ABSTRACT
Treatment of cutaneous leishmaniasis (CL) is difficult due to the scarce number of drugs able to eliminate completely the intracellular form of the parasite. In the present study, the aim was to evaluate: i) phytochemical properties of extracts from Libidibia ferrea; ii) antileishmanial activity of extracts from L. ferrea against promastigotes and amastigotes of Leishmania (Leishmania) amazonensis and Leishmania (Viannia) guyanensis; iii) the effects of topical treatment using hydrogel containing active extract of L. ferrea on golden hamsters infected with L. (L.) amazonensis. Extracts from leaves, branches and fruits of L. ferrea were obtained with hexane and methanol and were tested by in vitro assays in promastigotes and murine macrophages J774 experimentally infected with amastigotes of Leishmania. Groups of hamsters with CL received topical treatment with a formulation of extract (10%) hydrogels, 50 mg.day-1 for 40 days. In vitro activity of FrMeOH (methanolic extract from fruits without seeds) resulted in significant reduction of viable promastigotes of L. (L.) amazonensis (IC50 of 15.4 µg.mL-1) and demonstrated inhibition potential of amastigote forms of L. (L.) amazonensis and L. (V.) guyanensis and low cytotoxicity in macrophages. The overall data of topical treatment with extract hydrogels (GelFrMeOH) showed that lesion sizes were significantly reduced (42.78%), with low parasite burden by RT-qPCR and culture analysis by microscopy examination, and with histopathological findings such as lower inflammatory cell infiltration 40 days after treatment. Chemical analysis demonstrated FrMeOH contains high levels of phenolic compounds. The results indicate a possible alternative therapy for CL using phytotherapics.
Keywords:
cutaneous leishmaniasis; natural products; topical formulations; experimental treatment
RESUMO
O tratamento da leishmaniose cutânea (LC) é de difícil resultado, devido ao escasso número de fármacos capazes de eliminar completamente a forma intracelular do parasita. No presente estudo, objetivou-se avaliar: i) propriedades fitoquímicas dos extratos de Libidibia ferrea; ii) a atividade antileishmania de extratos de L. ferrea contra promastigotas e amastigotas de Leishmania (Leishmania) amazonensis e Leishmania (Viannia) guyanensis; iii) os efeitos do tratamento tópico utilizando hidrogel contendo extrato ativo de L. ferrea em hamsters dourados (Mesocricetus auratus) infectados com L. (L.) amazonensis. Extratos de folhas, galhos e frutos de L. ferrea foram obtidos com hexano e metanol e foram testados por ensaios in vitro contra promastigotas e macrófagos J774 infectados com amastigotas de Leishmania. Grupos de hamsters infectados receberam hidrogel tópico com extrato (10%), 50 mg.dia-1 durante 40 dias. A atividade in vitro de FrMeOH (extrato metanólico dos frutos) mostrou redução significativa de promastigotas de L. (L.) amazonensis (IC50 de 15,4 μg.mL-1), potencial de inibição de formas amastigotas de L. (L.) amazonensis e L. (V.) guyanensis (IC50 303,36 μg.mL-1) e baixa citotoxicidade em células de macrófagos J774. Os resultados destacaram que as lesões cutâneas de animais que receberam tratamento com hidrogel + extrato (GelFrMeOH) apresentaram redução significativa (42,78%), menos úlceração e redução da carga parasitária detectada por RT-qPCR, microscopia e análise em cultura e alterações histopatológicas, como menor infiltrado inflamatório após 40 dias de tratamento. A análise química demonstrou que FrMeOH contém altos níveis de compostos fenólicos. Os resultados apontam para uma possível terapia alternativa para CL utilizando fitoterápicos.
Palavras-chave:
leishmaniose cutânea; formulações tópicas; produtos naturais; tratamento experimental
INTRODUCTION
Leishmaniasis is an infectious-parasitic disease caused by protozoa of different species of the genus Leishmania, which has the ability to invade and reproduce within the cells of the infected host’s immune system (Gontijo & Carvalho 2003Gontijo, B.; De Carvalho, M.L.R. 2003. Leishmaniose tegumentar americana. Journal of the Brazilian Society of Tropical Medicine 36: 71-80.; Pace 2014Pace, D. 2014. Leishmaniasis. Journal of Infection, 69:10-18. ). When Leishmania promastigotes are introduced into the human skin, parasitism induces the proliferation of histiocytic cells and the phagocytosis of flagellates by these tissue macrophages that, besides not being able to destroy the parasites, still allow their multiplication within the digestive vacuoles (Sacks and Kamhawi 2001Sacks, D.; Kamhawi, S. 2001. Molecular aspects of parasite-vector and vector-host interactions in leishmaniasis. Annual Review of Microbiology, 55: 453-83.).
Cutaneous leishmaniasis (CL), although widely distributed, accounts for 1/3 of its occurrence in three epidemiological regions: the Americas, the Mediterranean and Central Asia (Alvar et al. 2012Alvar, J.; Vélez, I.D.; Bern, C.; Herrero, M.; Desjeux, P.; Cano, J.; Jannin, J.; DenBoer, M. 2012. Leishmaniasis worldwide and global estimates of its incidence. PLoS One, 9 e35671.). Brazil is among the five countries with the highest number of cases of this endemic disease that represents a serious public health problem, with emphasis on the North, Midwest and Northeast regions (Costa 2005Costa, L.M.J. 2005. Epidemiologia das Leishmanioses no Brasil. Gazeta médica da Bahia, 75: 3-17.).
The drugs currently used against CL are the pentavalent antimonial salt as first choice drug, pentamidine and amphotericin B as second choice (Almeida and Santos et al. 2011Almeida, O.L.S.A.; Santos, J.B. 2011. Advances in the treatment of cutaneous leishmaniasis in the new world in the last ten years: a systematic literature review. Anais Brasileiros de Dermatologia, 86: 497-506.). However, all these present a number of problems, including parasite resistance and induction of side effects, and all the drugs currently available are parenteral administration (Silva-Lopez 2010Silva-López, R.E. 2010. Proteases de Leishmania: novos alvos para o desenvolvimento racional de fármacos. Química Nova, 33: 1541 - 1548. ).
