Abstract
The developed model is an association of thermodynamic calculations for dissolution of alloys, slag formers and the deoxidation reaction in the molten steel with two artificial neural network (ANN) models trained with industrial data, to predict the molten steel temperature drop from the blowing end of the BOF until the first measurement at secondary metallurgy. To calculate the associated energy for deoxidation, an experiment was designed to set up the parameters for oxygen partitioning among deoxidants, with timed aluminum addition during teeming being the main parameter. The temperature control in the teeming stage presented a standard deviation for the error of prediction of 5.46 ºC, for transportation from the rinsing station to the secondary metallurgy of 2.79 ºC. The association of all calculations presented an error standard deviation of 7.49 ºC. The operational validation presented superior accuracy compared with the current method for controlling the temperature, resulting in a reduction in the aluminum consumption for heating at secondary metallurgy with a potential economy of U$ 4.07 million per year for a steel shop producing 5 million tons of steel yearly. The artificial neural network model confirmed its capacity for modeling a complex multivariable process and the separation of thermodynamic calculation provides a better adaptability to different steel grades with different teeming strategies.
Keywords:
molten steel temperature control; artificial neural network; thermodynamic calculations
1. Introduction
Heat losses during steelmaking are unavoidable but should be as small and predictable as is possible. Extreme temperatures at the casting station could lead to defects related to segregation and grain size/shape distribution, and possibly to breakouts. A too high Basic Oxygen Furnace (BOF) end blow temperature implies on higher energy and lime consumption and refractory losses.
As already stressed by others, the complexity of the steelmaking process and nonlinear relationship between the process parameters makes it difficult to model the input-output process parameters and forecast steelmaking operations (Laha, 2015LAHA, Dipak, REN, Ye, SUGANTHAN, Ponnuthurai N. Modeling of steelmaking process with effective machine learning techniques. Expert systems with applications, v. 42, n. 10, p. 4687-4696, 2015.). Due to this feature, statistical models and artificial neural networks (ANN) have been studied to model a variety of complex processes. Typical steelmaking operations, such as Ladle Furnace (LF), Tian (2008)TIAN, Huixin, MAO, Zhizhong, WANG, Yan. Hybrid modeling of molten steel temperature prediction in LF. ISIJ international, v. 48, n. 1, p. 58-62, 2008. and Sampaio (2006)SAMPAIO, Patrícia Teixeira, BRAGA, Antônio Pádua, FUJII, Takeshi. Modelagem térmica de um forno-panela utilizando redes neurais artificiais. Belo Horizonte: Universidade Federal de Minas Gerais, 2006. (Tese de Doutorado).; BOF blowing end dynamic control of carbon and/or temperature were performed by Wei (2016)Wei Li, Xinchun Wang, XushengWanga, Hong Wanga. Endpoint prediction of BOF steelmaking based on BP neural network combined with improved PSO. Chemical Engineering Transactions, v. 51, p.475-480, 2016., Liu (2014)LIU, Hui, WANG, Bin, XIONG, Xin. Basic oxygen furnace steelmaking end-point prediction based on computer vision and general regression neural network. Optik-International Journal for Light and Electron Optics, v. 125, n. 18, p. 5241-5248, 2014., Liu (2014)LIU, Hui, WANG, Bin, XIONG, Xin. Basic oxygen furnace steelmaking end-point prediction based on computer vision and general regression neural network. Optik-International Journal for Light and Electron Optics, v. 125, n. 18, p. 5241-5248, 2014., Wang (2012)Wei Li, Xinchun Wang, XushengWanga, Hong Wanga. Endpoint prediction of BOF steelmaking based on BP neural network combined with improved PSO. Chemical Engineering Transactions, v. 51, p.475-480, 2016., Bing-Yao (2011)BING-YAO, Cai, HUI, Zhao, YOU-JUN, Yue. Research on the BOF steelmaking endpoint temperature prediction. In: Mechatronic Science, Electric Engineering and Computer (MEC). In: INTERNATIONAL CONFERENCE ON. IEEE, 2011. p. 2278-2281., Rajesh (2010)RAJESH, Narra, KHARE, Malaya Ranjan, PABI, Shyamal Kumar. Feed forward neural network for prediction of end blow oxygen in LD converter steel making. Materials Research, v. 13, n. 1, p. 15-19, 2010., and Meradi (2008)MERADI, H., BOUHOUCHE, S., LAHRECHE, M. Prediction of bath temperature using neural networks. World Academy of Science, Engineering and Technology, v. 24, p. 946-950, 2008.; phosphorus at endpoint control Wang (2014)WANG, Hong-Bing, JUN, C. A. I., KAI, F. E. N. G. Predicting the endpoint phosphorus content