. 607-618, 2013.) and/or metabolites or even any modification of the bacterial surface. Initially, a reduction in the surface tension is related to an increase in the bacterial concentration. When the stationary phase of growth is achieved, the surface tension values should remain constant, and any reduction in the tension values may be related to metabolic production, as described by Didyk and Sadowski (2013)DIDYK, A.M., SADOWSKI, Z. Flotation of serpentinite and quartz using biosurfactans. Physicochemical Problems of Mineral Processing, v. 48, n. 2, p. 607-618, 2013.. These results are observed in Fig 2a, in which the surface tension of the culture broth (cells in the culture medium) and free-cells suspensions (cells separate from the culture) were measured. The first could determine the metabolite production, and the second could elucidate any modification of the bacterial cell wall.
A continuous reduction in the surface tension values of the culture broth after the stationary phase is achieved, may be considered an indicator of the production of metabolites that are able to reduce the surface tension, such as biosurfactants (Didyk and Sadowski, 2013DIDYK, A.M., SADOWSKI, Z. Flotation of serpentinite and quartz using biosurfactans. Physicochemical Problems of Mineral Processing, v. 48, n. 2, p. 607-618, 2013.). It is also observed that the surface tension of the free-cells suspensions presented a constant value (approximately 57 mN/m) as a function of time, suggesting that a longer culture time did not cause any modification on the bacterial surface, and if metabolites were produced, they probably were expelled to the medium.
Furthermore, a comparative analysis of the surface tension of the free-cell suspensions before and after cellular adaptation could reveal if "cellular adaptation" promoted any modification on the bacterial cell wall. The surface tension profile of the bacterial cells as a function of pH is shown in Fig. 2b. The surface tension of the unadapted bacterial cells decreased significantly below pH 7. The lowest value occurred at approximately the IEP of the microorganism (around pH 3)., where, according to Lambert et al. (2003)LAMBERT, W.,D., DU, L., MA, Y., LOHA, V., BURAPATANA, V., PROKOP, A., TANNER, R., D., PAMMENT, N.B. The effect of pH on the foam fractionation of b-glucosidase and cellulase. Bioresource Technology, v. 87, p. 247-253, 2003., the best condition to form foam using bacteria is at its IEP value, which is also important in flotation. However, above pH 7, higher surface tension values were observed, which are not beneficial for flotation.