A large part of the population, without access to medicines, seeks alternatives in popular medicine, with the therapeutic use of plants. Plant species produce a wide variety of secondary metabolites, with incomparable chemical and bioactivity diversity (Barreiro and Bolzani 2009Barreiro, E.; Bolzani, V.S. 2009. Biodiversidade: Fonte potencial para descoberta de fármacos. Química Nova, 32: 679-688.), which has allowed the development of hundreds of pharmaceutical products (Oliveira et al. 2013Oliveira, L.F.; Gilbert, B.; Villas-Boas, G.K. 2013. Potential for innovation in the treatment of leishmaniasis using plants and natural products as sources of new drugs. Revista Fitos, 8: 1-72. ). Libidibia ferrea (Mart. Ex Tul.) L.P. Queiroz (Fabaceae) is a native tree of Brazil, it is commonly known as “pauferro” or “jucá”, found mainly in Brazilian savannah areas and the Amazon region, where it was introduced by Northeastern immigrants (Costa et al. 2015Costa, L.M.; Guilhon-Simplicio, F.; Souza, T.P. 2015. Libidibia ferrea (Mart. ex Tul.) L.P. Queiroz var. ferrea: pharmacological, phytochemical and botanical aspects. International Journal of Pharmacy and Pharmaceutical Sciences, 4: 48-53.). Libidibia ferrea has a wide distribution, and many economic and pharmacological properties, being used from wood production to the afforestation of streets and avenues (Souza and Lorenzi 2012Souza, V.C.; Lorenzi, H. 2012. Botânica Sistemática - Guia ilustrado para identificação das famílias de angiospermas da flora brasileira, baseado em APG III. 3rd ed. Instituto Plantarum, Nova Odessa, 768p.). Pharmacological studies of extracts obtained from L. ferrea have demonstrated its biological anti-inflammatory (Carvalho et al. 1996Carvalho, J.C.T.; Teixeira, J.R.M.; Souza, P.J.C.; Bastos, J.K.; Filho, D.S.; Sarti, S. 1996. Preliminary studies of analgesic and anti-inflammatory properties of Caesalpinia ferrea crude extract. Journal of Ethnopharmacology, 53: 175-178.), bactericidal (Sampaio et al. 2009Sampaio, F.C.; Pereira, M.S.V.; Dias, C.S.; Costa, V.C.O.; Conde, N.C.O.; Buzalaf, M.A.R. 2009. In vitro antimicrobial activity of Caesalpinia ferrea Martius fruits against oral pathogens. Journal of Ethnopharmacology, 124: 289-294. ), hypoglycemic and antioxidant potentials (Batubara et al. 2009Batubara, I.; Mitsunanga, T.; Ohashi, H. 2009. Brazilian from Caesalpinia sappan wood as an antiacne agent. The Japan Wood Research Society, 56: 77-81.) and healing activity (Oliveira et al. 2010). Chemical studies with this species have shown the presence of phenolic compounds with possible pharmacological applications, such as chalcones and polyphenols (Nakamura et al. 2002Nakamura, E.S.; Kurosaki, F.; Arisawa, M.; Mukainaka, T.; Okuda, M.; Tokuda, H.; Nishino, H.; Junior, F.P. 2002. Cancer chemopreventive effects of constituents of Caesalpinia ferrea and related compounds. Cancer Letters, 177: 119-124. ; Nozaki et al. 2007Nozaki, H.; Hayashi, K.; Kido, M.; Akumoto, K.; Ikeda, S.; Matsuura, N. et al. 2007. Pauferrol A, a novel chalcone trimer with a cyclobutane ring from Caesalpinia ferrea mart exhibiting DNA topoisomerase II inhibition and apoptosis-inducing activity. Tetrahedron Letters, 48: 8290-8292. ).
Topical drug delivery offers numerous advantages for the treatment of CL. The hydrogels used in topical treatments have similar flexibility to natural tissues due to the high water content, which minimizes the irritation of the membranes or the tissues neighboring the application site (Koop 2012Koop, H.S. 2012. Hidrogéis binaries de Xantana e Galactomana com incorporação de curcumina visando uso tópico. Tese de Doutorado, Universidade Federal do Paraná. Curitiba, Paraná. 144p.), with a better patient compliance.
The objective of this study was to evaluate the chemical properties and antileishmanial activity of extracts obtained from L. ferrea against promastigotes and amastigotes of Leishmania that cause cutaneous leishmaniasis and to study the effect of topical treatment with hydrogels containing active extract in hamsters experimentally infected with L. (L.) amazonensis.
MATERIALS AND METHODS
Plant material
Samples of Libidibia ferrea (leaves, branches and fruits) were collected in Manaus, Amazonas, Brazil (3°4’34”S 60°0’11”W). A prepared voucher specimen was identified by Dr. Mike Hopkins, and deposited in the Herbarium of the National Institute of Amazonian Research (INPA) under the identification number 246150. Part of the plant material was dried in a forced circulation oven, with temperature below 45°C. After drying, the parts (leaves, branches and fruits without seeds) were pulverized and extracted with hexane and methanol solvent (VETEC®). Three extractions were done with each solvent using ultrasound bath for 20 min at each extraction. After ultrasound bath, the material was filtered, and concentrated on rotatory evaporator (Büchi®).
Phytochemical prospection
Crude extracts were submitted to a series of phytochemical characterization reactions to detect the presence of secondary metabolites as phenolic compounds (precipitation reaction with ferric chloride), flavonoids (magnesium metal in hydrochloric acid), triterpenes (Liebermann-Burchard reaction), alkaloids (Dragendorff) and characterization of saponins (foam index), according to the methodology described by Simões et al. (1999).
Determination of phenolics and flavonoids
For the dosage of total phenolics, extracts (1.0 mg ml-1) were dissolved in methanol or isopropanol (Prolabo®) and diluted in 5% triethanolamine and FeCl3 (0.165%). The reading of absorbance was performed on a spectrophotometer (510 nm), and the reactions were made in triplicate and compared with the standard curve of pyrogallol (Merk®). For the determination of total flavonoids, the extracts were dissolved in methanol or isopropanol and diluted in sodium acetate (0.1 M) and 10% AlCl3. The reading of absorbance was performed by spectrophotometer (415 nm) and reactions were made in triplicate and compared to standard curve of quercetin and naringenin (Merk®). The TPC (total phenolic content) and TFC (total flavonoid content) of the extracts was represented as milligrams of standard equivalents per gram of dry extract (mg pyrogallol or quercetin or naringenin / g extract), according to the calibration curve where a = linear coefficient of pyrogalol/quercetin/naringenin curve and b = slope of the curve).
Origin and maintenance of parasites
We used Leishmania (L.) amazonensis (MHO/BR/2006/IM5584), Leishmania (V.) guyanensis (MHO/BR/95/M4147) characterized, cryopreserved and kept in the Laboratory of Leishmaniasis and Chagas´s Disease, National Institute of Amazonian Research - INPA. The parasites were grown in Novy-MacNeal-Nicolle medium (NNN) and culture expanded in complete RPMI medium 1640 (Himedia), supplemented with 10% inactivated fetal calf serum (iFCS) and gentamycin (40 µg.mL-1) and cryopreserved in ultra-freezer at −80 °C. Murine macrophages (lineage J774) were maintained in complete (Himedia®) in culture bottles in an oven at 37°C.
In vitro promastigote assay
The assays were performed in 96 well plates using promastigotes in late log phase (106 cells.mL-1), according to Fumarola et al. (2004Fumarola L.; Spinelli, R.; Brandonisio, O. 2004. In vitro assays for evaluation of drug activity against Leishmania spp. Research in Microbiology, 155: 244-230.). The extracts were diluted in 3% v/v of dimethyl sulfoxide (DMSO, Vetec®) and Schneider’s Drosophila medium, filtered through 0.22 µm Millipore membranes in a sterile condition. Extracts of L. ferrea were evaluated at concentrations of 32 to 500 µg.mL-1. Controls of assay were pentamidine (Pentacarinat®) and 3% v/v of solvent DMSO was used as a negative control. The activity of the samples was evaluated by growth inhibition and mortality in periods of 24, 48 and 72 hours, at 25° C. Bioassays were made in triplicates and repeated twice. Parasite viability was quantified with hemocytometer.
Cell culture and cytotoxicity tests
Macrophages were placed in 24 mL of 105 cells.mL-1 of macrophage lineage J774 that were pre-cultured in 24-well polyethylene plates containing RPMI 1640 medium supplemented with 10% of iFCS for 24 h at 37°C, and after that they were placed in L. ferrea extracts. Incubation of macrophages was oven at 37°C for 48 h in the presence of the samples and MTT (3-[4,5-dimethyl-2-thiazolyl]-2,5-diphenyl-2H-tetrazolium/Roche®) reagent. Macrophages were incubated for another 12 h and then the colorimetric readings were made with a spectrophotometer (Bio-Tek®) using the wavelength of 590 nm.