of molten steel in BOF by two-stage hybrid method. Journal of Iron and Steel Research, International, v. 21, p. 65-69, 2014.. Different strategies were applied according to the operational data used for the training process, sub-lance measurements, visual characteristics of flame, gas analysis, etc. Chen et al (1997)CHEN, W., DUAN, G., OU, C. Neural network applied to predicting molten steel temperature profile from converter to continuous casting. Iron and Steel. China, v. 32, n. 8, p. 30-32, 1997. developed an ANN model aiming to predict the temperature evolution from the converter to the casting machine. A hybrid model defined as a thermal model based on first principles in which the tuning parameters were calculated by intelligent algorithm has been presented by Tian (2008)TIAN, Huixin, MAO, Zhizhong, WANG, Yan. Hybrid modeling of molten steel temperature prediction in LF. ISIJ international, v. 48, n. 1, p. 58-62, 2008. to control molten steel temperature at an LF. Fei et al(2014)FEI, H. E. et al. Hybrid model of molten steel temperature prediction based on ladle heat status and artificial neural network. Journal of Iron and Steel Research, International, v. 21, n. 2, p. 181-190, 2014. describe a hybrid model built to predict the steel temperature from BOF to a caster passing through a Bubbling Station(BS) and an LF, both forwards and backwards; their model assesses the ladle thermal status defined by tracking the teeming ladle operational data, integrating it with ANN trained with other operational factors, such as material addition.
The hybrid model herein proposed employs a combination of thermodynamic calculations and an ANN for each metallurgical station. Some heating/cooling effects such as those from alloy additions, deoxidation can be readily estimated using first principles. The ANN is trained with other operational factors correlated with molten steel temperature loss that present high mathematical complexity, such as the heat transfer from molten steel to refractory linings of the teeming ladle and BOF. Figure 1 shows a schematic of the steel producing routes. The regular route is Basic Oxygen Furnace (BOF), pouring, rinsing station (RS), secondary metallurgy at RH degasser or Aluminum Heating Station (AHS), Continuous Casting Machine (CCM); the so-called direct route is Basic Oxygen Furnace (BOF), pouring, rinsing station (RS), Continuous Casting Machine (CCM).
This study is concerned with temperature control between the BOF blowing end and the first temperature measurement at RS, T2 - T1 ; also with temperature control between departure at RS and arrival at the secondary metallurgy (RH, AHS), T3-T2. A better prediction of both T2-T1 and T3-T2 leads to a more predictable casting temperature and smaller end blow temperature. In both cases, a hybrid Neural Networks - Thermodynamic model will be employed. Such model requires: 1- to estimate the thermal effects from additions of fluxes and alloying elements using the first principles of thermodynamics); 2- to estimate the oxygen partitioning among deoxidants as a function of timing of addition and relative proportion through a series of controlled experiments and thereafter, to estimate the thermal effect for deoxidation; 3- to train the artificial neural networks after discounting the known thermal effects and to validate the model.
2. Materials and methods
Three aspects are central to this model. Calculation of thermal effects due to alloying; estimation of oxygen partition among deoxidizers, and subsequent thermal effects; ANN training after data filtering from these effects. They are described as follows.
Thermodynamic calculations
Thermal effects due to the addition of CaO, MgO, Al2O3, SiO2, CaCO3, MgCO3 and, C, Si, Mn, Al, Fe are taken into consideration. For each mol of a given slag former, the thermal effects are given by:
Where ( HT-H298 ) stands for heat content, x for fraction of molten oxide and ∆Hmelting for enthalpy of melting. Evaluating the effects of carbonate addition includes the heat of carbonate dissociation ( MCO3 = MO+ CO2) and carbon dioxide heating. For each grade of steel, processing temperature, timing and mass of each addition the above-mentioned variables can be evaluated using thermochemical data from Slag Atlas(1995), Kelley(1961)KELLEY, Kenneth Keith, KING, E. G. Contributions to the data on theoretical metallurgy. US Government Printing Office, 1961., Hultgren(1973)HULTGREN, Ralph et al. Selected values of the thermodynamic properties of binary alloys. National Standard Reference Data System, 1973. and others (Morris, 2011MORRIS, Arthur E., FINE, H. Alan, GEIGER, Gordon. Handbook on material and energy balance calculations in material processing, includes CD-ROM. John Wiley& Sons, 2011.).