Thus, from the results, a similar behavior could be observed for both mineral-adapted cells; an increase in the surface tension values in the acidic region and a decrease in the alkaline region. The quartz-adapted cells presented surface tension values between 58 and 60 mN/m for the pH scale studied, whereas the apatite-adapted cells presented lower values at approximately 55 mN/m. The surface tension results demonstrated the ability of the bacteria to form foam, which confirm its biofrother function. The "cellular adaptation" did not affect its biofrother function; however, a higher biofrother ability is expected for apatite-adapted cells.
3.3 Fourier Transform Infrared Spectroscopy (FTIR)
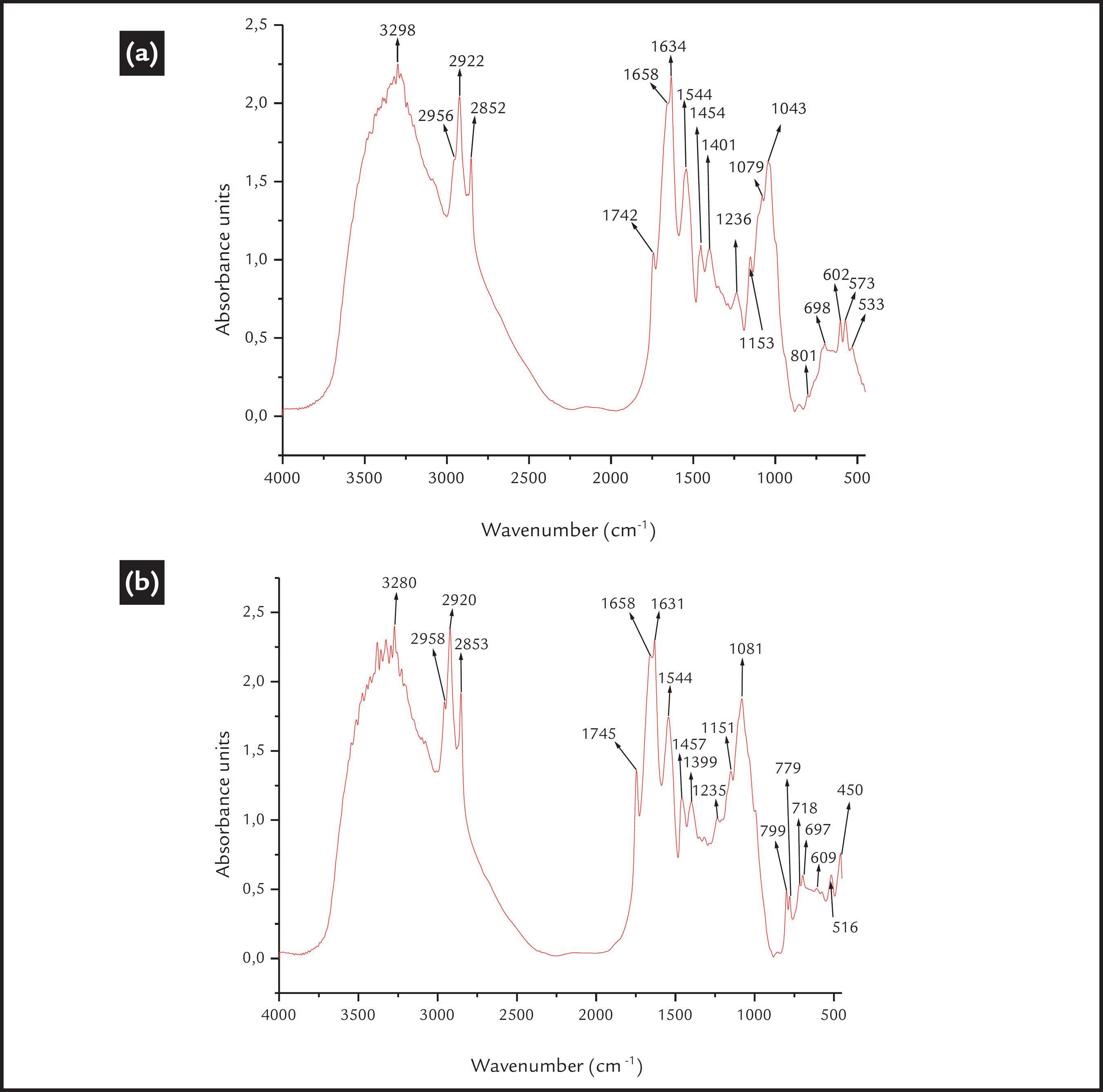
The FTIR spectra of the R. opacus cells exhibited many bands between 400 and 4000 cm-1. The principal bands and their respective functional groups are presented in Table 1. The intensities of the higher bands correspond to the principal components of the bacterial wall cell, such as the proteins, fatty acids and polysaccharides. Thus, the cell surface is partially hydrophobic and partially hydrophilic. This is a general representative FTIR spectrum of the bacteria (Schmitt and Fleming, 1998SCHMITT, J., FLEMING, HANS-CURT. FTIR-spectroscopy in microbial and material analysis. International Biodeterioration & Biodegradation, v. 41, p. 1-11, 1998.; Deo and Natarajan., 1988DEO, N., NATARAJAN, K.A. Studies on interaction of Paenibacillus polymyxa with iron ore minerals in relation to beneficiation. International Journal of Mineral Processing, v. 55, n. 1, p. 41-60, 1988.).
After the cellular adaptation, a change in the FTIR spectra of the bacterial cells was observed, which may imply that there was a modification in the bacterial cell wall functional groups. The spectrum of the apatite-adapted bacterium is shown in Fig. 3a. Slightly more intense bands are observed at 1455 cm-1 and at 1745 cm-1, suggesting an increase in the content of the fatty acids and triglycerides. However, a higher intensity increment in the spectrum was observed between 2920 cm-1 and 2850 cm-1, suggesting an increase in the content of lipids, proteins, fatty acids and polysaccharides; and also between 1150 and 990 cm-1, where higher signal intensities were especially detected for the band at 1051 cm-1, which also moved to 1043 cm-1. This indicated a higher content of phospholipids and polysaccharides. Additionally, the glycogen band (1150 cm-1) was slightly more intense, and the weak band at 992 cm-1 was no observed after the cellular adaptation. Finally, the bands between 609 and 573 cm-1 are relatively weak in intensity.
a) FTIR spectrum of the R. opacus bacteria adapted to apatite; b) FTIR spectrum of the R. opacus bacteria adapted to quartz.