Amastigote-macrophage assay
Culture of macrophage lineage J774 (105cells.mL-1) in 24-well plate was infected with a number of 106 promastigotes.mL-1 (1:10 cell/parasite) for four hours. Macrophages infected were incubated in the presence of L. ferrea extracts that showed higher antileishmanial potential against promastigotes of L. (V.) guyanensis and L. (L.) amazonensis and little or no cytotoxicity. After 48 h of incubation at 37°C, the wells were washed, glass cover slips were removed and the infected cells fixed and stained with Panotic (Laborclin®). We quantified by optical microscopy the number of infected macrophages (200 macrophages per slide/replicate), and the number of amastigotes per macrophage.
In vivo experiments
Origin and infection of animals: experimental animals used were 36 golden hamsters (Mesocricetus auratus), specific-pathogen-free, males and adults coming from and kept in the Experimental Care Center of INPA. Hamsters were housed in stainless steel cages in rooms conditioned with photoperiod and controlled temperature (22-24 °C) and humidity (50-60%) in feeding conditions ad libitum and free access to tap water. This study was approved under protocol number 009/2012 and carried out according to the regulations and recommendations of the Animal Research Ethics Committee - CEUA of INPA. The animals were separated into experimental groups and infected in the nose with 100 µL of L. (L.) amazonensis (106 cells.mL-1). The animals remained under supervision until the appearance of the lesions. Treatment with the hydrogel commenced 22 days after infection, and treatments were applied to the lesions once daily for 40 days, in a dose of 50 mg.day-1. Production of the formulations : hydrogel was prepared using macrogols (polyethylene glycols) in hydrophilic bases, in aqueous system as a cosolvent, and added to a base 10% of active extract FrMeOH, designated as GelFrMeOH. The hydrogel base with no extract was used as control placebo (GelPla). Experimental protocol: the animals were randomly allocated into four treatment groups: I - Infected untreated control group; II - Infected treated group with GelFrMeOH; III - Infected treated group with GelPla; IV - Infected treated group with Glucantime® intramuscular (IM) in a dose of 20 mg(Sb5).kg.day-1 .
Evaluation of treatment effects
Clinical evaluation: daily evaluation to determine total lesion volume using pachymeter (Zaas Precision®) to analyze the progression of the lesion. Lesions were also photo documented for morphological evolution. Euthanasia: The animals were euthanized with Euthanyle® (sodium pentobarbital - phenytoin sodium) in accordance with CEUA/INPA. Parasitological studies: lesions were obtained of groups in two different experimental times: i) from animals sacrificed at the end of treatment (40th day); ii) from animals sacrificed 20 days after treatment (60th day). A tissue sample sectioned from the lesion area of each animal was used for an imprint on glass slides. The slides were stained with Giemsa (Sigma), and the infectivity index was determined by multiplying the percentage of macrophages that had at least one intracellular parasite by the average number of intracellular parasites per infected macrophage (200 cells were examined/animal). Viability of parasites in culture medium: Tissue fragments of lesions were cultivated in NNN medium for eight days at 25 oC to observe the parasite viability. Parasite quantification by q-PCR: fragments of approximately 10 mg were transferred to microtubules (Eppendorf®) and then the DNA extraction procedure was performed using the Wizard® Genomic DNA Purification Kit (Promega), homogenized nuclear lysis solution, Proteinase K (20 mg / mL) and RNAse, according to the manufacturer’s instructions. For the q-PCR, reactions were prepared with a final volume of 25 μL with oligonucleotides at 100 nM and SYBR Green PCR Master Mix (Applied Biosystems). For the experiment, a standard curve was obtained. The thermal cycler used was the Rotor-Gene Q model (QIAGEM). Calculation of the absolute numbers of copies for samples of each assay was done taking into account the efficiency (E = 1.19248 (* = 10(-1/m) -1) of the reaction. Histopathological study: Tissue sections from skin lesions were used for histological study. The tissues were fixed with 10% buffered formalin and embedded in paraffin. Susequently, the sections were stained using hematoxylin and eosin (HE), and then they were dried overnight. The stained tissues were microscopically examined and the most detailed and clear tissue slides were chosen.
Statistical analysis
Statistical analysis was performed using the GraphPad Prism software (version 6.0 for Windows). The number of live cells was used to calculate by linear regression and to calculate the IC50 values (Inhibitory Concentration for 50% of the live parasite). A one-way ANOVA statistical test was used to assess the significance of the differences between the various groups followed by Tukey’s test to compare the means of different treatment groups, with 95% confidence to be considered significant.
RESULTS
Methanolic extracts from leaves, branches and fruits of Libidibia ferrea when revealed with ferric chloride showed strong reaction and well-marked bands, especially FrMeOH extract (Table 1). Dragendorff’s reagent was the only that showed no stain to extracts, indicating low concentration or absence of alkaloids (Table 1). The extract with the highest income was the FrMeOH, containing up to 40% yield (Table 2). The hexanic extracts, however, had very low income, where all parts extracted with hexane showed yield below 2% (Table 2). Methanolic extracts showed a high concentration of phenolic compounds. The most pronounced values of TPC were found in the methanolic extract from fruits without seeds (514.65 mg pyr/g dry extract) and leaves (494.20 mg pyr/g dry extract) (Table 2). Total flavonoid content (TFC) ranged from 5.67 to 69.35 mg quercetin/g of dry extract, and highest value was found in FrMeOH (Table 2).
Total phenolic content (TPC) and total flavonoid content (TFC) in extracts of leaves (Le), branch (Br) and fruits without seeds (Fr) from Libidibia ferrea obtained with hexane (Hex) or methanol (MeOH). Pyr = Pyrogallol; Quc = Quercetin; Nar = Naringenin. Values are means ± standard deviation.
Different parts of the same individual may have a large variation in the content and concentration of secondary metabolites, and thus may have a different biological activity. Table 3 shows the IC50 values calculated from results of extracts against Leishmania promastigotes. It was observed that the most promising extract was the methanolic extract of fruits (FrMeOH) with IC50 15.04 µg.mL-1 against L. (L.) amazonensis (Table 3). However, this extract showed low activity against L. (V.) guyanensis (173.11 µg.mL-1). The second extract with greater activity was the hexanic extract of the leaves (LeHex), which showed activity against L. (L.) amazonensis, with IC50 value of 53.09 µg.mL-1. Hexanic extracts from fruits and branches of L. ferrea showed no activity at the highest concentration tested for L. (L.) amazonensis. However, the LeHex extract showed low antileishmanial activity when evaluated against their potential L. (V.) guyanensis (Table 3). For L. (V.) guyanensis, inhibitory effects on promastigotes were observed for methanolic extract of the leaves (LeMeOH), with IC50 value of 129.42 µg.mL-1. EpMeOH exhibited an IC50 of 173.11 µg.mL-1 against promastigotes of L. (V.) guyanensis.
Anti-leishmanial activity in promastigotes of Leishmania spp. and IC50 values of Libidibia ferrea extracts of leaves (Le), branches (Br) and fruits without seeds (Fr) obtained with hexane (Hex) or methanol.
Among the most promising extracts (FrMeOH, LeMeOH and LeHex), FrMeOH extract from L. ferrea was less cytotoxic with IC50 above 500 µg.mL-1. Even when higher doses were used, such as 500 µg.mL-1 of FrMeOH extract, viable cells only decreased 18.13% (data not shown). For this reason this extract was selected for evaluation of antileishmanial potential in intracellular forms in infected macrophages. However, cytotoxicity in macrophages was observed when LeMeOH and LeHex were tested in 500 µg.mL-1. IC50 values of cytotoxicity for these extracts were 281.89 and 285.28 µg.mL-1.