A similar procedure is due to additions of alloying elements such as Silicon (and alike). Here the thermal contribution is given by
Where ∆Hdissolution is the heat of formation of a liquid Fe-Y solution. Thermodynamic data are from sources such as Kelley(1964) and Hultgren (1973HULTGREN, Ralph et al. Selected values of the thermodynamic properties of binary alloys. National Standard Reference Data System, 1973.).
The calculated ∆H value is divided by the heat capacity of molten steel to estimate the temperature variation ( ∆Tsteel ) regarding the additions of synthetic slags and alloys. Calculations are straightforward and agreement with the literature (Turkdogan et al) is apparent; as a matter of fact, the main difficulty is to assess the actual state of the presumed slag.
Evaluation of oxygen partitioning among deoxidants
Oxygen content at the BOF blow end is, of course, a function of the blow pattern. The main sources of oxygen are the steel dissolved content, oxygen from the carried over slag and slag formation additions, oxygen from the atmospheric pick-up. Deoxidants and alloying elements are added following a practice which includes a shape (bulk, powder, wire, etc), the sequence of addition and oxygen affinity specifics. Evaluating the fraction of oxygen reacting with a specific element, Zi, is a complex matter involving multi equilibria reactions in a metal-slag-gas system; evaluating this fraction is of considerable importance since, for each reaction, the amount of released heat varies remarkably.
Thus a series of experiments have been performed in order to assess Zi for a given grade of steel. The deoxidation mass balance can be written as
The amount of Aluminum spent for deoxidation purposes can be estimated as
Where MAl-pouring stands for the mass of aluminum added at the pouring station; %AlRS aluminum content as sampled at the rinsing station; Msteel is the mass of steel; MO and MAl are the atomic masses of oxygen and aluminum. The other contributions are evaluated following the same procedure.
The material addition during the pouring phase was divided into two batches: the first batch lasts till 10% of the total pouring time; the second batch starts at 30% or 50% of total pouring time. The steels targeted for these experiments show average end blow carbon and manganese contents of 0.03% and 0.10% and 600 ppm of oxygen; average additions are 50 kg of coke, 1100 kg of high carbon iron-manganese alloy and 500 kg of aluminum. The aluminum addition was made completely in the second batch or 70% of the total mass in the first batch. Pouring from BOF was automatically interrupted if slag was detected inside the pouring channel. The latter technique allowed to reduce the noise from the carryover slag. For all experiments, end blow temperature, oxygen content and steel sampling have been done by Multi-Lab Celox; temperature at RS, AHS and RH are given by immersion Pt-Pt/Rh thermocouples.
The heat for steel deoxidation during teeming ( ∆HDeoxidation ) can be estimated by the sum of the thermal effects of each deoxidizing reaction assuming that the available oxygen is partitioned according to Zi.
Artificial Neural Network
According to BISHOP(1995)BISHOP, Christopher M. Neural networks for pattern recognition. Oxford university press, 1995. and SIMON(2001), the Artificial Neural Networks (ANN) have the capacity of inferring functions as a result of the supervised learning process. The regression problem to predict the temperature changes related to operational factors other than from material additions in the molten steel will be addressed by two ANN models. The capacity for modeling complex multivariable nonlinear functions from a noisy operational dataset and the low computational time needed to perform an online prediction are the main characteristics that make the ANN model a suitable tool to perform the calculation of temperature loss of molten steel in an industrial environment. The ANN was developed using Statistica software, version 13.1. The models were a multi-layer perceptron (MLP), retro-propagation trained using the sum of squares as the error function.