Additionally, the spectrum of the quartz-adapted cells is shown in Fig. 3b. A more intense band at 2920 cm-1 was observed, corresponding to CH2 asymmetric stretching from lipids. Additionally, a higher intensity of the band corresponding to the triglycerides (1745 cm-1) was observed. However, the bands corresponding to amide I (proteins) and carboxylate ions (1658 cm-1, 1633 cm-1 and 1400 cm-1) were significantly reduced in intensity. Again, a higher intensity spectrum after cellular adaptation was observed for the bands between 1150 and 990 cm-1. The bands corresponding to nucleic acid and/or glycogen (1151 cm-1 and 1081 cm-1) increased in intensity. Finally, slightly higher intensities of several bands between 800 and 700 cm-1 were observed due to amide IV and C=O bending in proteins (Schmitt and Fleming., 1998SCHMITT, J., FLEMING, HANS-CURT. FTIR-spectroscopy in microbial and material analysis. International Biodeterioration & Biodegradation, v. 41, p. 1-11, 1998.).
The literature review showed that there is a lack in the characterization of mineral-adapted cells; however, Sharma et al. (2001)SHARMA, P.K., RAO, K.H., FORSSBERG, K.S.E., NATARAJAN, K.A., Surface chemical characterisation of Paenibacillus polymyxa before and after adaptation to sulfide minerals. International Journal of Minerals Processing, v. 62, p. 3-25, 2001. studied the characterization of P. polymyxa before and after adaptation to sulfide minerals. According to the authors, the adaptation process caused a higher secretion of metabolites (proteins and polysaccharides), but the galena-adapted cells produced the highest amount of secretions. The FTIR spectra of the bacteria after adaptation presented some changes, and in general, higher bands were present for 3053 (amide B vibrations) 2958, and 2922 cm-1 (characteristic of hydrocarbon chains).
Therefore, in our study, the cellular adaptation promoted a higher content of fatty acids, proteins, phospholipids and polysaccharides in the structure of the mineral-adapted bacteria, whereas a lower content of proteins was observed in the quartz-adapted bacteria. This higher production of biopolymers is also shown in Table 2, and this is in accordance with related works (Botero et al., 2008BOTERO, A.E.C., TOREM, M.L., MESQUITA, L.M.S. Surface chemistry fundamentals of biosorption of Rhodococcus opacus and its effect in calcite and magnesite flotation. Minerals Engineering, v. 21, n. 1, p. 83-92, 2008.; Natarajan, 2006NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006.; Sharma et al., 2001SHARMA, P.K., RAO, K.H., FORSSBERG, K.S.E., NATARAJAN, K.A., Surface chemical characterisation of Paenibacillus polymyxa before and after adaptation to sulfide minerals. International Journal of Minerals Processing, v. 62, p. 3-25, 2001.). Consequently, the presence of these components in the mineral-adapted bacteria will promote a higher hydrophobic and partially hydrophilic character.
3.4 Microflotation experiments
The flotability of apatite and quartz (highly hydrophilic minerals) as a function of pH value, using the bacterial cells and mineral-adapted cells, are presented in Fig. 4a and 4b, respectively. Before cellular adaptation, we observed that the highest flotability of both minerals was attained at pH 5. We achieved values of approximately 45% and 14% for apatite and quartz, respectively, after 2 min of flotation. Additionally, we observed that apatite can be floated at a pH range between 3 and 7. However, the flotability of quartz was insignificant for other values of pH. The use of the apatite-adapted cells during the bioflotation of apatite promoted a higher flotability in the pH range between 3 and 9 (Fig. 4.a). Nevertheless, a significant increment was only observed at pH 3, where the flotability increases from approximately 10% to 55%. However, when the quartz-adapted cells were used, the flotability of apatite only improved at pH 3 to a value of 45%. A similar behavior was observed during the bioflotation of quartz (See Fig. 4b) in which a higher flotability of quartz was observed when the quartz-adapted cells were used. This is in accordance with flotation studies using mineral-adapted bacteria (Natarajan, 2006NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006.) in which the flotability of quartz was enhanced after cellular adaptation. The improvement presented in quartz flotability was not as significant as in the bioflotation of apatite. Additionally, the flotability of quartz was not affected when the apatite-adapted bacterium was used. This conclusion demonstrates the generation of a mineral-specific bioreagent, which can increase the selectivity of the flotation process (Natarajan, 2006NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006.). Lastly, it is possible to identify a selectivity window to separate apatite from quartz in the pH 3, by using the apatite-adapted bacteria as collector.