Thus, the anti-amastigote effects of FrMeOH on in vitro infected-macrophages were tested. A screening showed that 500 µg.mL-1 of FrMeOH when incubated with amastigotes of L. (V.) guyanensis inhibited 62% of amastigote survival in comparison to controls. The IC50 for amastigotes of L. (V.) guyanensis was 330.36 µg.mL-1. Activity against amastigotes of L. (L.) amazonensis was observed in 500 µg.mL-1 of FrMeOH, with reduction of 54% of infected macrophages.
The parameters analyzed to evaluate the effects of treatments applied by topical administration on the lesions caused by L. (L.) amazonensis in hamsters allowed to follow the clinical evolution during the treatment, and the influence on parasites and tissues at the end of the 40-day treatment. The clinical evolution of lesion volumes is an important aspect to be evaluated, since cutaneous leishmaniasis (CL) can produce in the infected hamsters the development of histiocytic lesions, with broad tissue growth similar to benign lesions that develop in a chronic way to ulcers or nodular lesions. This experiment showed a 445.94% increase in the total lesion volume in the hamster noses of the control group compared to the first day of treatment.
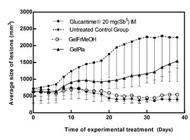
During the clinical analysis we observed progressive growth in the size of lesions. The group that received treatment with the pentavalent antimonial Glucantime® (20 mg (Sb5) kg.day-1) IM showed statistical differences with the negative control group (infected untreated control group) at the 14th day of treatment, with P = 0.0138. The statistical difference between the groups increased by the 20th day (P < 0.001). The animals continued to receive antimonial treatment until the 40th day of treatment, even though the reduction occurred after 20 days of treatment. At the end of treatment, a 43.42% reduction in lesion volume was observed in relation to the start of treatment (Figure 1).
Effect of topical treatment on lesion of golden hamsters (Mesocricetus auratus) infected with Leishmania (Leishmania) amazonensis, using 50 mg.day-1 of hydrogel with extract from Libidibia ferrea. Negative Control: animals infected and untreated. Positive control: animals treated with Glucantime® IM 20 mg(Sb5).kg.day-1. The data represent the mean ± SD.
The group receiving topical GelFrMeOH treatment showed constancy of the lesion volumes, not allowing the increase observed in the untreated group (Figure 1). There was a statistical difference between the lesion volumes of the GelFrMeOH group and the untreated group by the 14th day, with P = 0.0218. Statistical differences between the group treated with GelEpMeOH and the untreated group increased with continuity of treatment until reaching maximum differences in lesion size by the 24th day of treatment (P < 0.001). Animals treated with GelEpMeOH showed 42.78% reduction in lesion volume after 38 days of treatment compared to the first day of treatment (22th day post-infection).
However, the group that received topical gel excipients (GelPla) also showed significant difference between the means of lesion size when compared to the group at the 20th day of treatment (P = 0.0178). The mean values of the lesions of the GelPla group assume an intermediate position in the graph (Figure 1), showing difference between the control group and the groups treated with GelFrMeOH and with antimonial (Glucantime®). The difference between the GelFrMeOH group and the GelPla group occurred after the 30th day of treatment (P = 0.0411), and the difference increased with treatment time, reaching maximum difference on the last (40th) day of treatment (P = 0.0015). On the last day of treatment, mean lesions of the GelPla group corresponded to a 241% increase compared to day 0 of the treatment.
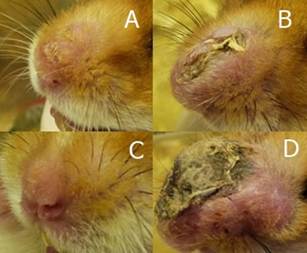
Lesions of the animals of the group that did not receive any treatment increased gradually during the experimental time, reaching indices superior to 400% of increase. The lesions of the group that received topical treatment with GelPla also increased, but in smaller proportions than in the control group, with an increase of 2.4 times its total volume. Lesions in the GelFrMeOH and Glucantime® treated groups reduced their total volume by more than 40%. There was presence of scar ulceration of varying diameter and magnitude in animals of control group (without treatment) as shown in Figure 2. Lesions of the group that received treatment with GelFrMeOH continued with its nodular aspect, or regression of the nodule, presenting 11% of animals with scar ulceration. The animals treated with GelPla had nodular lesions at the beginning, but 67% of the lesions presented ulceration. Animals treated with IM antimonial (Glucantime®) did not present ulceration until the end of treatment.
Clinical aspect of macroscopic lesions of Golden Hamster (Mesocricetus auratus) nose infected by Leishmania (Leishmania) amazonensis. Image of 30rd day of treatment. A - animal treated with 50 mg.day-1 of hydrogel with 10% of extract from Libidibia ferrea. B - animal treated with 50 mg.day-1 of hydrogel excipients. C - animal treated with Glucantime® IM 20 mg(Sb5).kg.day-1. D - Control Group: animal infected and untreated. This figure is in color in the electronic version.
Parasitological studies of lesions were obtained of groups in two different experimental times: at the end of treatment (40th day) and 20 days after treatment (60th day). The parasite viability verified by the NNN culture was semi-quantitative, and revealed the presence of higher scores of viable flagellate parasites in the cultures from fragments of the untreated group (score > 2). After 40th day, the lowest score was of the positive control (treatment with antimonial), with few viable parasites. Treatment groups GelFrMeOH and GelPla presented few viable parasites (score < 1) (Figure 3). On the 60th day the untreated group continued with high viability (score >3) of parasites, while viable parasites were present in the group treated with Glucantime®, GelFrMeOH and GelPla with score = 2.
Parasitological studies of material from lesions of golden hamster (Mesocricetus auratus) nose infected by Leishmania (Leishmania) amazonensis. A - at the end of treatment (40th day). B - 20 days after treatment (60th day). 1 - Parasite index counted by imprinting lesions fragments stained with Giemsa and counted in 1000 X. 2 - Parasite quantification by q-PCR. Control: animals inoculated but without topical treatment. The data represent the mean ± SD. *P < 0.05 when compared with all groups.
The evaluation of the parasite load by impression smears obtained after 40th day of treatment showed statistical differences between the GelFrMeOH and GelPla groups (P < 0.0001), the GelFrMeOH group and the untreated group (P < 0.0001). The group that received antimonial treatment (Glucantime®) also presented statistical difference compared with the negative control (P = 0.0086). Summarizing these results, the mean values visualized for the groups treated with GelFrMeOH, and Glucantime® showed values well below those found in their respective placebos and in the negative control. After 60 days of evaluation (20 days post treatment), only the GelFrMeOH group showed difference between GelPla (P = 0.005) and the group that received no treatment (P = 0.0004). We observed significant decrease in the parasite density in the group treated with GelFrMeOH when compared with their corresponding infected control group.
Parasite quantification by the real-time PCR (q-PCR) method, which is based on the detection of Leishmania DNA present in the lesions, showed that all the groups that received some treatment presented statistical differences in the concentration of parasite DNA when compared to the untreated group of samples obtained on the 40th day of treatment (Figure 3). The groups that received treatment with GelFrMeOH presented the lowest concentrations (<0.01), presenting P = 0.0276. GelPla also showed difference in relation to the untreated group (P = 0.0279). The group treated with pentavalent antimonial (Glucantime®) had values lower than 1.62 parasitic load and P = 0.027 compared to the untreated group. However, there was a change in the profiles of the parasite concentration in samples from the animals that remained for 20 days at the end of treatment (60th day). Mean values increased for the Glucantime® treated group (> 14,000) and decreased for the untreated group (287). The lowest values were found for the GelFrMeOH group (< 4.0) and the GelPla group (5.88). All groups presented statistical differences when compared to the group treated with Glucantime® (p < 0.05).