A first ANN was developed for controlling/predicting the temperature loss between the start of tapping at BOF and the first temperature measurement at RS, T2 - T1, taking into consideration the production of Ultra Low Carbon Steel (ULC) with the specification of C ≤ 0.003%, Si ≤ 0.034%, 0.105% ≤ Mn ≤ 0.155%, P ≤ 0.020%, S ≤ 0.015%, 0.020%≤ Al ≤ 0.045%, 0.050% ≤ Ti ≤ 0,090%. The ULC steel grades have been chosen because they are the simplest steel grades regarding the complexity of material addition during the teeming process, since the deoxidation and alloying is performed at secondary metallurgy, requiring only 500 Kg of lime ( CaO = 92%, MgO=0.7%, SiO2=1.4%, CO2 = 3.30%, H2O = 2.15%, Al2O3=0.2%, Fe2O3=0.2%) and 1500 Kg of synthetic slag (CaO = 54.5%, MgO=23.8%, SiO2=1.52%, CO2 = 12%, H2O = 5.50%, Al2O3=2.42%, Fe2O3=0.2%). The thermal effects from these additions are discounted from measured T2-T1 to provide the temperature drop ∆TANN Teeming associated with other operational variables. Then training the ANN takes into consideration: ladle life (number of heats); time of empty ladle (time elapsed from end of casting and beginning of pouring); steel resting time at the ladle during the previous heat; time of empty BOF (time elapsed between successive heats); elapsed time between measuring temperature at blow end and starting of teeming; end blow temperature; rinsing time till first sampling at RS; pouring time; slag mass; and steel mass. Data from some 450 heats of ultra-low carbon steel have been used for training with eleven hidden layers, and hyperbolic tangent as activation function both at the hidden layer and exit layer.
The second ANN deals with temperature control (∆TANN RS ) during the transportation period from RS to secondary metallurgy (RH or AHS), T3-T2. It takes into consideration only the thermal effects related to heat transfer phenomena. For this one, the main variables were: time elapsed between the first temperature measurement at RS and RH degasser; rinsing time; time of empty ladle; refractory life (number of heats); the first temperature as measured at the RS; and time with steel at the previous heat. Data from some 400 heats of ultra-low carbon steel where no addition was made between the T2 and T3 measurements have been used for training with seven hidden layers, and exponential activation function both at the hidden layer and exit layer.
3. Results and discussion
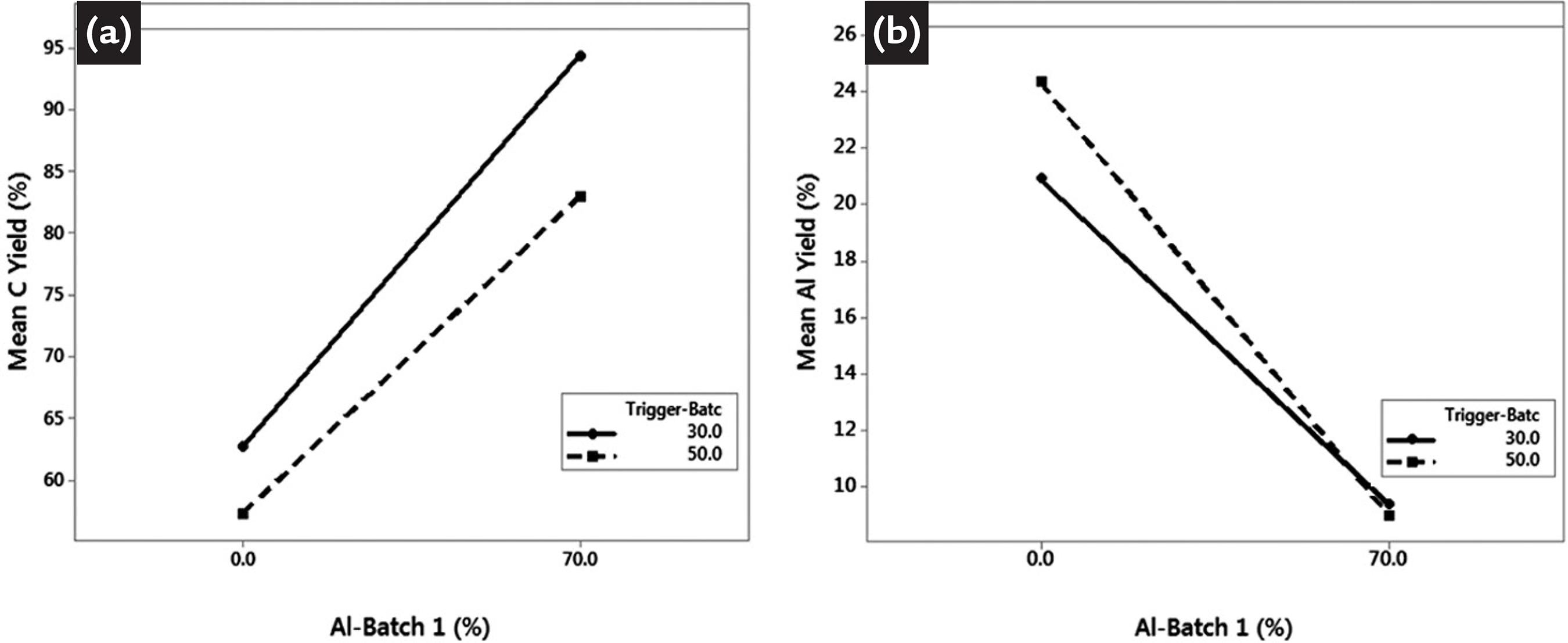