a) Flotability of apatite using the adapted and unadapted R.opacus bacteria as a bioreagent. Bacterial concentration 0.15 g L-1. Flotation time: 2 min. b) Flotability of quartz using the adapted and unadapted R.opacus bacteria as a bioreagent. Bacterial concentration 0.15 g L-1. Flotation time: 2 min.
Finally, the flotability of either mineral was unaffected when the bacterial cells grown under different culture times were used. As example the flotability of apatite and quartz was around 46% and 12%, respectively, when the culture time of the bacteria was 96 h. Thus, if the metabolic products were excreted to the medium and the flotability of the minerals was not affected, the wall cell of the bacteria was not affected by the longer culture times.
Our results may suggest that higher culture times did not affect the cell wall of the bacteria; however, a higher metabolic production into the suspension was detected, as supported by the zeta potential, surface tension and microflotation results. This effect was also confirmed by Didyk and Sadoswski (2013)DIDYK, A.M., SADOWSKI, Z. Flotation of serpentinite and quartz using biosurfactans. Physicochemical Problems of Mineral Processing, v. 48, n. 2, p. 607-618, 2013. for the culture of Bacillus circulans and Streptomyces sp. However, the results also showed that the cellular adaptation caused a modification in the bacterial cell wall. A more detailed explanation of these behaviors can be found in other works (Natarajan, 2006NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006.; Sarvamangala and Natarajan, 2011SARVAMANGALA, H., NATARAJAN, K.A. Microbially induced flotation of alumina, silica/calcite from haematite. International Journal of Minerals processing, v. 99, p. 70-77, 2011.; Sarvamangala et al., 2012SARVAMANGALA, H., NATARAJAN, K.A., GIRISHA, S.T. Biobeneficiation of iron ores. International Journal of Mining Engineering and Mineral Processing, v. 1, v. 2, p. 21-30, 2012., 2013SARVAMANGALA, H., NATARAJAN, K.A., GIRISHA, S.T. Microbially-induced pyrite removal from galena using Bacillus subtilis. International Journal of Minerals Processing, v. 120, p. 15-21, 2013.) in which the authors confirmed that the presence of different minerals during the growth of various microbial promotes the generation of different metabolic products (mainly proteins and polysaccharides), or changes their production rate. The authors also confirmed that the predominant adsorption of polysaccharides will render a mineral surface more hydrophilic. In regards to proteins, it will enhance their hydrophobicity. This result can help explain the results presented in this work. The cellular adaptation may stimulate a higher production of proteins on the bacterial cell wall, which will adsorb on the mineral surface, rendering it more hydrophobic. Furthermore, this effect was more relevant during the cellular adaptation to apatite, and the proteins produced may have a higher selectivity for the apatite surface, which will explain the microflotation results.
4. Conclusions
The zeta potential evaluation of the R. opacus before and after adaptation to the presence of a mineral substrate showed that the bacterial cells modified its zeta potential profiles; however, no significant change was observed with higher culture times. Additionally, the surface tension measurements associated with the bacterial growth curve showed that higher culture times caused a higher production of metabolites, whereas the cellular adaptation may promote a modification in the bacterial cell surface. Additionally, the FTIR results showed that the cellular adaptation caused a modification in the bacterial spectra. The adaptation to apatite promoted a slight increase in the fatty acids, proteins, phospholipids and polysaccharides. Meanwhile, the adaptation to quartz had a slightly similar effect; however, a lower content of proteins was observed. Finally, the use of mineral-adapted bacteria as biocollectors produced an increase in the flotability of both minerals. The flotability of apatite achieved values of approximately 55% and 45% for a pH of 3 and 5, respectively, when apatite-adapted bacterium was used, whereas the flotability of quartz attained an improvement only at pH 3 when the quartz-adapted bacterium was used. However, we did not observe any change in the flotability of both minerals when the microorganism that was developed under a higher culture time was used. Thus, this suggests that the flotability of both minerals can be enhanced if the microorganism is developed in a culture medium that contains the same mineral as the substrate. Moreover, it is possible to increase the ability of the R. opacus bacteria to act as a biocollector of apatite due to the cellular adaptation process.