The different diagnoses for parasite load (Figure 3) seem to converge on the results of the parasitism reduction in the lesions of treated animals with gel containing the extract more efficiently than the animals treated with Glucantime®, mainly when comparing the results after 20 days of the end of the treatment.
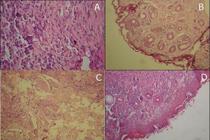
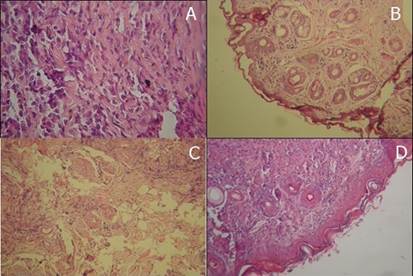
The dermal inflammatory pattern and cell population were estimated histologically in sections stained with HE. Pathologically, in the control group (untreated) the epidermis showed ulceration overlying connective tissue heavily infiltrated by inflammatory cells, predominantly composed of histocytes, and the most common lesion pattern was perifollicular dermatitis, mainly around the capillary isthmus (Figure 4A). The inflammatory infiltrate extended to the region of the sebaceous gland, obliterating it in some cases. Diffuse or perivascular infiltrate usually accompanied perifollicular inflammation. The intensity of the inflammatory infiltrate is shown in Figure 4A where the invasion at the border of the fibrous perichondrium of the nasal cartilage can be observed. Macrophages were predominant, with a discrete presence of lymphocytes and plasma cells. Isolated neutrophils were also observed in some laminae. In the animals treated with Glucantime® (positive control), the dermatitis was mild, papillary dermis, with a low mononuclear infiltrate, not extending to the hair follicles. Sebaceous adenitis was not observed in the analyzed samples (Figure 4B). The samples obtained from the animals of the GelFrMeOH group showed slight infiltration of inflammatory cells and absence of amastigote forms of parasites (Figure 4C), whereas in the GelPla group there was an intense and diffuse infiltration of inflammatory cells (Figure 4D). In the placebo group, we observed infiltration of epithelioid cells, lymphocytes and neutrophils in addition to plasma cells. In some areas, adenitis of the sebaceous gland and granulomatous area.
Light micrograph of nose skin of infected hamster (Mesocricetus auratus) with Leishmania (Leishmania) amazonensis showing infiltration of connective tissue by acute and chronic nonspecific inflammatory cells (H&E X400). A - Control Group: animal infected and untreated. B - animal treated with Glucantime® IM 20 mg(Sb5).kg.day-1. C - animal treated with 50 mg.day-1 of hydrogel with 10% of extract from Libidibia ferrea. D - animal treated with 50 mg.day-1 of hydrogel excipients. This figure is in color in the electronic version.
DISCUSSION
The search for new treatments that are less toxic and easily bioavailable to treat poor population, generally more affected by leishmaniasis, is imperative, and natural products are potential sources of a great variety of biological activity substitutes (Oliveira et al. 2013Oliveira, L.F.; Gilbert, B.; Villas-Boas, G.K. 2013. Potential for innovation in the treatment of leishmaniasis using plants and natural products as sources of new drugs. Revista Fitos, 8: 1-72. ). In this study, promising results obtained are similar to other ones from natural sources, as reported by Singh et al. (2012Singh, N.; Kumar1, M.; Singh, R. K. 2012. Leishmaniasis: Current status of available drugs and new potential drug targets Asian Pacific Journal of Tropical Medicine, 2012: 485-497.). Nevertheless, no natural-based therapy for the treatment of CL is currently advocated by health agencies in countries where leishmaniasis is endemic (Croft et al. 2006Croft, S.L.; Seifert, K.; Yardley, V. 2006. Current scenario of drug development for leishmaniasis. Indian Journal of Medical Research, 123: 399-410.).
Prospection of natural products present in Libidibia ferrea extracts showed that phenolic compounds are the most representative, corroborating the findings of Ueda et al. (2011). Phenolic compounds derived from vegetable products are recognized to have antioxidant activities that act as auxiliaries in the functioning of the endogenous immune system (Hakime-Silva et al. 2013Hakime-Silva, R.A.; Vellosa, J.C.; Khalil, N.M.; Khalil, O.A.; Brunetti, I.L.; Oliveira, O.M. 2013. Chemical, enzymatic and cellular antioxidant activity studies of Agaricus blazei Murrill. Annals of the Brazilian Academy of Sciences, 85: 1073 - 1081.). Among the chemical components described in the literature for the genus Libidibia the presence of polyphenols is highlighted. Another class of substances that have been characterized are terpenoids, which are usually extracted with low polarity solvents (Dias et al. 2013Dias, A.M.A.; Rey-Rico, A.; Oliveira, R.A.; Marceneiro, S.; Alvarez-Lorenzo, C.; Concheiro, A.; Júnior, R.N.C.; Braga, M.E.M.; Sousa, H.C. 2013. Wound dressings loaded with an anti-inflammatory jucá (Libidibia ferrea) extract using supercritical carbon dioxide technology. The Journal of Supercritical Fluids, 74: 34-45.).
Port’s (2011) noted in a study of nine species of plants, prepared as teas, that higher levels of phenolic compounds were present in L. ferrea. Gallic acid and ellagic acid were quantified by HPLC in extracts of the fruits of L. ferrea by Cunha et al. (2012Cunha, A.P.; Uchoa, A.F.J.; Almeida, R.R.; Ribeiro, A.C.B.; Brito, D.H.A.; Vieira, I.G.P.; Gomes, E.C.C.; Ricardo, N.M.P.S. 2012. Desenvolvimento de membranas de alginato e pectina associadas a bioativos vegetais para tratamento em queimaduras. 7o
Congresso Latino Americano de Órgãos Artificiais e Biomateriais. ( (www.metallum.com.br/7colaob/resumos/trabalhos_completos/01-215.docx
). Accessed on 26/02/2017.
www.metallum.com.br/7colaob/resumos/trab...
), indicating values above 2.49% of gallic acid and 5.89% of ellagic acid. These values can indicate a high level of phenolic compounds produced by L. ferrea even in individuals present in regions of different environmental conditions.
In the present study, we observed that this extract also showed high concentrations of phenolics and flavonoids, classes of metabolites already widely described and isolated from the genus Libidibia, as gallic acid and methyl-3,4,5-trihydroxybenzoate (Nakamura et al. 2002Nakamura, E.S.; Kurosaki, F.; Arisawa, M.; Mukainaka, T.; Okuda, M.; Tokuda, H.; Nishino, H.; Junior, F.P. 2002. Cancer chemopreventive effects of constituents of Caesalpinia ferrea and related compounds. Cancer Letters, 177: 119-124. ; Das et al. 2010Das, B.; Srinivas, Y.; Sudhakar, C.; Mahender, I.; Laxminarayana, K.; Reddy, P.R. et al. 2010. New diterpenoids from Caesalpinia species and their cytotoxic activity. Bioorganic & Medicinal Chemistry Letters, 20: 2847-2850.). From stem extract of L. ferra chalcone pauferrol A, a topoisomerase II inhibitor, was isolated (Nozaki et al. 2007Nozaki, H.; Hayashi, K.; Kido, M.; Akumoto, K.; Ikeda, S.; Matsuura, N. et al. 2007. Pauferrol A, a novel chalcone trimer with a cyclobutane ring from Caesalpinia ferrea mart exhibiting DNA topoisomerase II inhibition and apoptosis-inducing activity. Tetrahedron Letters, 48: 8290-8292. ). Different structures of metabolites of the phenolic class have already shown interaction in topoisomerase II of Leishmania species (Sen and Chattergee 2011Sen, R.; Chatterjee, M. 2011. Plant derived therapeutics for the treatment of Leishmaniasis. Phytomedicine, 18: 1056-1069. ), being this a possible mechanism of inhibitory action.