The experiments for determination of oxygen partitioning among deoxidants have shown that when 70% of total mass of aluminum for deoxidation and alloying the steel is added at the first batch of materials (10% of total pouring time) to be added during pouring phase, the obtained carbon yield measured after stirring time at ladle rinsing station increases as expected. If the second batch of materials to be added during pouring phase is delayed to 50% of total pouring time and 100% of the aluminum mass is added only in the second batch, less aluminum protection is provided. These effects can be seen in the factorial plot; notice that the carbon yield changes in Figure 2 (a). In accordance, the aluminum yield measured after stirring time at the ladle rinsing station is affected in an opposing way, if aluminum addition to the second batch of materials is delayed; its yield will naturally increase, since some of the oxygen has had the opportunity to react with other elements, such as carbon, see Figure 2 (b).
Factorial plot for different configuration of aluminum addition during teeming to (a) carbon yield and (b) aluminum yield.
In order to save aluminum, the standard procedure comprises no aluminum addition during the first batch (10% of the teeming time) and the total mass of aluminum being added at the second batch (50% of the teeming time). The average oxygen partition among deoxidants for the studied steel grade is calculated by the mass balance as follows: ZAl=0.92 ; ZC=0.07 ; ZMn=0.01 and ZSi=0. These model parameters must be evaluated for each different steel grade deoxidized during teeming.
The resulting correlation coefficient during the model validation with a new operational dataset was equal 0.85. The model error (measured value - predicted value) standard deviation is equal 5.46ºC, error mean = 0.301 ºC and 95% of the prediction error is in the interval of -10.79 ºC to +10.66 ºC
Figure 3 shows a comparison between the temperature drop during teeming,( T2- T1 )+∆ TSlagForm, measured values against values given by the neural network trained with ULC grade operational data.
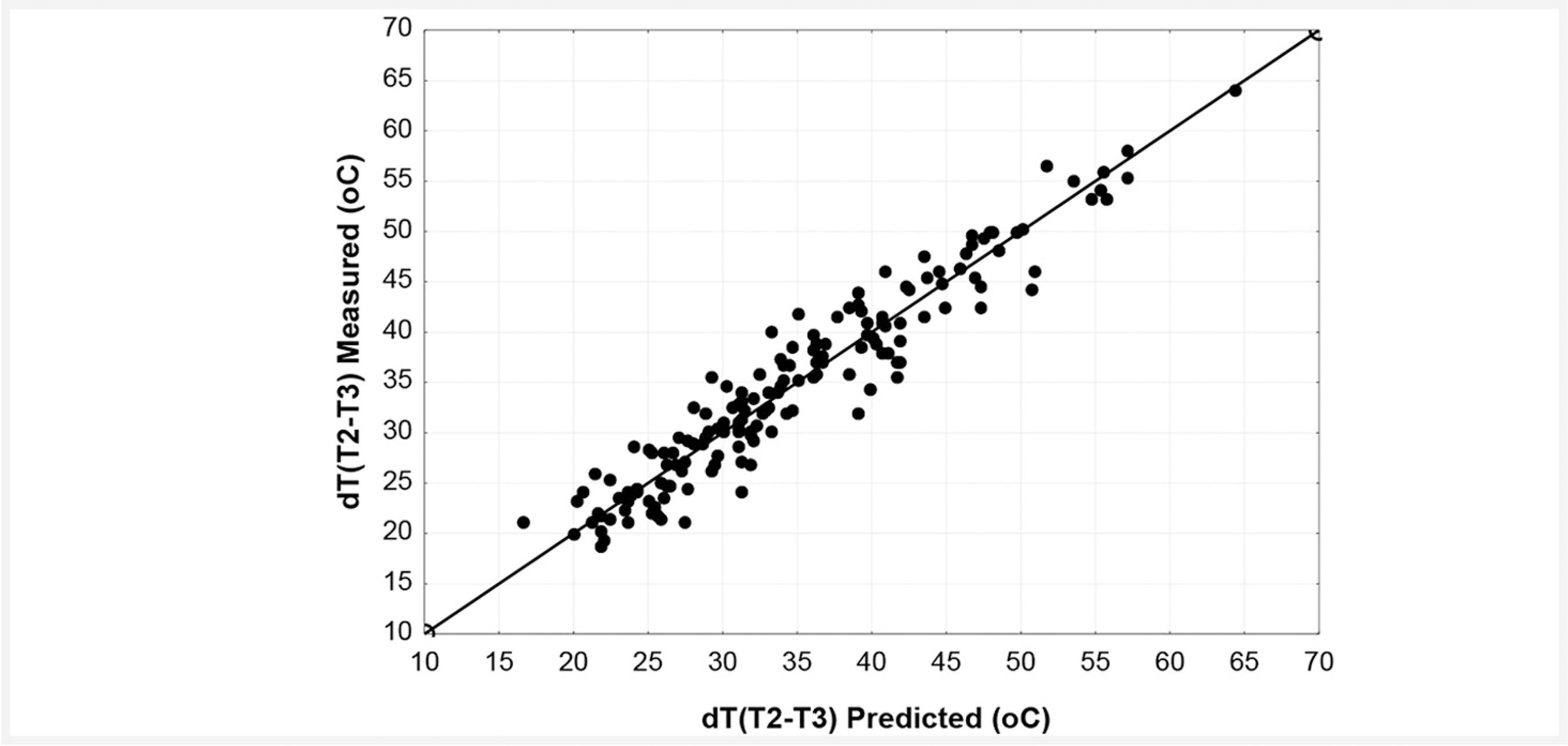
The metallurgical work at the rinsing station is simpler than at the teeming station. It consists essentially of rinsing inert gas and keeping control of composition and temperature. Thus a much better prediction of temperature drop from temperature measurement at the rising station (T2) to first temperature measurement at secondary metallurgy (T3) can be done, as is seen in Figure 4. As a matter of fact, the resulting correlation coefficient during the model validation with a new operational dataset can be as high as 0.95.