Acknowledgments
The authors acknowledge CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico), ITV-VALE, CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) and FAPERJ (Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro) for their financial support.
References
- AMÉZCUA-VEGA, C., POGGI-VARALDO, H.M., ESPARZA-GARCÍA, F., RÍOS-LEAL, E., AND RODRÍGUEZ-VÁZQUEZ, R. Effect of culture conditions on fatty acids composition of a biosurfactant produced by Candida ingens and changes of surface tension of culture media. Bioresource Technology, v. 98, n. 1, p. 237-240, 2007.
- BOTERO, A.E.C., TOREM, M.L., MESQUITA, L.M.S. Fundamental studies of Rhodococcus opacus as a biocollector of calcite and magnesite. Minerals Engineering, v. 20, n. 10, p. 1026-1032, 2007.
- BOTERO, A.E.C., TOREM, M.L., MESQUITA, L.M.S. Surface chemistry fundamentals of biosorption of Rhodococcus opacus and its effect in calcite and magnesite flotation. Minerals Engineering, v. 21, n. 1, p. 83-92, 2008.
- CAMMAROTA, M.C. Exopolysaccharides production and microbial adhesion Rio de Janeiro: Departamento de Bioquímica, Univerdidade Federal do Rio de Janeiro, 1998. (Tese de Doutorado).
- DEO, N., NATARAJAN, K.A. Studies on interaction of Paenibacillus polymyxa with iron ore minerals in relation to beneficiation. International Journal of Mineral Processing, v. 55, n. 1, p. 41-60, 1988.
- DEO, N., NATARAJAN, K.A. Interaction of Bacillus polymyxa with some oxide minerals with reference to mineral beneficiation and environmental control. Minerals Engineering v. 10, n. 12, p. 1339-1354, 1997.
- DIDYK, A.M., SADOWSKI, Z. Flotation of serpentinite and quartz using biosurfactans. Physicochemical Problems of Mineral Processing, v. 48, n. 2, p. 607-618, 2013.
- EL-MIDANY, A.A., ABDEL-KHALEK, M.A. Reducing sulfur and ash from coal using Bacillus subtilis and Paenibacillus polymyxa. Fuel, v. 115, p. 589-595, 2014.
- FAHARAT, M., HIRAJIMA, T., SASAKI, K., AIBA, Y., DOI, K. Adsorption of SIP E. coli onto quartz and its applications in froth flotation. Minerals Engineering, v. 21, p. 389-395, 2008.
- GARIP, S., GOZEN, A.C., SEVERCAN, F. Use of Fourier transform infrared spectroscopy for rapid comparative analysis of Bacillus and Micrococcus isolates. Food Chemistry, v. 113, p. 1301-1307, 2009.
- LAMBERT, W.,D., DU, L., MA, Y., LOHA, V., BURAPATANA, V., PROKOP, A., TANNER, R., D., PAMMENT, N.B. The effect of pH on the foam fractionation of b-glucosidase and cellulase. Bioresource Technology, v. 87, p. 247-253, 2003.
- MERMA, A.G. Fundamental aspects in the apatite-quartz bioflotation system using the R. opacus bacteria as bioreagent Rio de Janeiro: Departamento de Engenharia Química e de Materiais, Pontifícia Universidade Católica do Rio de Janeiro, 2012. (Tese de Doutorado).
- MERMA, A.G., TOREM, M.L., MORAN, J.V., MONTE, M.B.M. On the fundamental aspects of apatite and quartz flotation using a gran positive strain as a bioreagent. Minerals Engineering, v., 48, p. 61-67, 2013.
- MESQUITA, L.M.S., LINS, F.A.F., TOREM, M.L. Interaction of a hydrophobic bacterium strain in a hematite-quartz flotation system. International Journal of Mineral Processing, v. 71, p. 31-44, 2003.