Leaves of Tephrosia cinerea (L.) Pers., a species of the Fabaceae family, showed IC50 of 43,6 µg.mL-1 against L. amazonensis in a study of activity from species used as medicinal plants, and it was one of great results of Bezerra et al. (2006Bezerra, J.L.; Costa, G.C.; Lopes, J.T.; Carvalho, I.C.D.S.; Patrício, F.J.; Sousa, S.M. et al. 2006. Avaliação da atividade leishmanicida in vitro de plantas medicinais. Revista Brasileira de Farmacognosia. 16: 631-637.). Previous studies have shown biological activities of the extracts of fruit and stem L. ferrea, such as anti-inflammatory (Carvalho et al. 1996Carvalho, J.C.T.; Teixeira, J.R.M.; Souza, P.J.C.; Bastos, J.K.; Filho, D.S.; Sarti, S. 1996. Preliminary studies of analgesic and anti-inflammatory properties of Caesalpinia ferrea crude extract. Journal of Ethnopharmacology, 53: 175-178.; Pereira et al. 2012Pereira, L.P.; Silva, R.O.; Bringel, P.H.S.F.; Silva, K.E.S.; Assreuy, A.M.S.; Pereira, M.G. 2012. Polysaccharide fractions of Caesalpinia ferrea pods: Potential anti-inflammatory usage. Journal of Ethnopharmacology 139: 642-648.), and antimicrobial, whereas fruit extract can inhibit in vitro growth of oral pathogens (Sampaio et al. 2009Sampaio, F.C.; Pereira, M.S.V.; Dias, C.S.; Costa, V.C.O.; Conde, N.C.O.; Buzalaf, M.A.R. 2009. In vitro antimicrobial activity of Caesalpinia ferrea Martius fruits against oral pathogens. Journal of Ethnopharmacology, 124: 289-294. ), and antioxidant (Silva et al. 2011Silva, L.C.N.; Silva-Junior, C.A.; Souza, R.M.; Macedo, A.J.; Silva, M.V.; Correia, M.T.S. 2011. Comparative analysis of the antioxidant and DNA protection capacities of Anadenanthera colubrina, Libidibia ferrea and Pityrocarpa moniliformis fruits. Food and Chemical Toxicology, 49: 2222-2228.), but this was the first screening with different extracts obtained from L. ferrea evaluated for Leishmania species. However, previous studies showed that the topical use of L. ferrea ointment was significantly efficient to help healing cutaneous wounds in goats (Oliveira et al. 2010Oliveira, A.F.; Batista, J.S.; Paiva, E.S.; Silva, A.E.; Farias, Y.J.M.D.; Damasceno, C.A.R.; Brito, P.D.; Queiroz, S.A.C.; Rodrigues, C.M.F.; Freitas, C.I.A. 2010. Avaliação da atividade cicatrizante do jucá (Caesalpinia ferrea Mart. ex Tul. var. ferrea) em lesões cutâneas de caprinos. Revista Brasileira de Plantas Medicinais, 12: 302-310.). Evaluating the chemical results together with the biological, the methanolic extract obtained from fruits without seeds is the most promising for future studies, with high extraction yield, high content of phenolics and flavonoids, activity against promastigotes of L. (V.) guyanensis and L. (L.) amazonensis and amastigotes of L. (V.) guyanensis and low cytotoxicity in J774 macrophages. The extract FrMeOH showed high concentrations of phenolics which have shown inhibitory activity on DNA synthesis, by interactions with topoisomerase II of Leishmania species (Sen and Chatterje 2011Sen, R.; Chatterjee, M. 2011. Plant derived therapeutics for the treatment of Leishmaniasis. Phytomedicine, 18: 1056-1069. ).
Studies that seek new therapeutic forms against cutaneous leishmaniasis evaluate several parameters to determine the efficacy of the compound and its use (Schwartz et al. 2014Schwartz, J.; Moreno, E.; Fernández, C.; Navarro-Blasco, I.; Nguewa, P.A.; Palop J.A.; Irache, J.M.; Sanmartín, C.; Espuelas, S. 2014. Topical treatment of L. major infected BALB/c mice with a novel diselenide chitosan hydrogel formulation. European Journal of Pharmaceutical Sciences, 62: 309 - 316. ). Comparison between the clinical results observed in animals that received treatment with hydrogel + 10% of methanolic extract of L. ferra and the untreated group showed strong evidence of the activity of the formulation on the development and evolution of the lesions, both by controlling the increase in the total volume of the lesion and other clinical aspects. In the groups that received the hydrogel, the lesions did not present an increase in their volume, with statistically significant differences when compared to the control group, and this evolution was similar to the group that received standard drug treatment (Glucantime®). In addition, the hydrogel with FrMeOH appears to have produced action in preventing the formation of ulcers in the lesions, according to the clinical evaluations. Promising results were found employing second choice drugs to the treatment of CL, as topical formulation with paromomycin sulfate, that was used topically twice a day for 4 weeks to treat L. major lesions on BALB/c mice, and the results showed a significantly smaller lesion size in the mice in the treated groups than in the mice in the control groups (Bavarsad et al. 2012Bavarsad, N.B.; Sedigheh, F.; Bazzaz, A.; Khamesipour, M.R. 2012. Colloidal in vitro and in vivo anti-leishmanial properties of transfersomes containing paromomycin sulfate in susceptible BALB/c mice. Acta Tropica, 124: 33 - 41.).
Several parameters were analyzed to allow a more accurate diagnosis of the parasite load present in the lesions of hamsters (M. auratus), both at the end of the treatment and after a period without receiving it. Summary results showed that there is a correlation between the different methods applied (tissue/histological aspect, detection of the presence of parasitic/molecular DNA and parasitological aspects), as it was detected in Swiss albino mice model infected with L. major (Eissa et al. 2011Eissa, M.M.; Amer, E.I.; Sawy, S.M.F. 2011. Leishmania major: Activity of tamoxifen against experimental cutaneous leishmaniasis. Experimental Parasitology, 128: 382-390.). Hydrogel treatment presented parasite load well below the other treatments, when the lesions were analyzed at the end of the treatment. However, there was an increase, although lower than in the negative control and also in the group treated with Glucantime®, regarding the presence of parasites after 20 days without receiving treatment. Hamsters treated with a topical formulation with pentamidine also showed viable parasites after end of treatment (Comandolli-Wyrepkowski et al. 2017Comandolli-Wyrepkowski, C.D.; Grafova, I.; Naiff, M.F.; Avella, M.; Gentile, G.; Grafov, A.; Franco, A.M.R. 2017. Topical treatment of experimental cutaneous leishmaniasis in golden hamster (Mesocricetus auratus) with formulations containing pentamidine. Acta Amazonica, 47: 39 - 46.).