Temperature drop during transportation; a comparison between measured and predicted values.
When the quality of the prediction is improved, the standard deviation of the error during model validation drops to 2.79 ºC. Other basic statistics are error mean= -0.042 ºC and 95% of prediction error is in the interval of -5.58 ºC to +5.49 ºC. The expected effects of elapsed time from temperature measurement at the rinsing station to first temperature measurement at secondary metallurgy and rinsing time can be obtained with a higher degree of confidence.
Consolidating the thermodynamic calculation, operational parameters for oxygen partitioning among deoxidants and the two artificial neural networks, the global model to calculate T3-T1 can be written as:
Where: ∆TSlagForm, ∆TAlloy and ∆TDeox are the steel temperature changes related to the addition of slag formers, alloying and deoxidation respectively. ∆TANN Teeming and ∆TANN RS are the temperature changes calculated by ANN models for teeming and transportation from rinsing station to secondary metallurgy, respectively. The combined models have resulted in a global (T3-T1) model. Herein, 34 industrial runs have been analyzed using this model. Figure 5 exemplifies the findings. There seems to be a good agreement between measured and predicted values for the first temperature measurement at the secondary metallurgy (T3).
Measured vs predicted values of first temperature at secondary metallurgy taking into consideration alloying, deoxidation and slag formers addition.
The error standard deviation of (T3-T1) model is 7.49 ºC, while other basic statistics are error mean= -2.33 ºC and 95% of the prediction error is in the interval of -11.67 ºC to +16.33 ºC. The heat complying with this model required for temperature adjustment 0.1 Kg of Al/t steel and 1.77 Kg of scrap/t of steel. The current values are 0.56 Kg of Al/t of steel and 1.74 Kg of scrap/t of steel. Taking into consideration a steel shop producing 5 Million tons of steel/year and aluminum costs of 1.77 U$/kg, the expected saving would be in the order of U$ 4.07 million/year. Less apparent, yet very important, improvements are higher alloy yield and secondary refining productivity. As less alumina is formed, the steel would be cleaner and less prone to cause nozzle blockage and internal defects.