- MOZES, N., LÉONARD, A.J., ROUXHET, P.G. On the relations between the elemental surface compositions of yeasts and bacteria and their charge and hydrophobicity. Biochimica et Biophysica Acta, v. 945, p. 324-334, 1988
- NATARAJAN, K.A., DEO, N. Role of bacterial interaction and bioreagents in iron ore flotation. International Journal of Minerals Processing, v. 62, p. 62: 143-147, 2001.
- NATARAJAN, K.A., Microbially-induced mineral flotation and flocculation: prospects and challenges. Proceedings of XXIII International Mineral Processing Congress, p. 487-498, 2006.
- OLIVEIRA, J.A. Saponification degree of vegetable oils at selective flotation of apatite from carbonates ore Ouro Preto: Departamento de Engenharia de Minas, Universidade Federal de Ouro Preto, 2005. (Dissertação de Mestrado).
- RAO, K.H., SUBRAMANIAN, S. Bioflotation and Bioflocculation of relevance to minerals bioprocessing. IN: Donati, R. E., SAND, W. Microbial processing of metal sulfides 2007. p. 267-286.
- SANTHIYA, D., SUBRAMANIAN'S. NATARAJAN, K.A. Surface chemical studies on sphalerite and galena using extracellular Polysaccharides isolated from Bacillus polymyxa. Journal of Colloid and Interface Science, v. 256, n. 2, p. 237-248, 2002.
- SARVAMANGALA, H., NATARAJAN, K.A., GIRISHA, S.T. Microbially-induced pyrite removal from galena using Bacillus subtilis. International Journal of Minerals Processing, v. 120, p. 15-21, 2013.
- SARVAMANGALA, H., NATARAJAN, K.A. Microbially induced flotation of alumina, silica/calcite from haematite. International Journal of Minerals processing, v. 99, p. 70-77, 2011.
- SARVAMANGALA, H., NATARAJAN, K.A., GIRISHA, S.T. Biobeneficiation of iron ores. International Journal of Mining Engineering and Mineral Processing, v. 1, v. 2, p. 21-30, 2012.
- SCHMITT, J., FLEMING, HANS-CURT. FTIR-spectroscopy in microbial and material analysis. International Biodeterioration & Biodegradation, v. 41, p. 1-11, 1998.
- SHARMA, P.K., RAO, K.H., FORSSBERG, K.S.E., NATARAJAN, K.A., Surface chemical characterisation of Paenibacillus polymyxa before and after adaptation to sulfide minerals. International Journal of Minerals Processing, v. 62, p. 3-25, 2001.
- STICKLAND, L. H. The determination of small quantities of bactéria by means of biuret reaction. J. Gen. Microbiol, v. 5, p. 698-703, 1951.
- SUBRAMANIAN, S., SANTHIYA, D., NATARAJAN, K.A. Surface modification studies on sulphide minerals using bioreagents. International Journal of Mineral Processing, v. 72, p. 175-188, 2003.
- VAN DER WAL, A., NORDE, A.A.J.B., ZEHNDER, B., LYKLEMA, J. A determination of the total charge in the cell walls of gram-positive bacteria. Colloids and Surfaces B: Biointerfaces, v. 9, p. 81-100, 1997.
- VASANTHAKUMAR, B., RAVISHANKAR, H., SUBRAMANIAN, S. Microbially induced selective flotation of sphalerite from galena using mineral-adapted strains of Bacillus megaterium. Colloids and Surfaces B: Biointerfaces, v. 112, p. 279-286, 2013.
- VENYAMINOV, S.Y., KALNIN, N.N. Quantitative IR spectrophotometry of peptide compounds in water (H2O) solutions. I. spectral parameters of Amino Acid Residue Absorption Bands. Biopolymers, v. 30, p. 1243-1257, 1990.
- VILINSKA, A., RAO, K.H. Leptosririllum ferrooxidans-sulfide mineral interactions with reference to bioflotation and bioflocculation. Trans. Nonferrous Met. Soc. China, v. 18, p. 1403-409, 2008.
- YEMN, E.W. WILLIS, A.J. The estimation of carbohydrate in plant extracts by anthrone. The Biochemical Journal, v. 57, p. 508-514, 1954.
Publication Dates
-
Publication in this collection
Jan-Mar 2017
History
-
Received
11 May 2016 -
Accepted
01 July 2016