The effect observed for the FrMeOH hydrogel can be explained by the penetration of the active principle and its diffusion rate, parameters that should be analyzed in future works. The question of penetration is one of the factors that determines efficacy in the treatment of tegumentary leishmaniasis, since the active principle must act in deeper tissues of the dermis, where many infected macrophages are located (Garnier et al. 2002Garnier, T.; Croft, S.L. 2002. Topical treatment for cutaneous leishmaniasis. Current Opinion in Investigational Drugs, 3: 538-544.). The other relevant question is the intrinsic antileishmanial activity of the sample (Schwartz et al. 2014Schwartz, J.; Moreno, E.; Fernández, C.; Navarro-Blasco, I.; Nguewa, P.A.; Palop J.A.; Irache, J.M.; Sanmartín, C.; Espuelas, S. 2014. Topical treatment of L. major infected BALB/c mice with a novel diselenide chitosan hydrogel formulation. European Journal of Pharmaceutical Sciences, 62: 309 - 316. ), which was observed for the FrMeOH extract in in vitro systems. In relation to hydrophobicity characteristics, it was added in hydrogel formulations due to its more hydrophilic character, increasing the interaction with the excipients, according to recommendations of the pharmaceutical technology (Nagelreiter et al. 2015Nagelreiter, C. Kratochvilova A.; Valenta, C. 2015. Dilution of semi-solid creams: Influence of various production parameters on rheological properties and skin penetration. International Journal of Pharmaceutics, 478: 429-438. ).
A study using chitosan hydrogel with the antibiotic paromomycin in the treatment of experimental CL also found promising results in lesions caused by L. (L.) amazonensis (Gonçalves et al. 2005Gonçalves, G.S.; Fernandes, A.P.; Souza, R.C.C.; Cardoso, J.E.; Oliveira-Silva, F.; Maciel, F.C. et al. 2005. Activity of a paromomycin hydrophilic formulation for topical treatment of infections by Leishmania (Leishmania) amazonensis and Leishmania (Viannia) braziliensis. Acta Tropica, 93: 161-167.). The hydrogels used in topical treatments have similar flexibility to natural tissues due to the high water content, which minimizes the irritation of the membranes or tissues neighboring the application site (Koop 2012Koop, H.S. 2012. Hidrogéis binaries de Xantana e Galactomana com incorporação de curcumina visando uso tópico. Tese de Doutorado, Universidade Federal do Paraná. Curitiba, Paraná. 144p.).
Topical therapies are more attractive options, offering reduced systemic toxicity and ease for the patient himself. However, when analyzing the therapeutic efficacy for the treatment of CL in a more comprehensive way, one should not only verify the elimination of the parasite, but also look in an integrated manner at the questions related to the aspects that involve the anti-inflammatory reactions and the healing capacity, thus promoting tissue repair and re-epithelialization, avoiding secondary infections (Schwartz et al. 2014Schwartz, J.; Moreno, E.; Fernández, C.; Navarro-Blasco, I.; Nguewa, P.A.; Palop J.A.; Irache, J.M.; Sanmartín, C.; Espuelas, S. 2014. Topical treatment of L. major infected BALB/c mice with a novel diselenide chitosan hydrogel formulation. European Journal of Pharmaceutical Sciences, 62: 309 - 316. ). Thus, this work allowed to verify the activity against the parasites and the effects on an experimentally infected host model, and to indicate a promising herbal treatment in the topical form of a hydrogel for the treatment of CL.
CONCLUSIONS
The methanolic extract of the fruits (FrMeOH) obtained from L. ferrea is the most promising, since it has in vitro activity against promastigotes and amastigotes of L. (L.) amazonensis and L. (V.) guyanensis and does not present cytotoxicity in macrophages J774. This extract is obtained with high values of yield of the vegetal material, besides having the highest values of phenolics between the extracts and high concentrations of flavonoids. The animals treated with the hydrogel containing the L. ferrea MeOH extract of fruits, after 40 days of treatment, presented reduction of lesion volume, decreased inflammation in the lesion region and lower parasitic load when compared to infected animals untreated, or that received only the gel without the extract.
ACKNOWLEDGMENTS
The authors would like to thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq, Fundação de Amaparo à Pesquisa do Estado do Amazonas - FAPEAM, and FP7-PEOPLE-2011-IRSES- 295262: VAIKUTUS for financial support, Master Leonardo Brandão from Biotério Central -INPA for assistance with animal care and Dr. Francimeire Gomes Pinheiro for technical assistance in the Laboratory of Leishmaniasis.
- Almeida, O.L.S.A.; Santos, J.B. 2011. Advances in the treatment of cutaneous leishmaniasis in the new world in the last ten years: a systematic literature review. Anais Brasileiros de Dermatologia, 86: 497-506.
- Alvar, J.; Vélez, I.D.; Bern, C.; Herrero, M.; Desjeux, P.; Cano, J.; Jannin, J.; DenBoer, M. 2012. Leishmaniasis worldwide and global estimates of its incidence. PLoS One, 9 e35671.
- Barreiro, E.; Bolzani, V.S. 2009. Biodiversidade: Fonte potencial para descoberta de fármacos. Química Nova, 32: 679-688.
- Batubara, I.; Mitsunanga, T.; Ohashi, H. 2009. Brazilian from Caesalpinia sappan wood as an antiacne agent. The Japan Wood Research Society, 56: 77-81.
- Bezerra, J.L.; Costa, G.C.; Lopes, J.T.; Carvalho, I.C.D.S.; Patrício, F.J.; Sousa, S.M. et al 2006. Avaliação da atividade leishmanicida in vitro de plantas medicinais. Revista Brasileira de Farmacognosia 16: 631-637.
- Carvalho, J.C.T.; Teixeira, J.R.M.; Souza, P.J.C.; Bastos, J.K.; Filho, D.S.; Sarti, S. 1996. Preliminary studies of analgesic and anti-inflammatory properties of Caesalpinia ferrea crude extract. Journal of Ethnopharmacology, 53: 175-178.
- Costa, L.M.J. 2005. Epidemiologia das Leishmanioses no Brasil. Gazeta médica da Bahia, 75: 3-17.
- Costa, L.M.; Guilhon-Simplicio, F.; Souza, T.P. 2015. Libidibia ferrea (Mart. ex Tul.) L.P. Queiroz var. ferrea: pharmacological, phytochemical and botanical aspects. International Journal of Pharmacy and Pharmaceutical Sciences, 4: 48-53.
- Croft, S.L.; Seifert, K.; Yardley, V. 2006. Current scenario of drug development for leishmaniasis. Indian Journal of Medical Research, 123: 399-410.
- Cunha, A.P.; Uchoa, A.F.J.; Almeida, R.R.; Ribeiro, A.C.B.; Brito, D.H.A.; Vieira, I.G.P.; Gomes, E.C.C.; Ricardo, N.M.P.S. 2012. Desenvolvimento de membranas de alginato e pectina associadas a bioativos vegetais para tratamento em queimaduras. 7o Congresso Latino Americano de Órgãos Artificiais e Biomateriais ( (www.metallum.com.br/7colaob/resumos/trabalhos_completos/01-215.docx ). Accessed on 26/02/2017.
» www.metallum.com.br/7colaob/resumos/trabalhos_completos/01-215.docx - Bavarsad, N.B.; Sedigheh, F.; Bazzaz, A.; Khamesipour, M.R. 2012. Colloidal in vitro and in vivo anti-leishmanial properties of transfersomes containing paromomycin sulfate in susceptible BALB/c mice. Acta Tropica, 124: 33 - 41.
- Comandolli-Wyrepkowski, C.D.; Grafova, I.; Naiff, M.F.; Avella, M.; Gentile, G.; Grafov, A.; Franco, A.M.R. 2017. Topical treatment of experimental cutaneous leishmaniasis in golden hamster (Mesocricetus auratus) with formulations containing pentamidine. Acta Amazonica, 47: 39 - 46.