The beneficial effects of implementing such hybrid ANN-Thermodynamics model are made apparent.
4. Conclusion
A hybrid ANN model coupled with first principles of thermodynamic estimations has been successfully developed in order to provide for a more accurate temperature prediction from the BOF blowing end to treatment start at secondary metallurgy. Filtering the raw data from known thermal effects allows a more precise ANN training. The oxygen partitioning among deoxidants must be evaluated for different addition practices during the teeming stage, since the thermal effect for deoxidation reaction can change significantly. The artificial neural network model demonstrated its capacity of mod eling nonlinear functions for complex multivariable industrial processes.
Taking in consideration the high tonnages related to the steelmaking industry the potential savings in regard to aluminum consumption are sizable.
Acknowledgements
The authors thank to CNPq, CAPES, FAPEMIG and Ternium Brasil for financial support.
References
- BISHOP, Christopher M. Neural networks for pattern recognition Oxford university press, 1995.
- BING-YAO, Cai, HUI, Zhao, YOU-JUN, Yue. Research on the BOF steelmaking endpoint temperature prediction. In: Mechatronic Science, Electric Engineering and Computer (MEC). In: INTERNATIONAL CONFERENCE ON. IEEE, 2011. p. 2278-2281.
- CHEN, W., DUAN, G., OU, C. Neural network applied to predicting molten steel temperature profile from converter to continuous casting. Iron and Steel. China, v. 32, n. 8, p. 30-32, 1997.
- EISENHÜTTENLEUTE, Verein Deutscher, ALLIBERT, M. Slag atlasVerlagStahleisen, 1995.
- FEI, H. E. et al. Hybrid model of molten steel temperature prediction based on ladle heat status and artificial neural network. Journal of Iron and Steel Research, International, v. 21, n. 2, p. 181-190, 2014.
- HAYKIN, Simon. Redes neurais: princípios e prática Bookman Editora, 2001.
- HULTGREN, Ralph et al. Selected values of the thermodynamic properties of binary alloys National Standard Reference Data System, 1973.
- KELLEY, Kenneth Keith, KING, E. G. Contributions to the data on theoretical metallurgy US Government Printing Office, 1961.
- LAHA, Dipak, REN, Ye, SUGANTHAN, Ponnuthurai N. Modeling of steelmaking process with effective machine learning techniques. Expert systems with applications, v. 42, n. 10, p. 4687-4696, 2015.
- LIU, Hui, WANG, Bin, XIONG, Xin. Basic oxygen furnace steelmaking end-point prediction based on computer vision and general regression neural network. Optik-International Journal for Light and Electron Optics, v. 125, n. 18, p. 5241-5248, 2014.
- MERADI, H., BOUHOUCHE, S., LAHRECHE, M. Prediction of bath temperature using neural networks. World Academy of Science, Engineering and Technology, v. 24, p. 946-950, 2008.
- MORRIS, Arthur E., FINE, H. Alan, GEIGER, Gordon. Handbook on material and energy balance calculations in material processing, includes CD-ROM John Wiley& Sons, 2011.
- RAJESH, Narra, KHARE, Malaya Ranjan, PABI, Shyamal Kumar. Feed forward neural network for prediction of end blow oxygen in LD converter steel making. Materials Research, v. 13, n. 1, p. 15-19, 2010.
- SAMPAIO, Patrícia Teixeira, BRAGA, Antônio Pádua, FUJII, Takeshi. Modelagem térmica de um forno-panela utilizando redes neurais artificiais Belo Horizonte: Universidade Federal de Minas Gerais, 2006. (Tese de Doutorado).
- TIAN, Huixin, MAO, Zhizhong, WANG, Yan. Hybrid modeling of molten steel temperature prediction in LF. ISIJ international, v. 48, n. 1, p. 58-62, 2008.
- WANG, Hong-Bing, JUN, C. A. I., KAI, F. E. N. G. Predicting the endpoint phosphorus content of molten steel in BOF by two-stage hybrid method. Journal of Iron and Steel Research, International, v. 21, p. 65-69, 2014.
- Wei Li, Xinchun Wang, XushengWanga, Hong Wanga. Endpoint prediction of BOF steelmaking based on BP neural network combined with improved PSO. Chemical Engineering Transactions, v. 51, p.475-480, 2016.
Publication Dates
-
Publication in this collection
Oct-Dec 2018
History
-
Received
11 Dec 2017 -
Accepted
12 Aug 2018