- Das, B.; Srinivas, Y.; Sudhakar, C.; Mahender, I.; Laxminarayana, K.; Reddy, P.R. et al 2010. New diterpenoids from Caesalpinia species and their cytotoxic activity. Bioorganic & Medicinal Chemistry Letters, 20: 2847-2850.
- Dias, A.M.A.; Rey-Rico, A.; Oliveira, R.A.; Marceneiro, S.; Alvarez-Lorenzo, C.; Concheiro, A.; Júnior, R.N.C.; Braga, M.E.M.; Sousa, H.C. 2013. Wound dressings loaded with an anti-inflammatory jucá (Libidibia ferrea) extract using supercritical carbon dioxide technology. The Journal of Supercritical Fluids, 74: 34-45.
- Eissa, M.M.; Amer, E.I.; Sawy, S.M.F. 2011. Leishmania major: Activity of tamoxifen against experimental cutaneous leishmaniasis. Experimental Parasitology, 128: 382-390.
- Fumarola L.; Spinelli, R.; Brandonisio, O. 2004. In vitro assays for evaluation of drug activity against Leishmania spp. Research in Microbiology, 155: 244-230.
- Garnier, T.; Croft, S.L. 2002. Topical treatment for cutaneous leishmaniasis. Current Opinion in Investigational Drugs, 3: 538-544.
- Gonçalves, G.S.; Fernandes, A.P.; Souza, R.C.C.; Cardoso, J.E.; Oliveira-Silva, F.; Maciel, F.C. et al 2005. Activity of a paromomycin hydrophilic formulation for topical treatment of infections by Leishmania (Leishmania) amazonensis and Leishmania (Viannia) braziliensis Acta Tropica, 93: 161-167.
- Gontijo, B.; De Carvalho, M.L.R. 2003. Leishmaniose tegumentar americana. Journal of the Brazilian Society of Tropical Medicine 36: 71-80.
- Hakime-Silva, R.A.; Vellosa, J.C.; Khalil, N.M.; Khalil, O.A.; Brunetti, I.L.; Oliveira, O.M. 2013. Chemical, enzymatic and cellular antioxidant activity studies of Agaricus blazei Murrill. Annals of the Brazilian Academy of Sciences, 85: 1073 - 1081.
- Koop, H.S. 2012. Hidrogéis binaries de Xantana e Galactomana com incorporação de curcumina visando uso tópico Tese de Doutorado, Universidade Federal do Paraná. Curitiba, Paraná. 144p.
- Nakamura, E.S.; Kurosaki, F.; Arisawa, M.; Mukainaka, T.; Okuda, M.; Tokuda, H.; Nishino, H.; Junior, F.P. 2002. Cancer chemopreventive effects of constituents of Caesalpinia ferrea and related compounds. Cancer Letters, 177: 119-124.
- Nagelreiter, C. Kratochvilova A.; Valenta, C. 2015. Dilution of semi-solid creams: Influence of various production parameters on rheological properties and skin penetration. International Journal of Pharmaceutics, 478: 429-438.
- Nozaki, H.; Hayashi, K.; Kido, M.; Akumoto, K.; Ikeda, S.; Matsuura, N. et al 2007. Pauferrol A, a novel chalcone trimer with a cyclobutane ring from Caesalpinia ferrea mart exhibiting DNA topoisomerase II inhibition and apoptosis-inducing activity. Tetrahedron Letters, 48: 8290-8292.
- Oliveira, L.F.; Gilbert, B.; Villas-Boas, G.K. 2013. Potential for innovation in the treatment of leishmaniasis using plants and natural products as sources of new drugs. Revista Fitos, 8: 1-72.
- Oliveira, A.F.; Batista, J.S.; Paiva, E.S.; Silva, A.E.; Farias, Y.J.M.D.; Damasceno, C.A.R.; Brito, P.D.; Queiroz, S.A.C.; Rodrigues, C.M.F.; Freitas, C.I.A. 2010. Avaliação da atividade cicatrizante do jucá (Caesalpinia ferrea Mart. ex Tul. var. ferrea) em lesões cutâneas de caprinos. Revista Brasileira de Plantas Medicinais, 12: 302-310.
- Pace, D. 2014. Leishmaniasis. Journal of Infection, 69:10-18.
- Pereira, L.P.; Silva, R.O.; Bringel, P.H.S.F.; Silva, K.E.S.; Assreuy, A.M.S.; Pereira, M.G. 2012. Polysaccharide fractions of Caesalpinia ferrea pods: Potential anti-inflammatory usage. Journal of Ethnopharmacology 139: 642-648.
- Port’s, P.S.; Chisté, R.C.; Godoy, H.T.; Prado, M.A. 2013. The phenolic compounds and the antioxidant potential of infusion of herbs from the Brazilian Amazonian region. Food Research International, 53: 875-81.
- Sacks, D.; Kamhawi, S. 2001. Molecular aspects of parasite-vector and vector-host interactions in leishmaniasis. Annual Review of Microbiology, 55: 453-83.
- Sampaio, F.C.; Pereira, M.S.V.; Dias, C.S.; Costa, V.C.O.; Conde, N.C.O.; Buzalaf, M.A.R. 2009. In vitro antimicrobial activity of Caesalpinia ferrea Martius fruits against oral pathogens. Journal of Ethnopharmacology, 124: 289-294.
- Sen, R.; Chatterjee, M. 2011. Plant derived therapeutics for the treatment of Leishmaniasis. Phytomedicine, 18: 1056-1069.
- Silva, L.C.N.; Silva-Junior, C.A.; Souza, R.M.; Macedo, A.J.; Silva, M.V.; Correia, M.T.S. 2011. Comparative analysis of the antioxidant and DNA protection capacities of Anadenanthera colubrina, Libidibia ferrea and Pityrocarpa moniliformis fruits. Food and Chemical Toxicology, 49: 2222-2228.
- Schwartz, J.; Moreno, E.; Fernández, C.; Navarro-Blasco, I.; Nguewa, P.A.; Palop J.A.; Irache, J.M.; Sanmartín, C.; Espuelas, S. 2014. Topical treatment of L. major infected BALB/c mice with a novel diselenide chitosan hydrogel formulation. European Journal of Pharmaceutical Sciences, 62: 309 - 316.
- Silva-López, R.E. 2010. Proteases de Leishmania: novos alvos para o desenvolvimento racional de fármacos. Química Nova, 33: 1541 - 1548.
- Simões. C.M.O.; Schenkel, E.P.; Gosmann, G.; Mello, J.C.P. de; Mentz, L.A.; Petrovik, P.R. 1999. Farmacognosia: da planta ao medicamento 2da ed. Editora UFRGS, Porto Alegre, 821p.
- Singh, N.; Kumar1, M.; Singh, R. K. 2012. Leishmaniasis: Current status of available drugs and new potential drug targets Asian Pacific Journal of Tropical Medicine, 2012: 485-497.
- Souza, V.C.; Lorenzi, H. 2012. Botânica Sistemática - Guia ilustrado para identificação das famílias de angiospermas da flora brasileira, baseado em APG III 3rd ed. Instituto Plantarum, Nova Odessa, 768p.
- Ueda, H.; Tachibana, Y.; Moriyasu, M.; Kawanishi, K.; Alves, S.M. 2001. Aldose reductase inhibitors from the fruits of Caesalpinia ferrea Mart. Phytomedicine, 8: 377-381.
Publication Dates
-
Publication in this collection
Oct-Dec 2017
History
-
Received
22 Mar 2017 -
Accepted
23 Aug 2017