Abstracts
Calcium carbonate, a pharmaceutical excipient, is widely used as diluent in solid dosage forms. It is also used as a base for medicinal and dental preparations, a buffering and dissolution aid for dispersible tablets, a food additive and a calcium supplement. Egg shells are a rich source of mineral salts, mainly calcium carbonate, which corresponds to about 94% of the shell. Layer farms produce large amounts of shells, whose final disposal poses a challenge from the environmental standpoint. This work was designed to evaluate the physicochemical and thermal properties of calcium carbonate obtained from egg shells. The findings indicated that calcium carbonate from egg shells can be used as an alternative pharmaceutical excipient.
thermal analysis; X-ray powder diffraction; egg shells; calcium carbonate
O carbonato de cálcio é um excipiente farmacêutico amplamente utilizado como diluente em formas farmacêuticas sólidas. Também é utilizado para auxiliar na dissolução de comprimidos dispersíveis, como base para preparações dentais e medicinais, e como aditivos em suplementos alimentares. A casca de ovo é rica em sais minerais, principalmente o carbonato de cálcio que corresponde a 94% da casca. As indústrias de ovos produzem grande quantidade de casca e seu destino final é um desafio em termos ambientais. Este trabalho teve como objetivo avaliar as propriedades físico-químicas do carbonato de cálcio obtido a partir da casca de ovo. Os resultados demonstraram que o carbonato de cálcio obtido pode ser uma alternativa como excipiente farmacêutico.
análise térmica; difração de Raios-x; casca de ovo; carbonato de cálcio
Physicochemical study of CaCO3 from egg shells
Estudo físico-químico do carbonato de cálcio obtido a partir da casca de ovo
Fabio Seigi MurakamiI, * * A quem a correspondência deve ser enviada ; Patrik Oening RodriguesI, III; Célia Maria Teixeira de CamposII; Marcos Antônio Segatto SilvaI
ILaboratório de Controle de Qualidade, Universidade Federal de Santa Catarina UFSC, Campus Universitário Trindade, CEP 88040-900, Florianópolis - SC, Brasil, E-mail: fsmurakami@gmail.com
IILaboratório de Farmacotécnica, Departamento de Ciências Farmacêuticas, Universidade Federal de Santa Catarina UFSC, Florianópolis - SC, Brasil
IIIDepartamento de Farmácia, Universidade da Região de Joinville, Joinville - SC, Brasil
ABSTRACT
Calcium carbonate, a pharmaceutical excipient, is widely used as diluent in solid dosage forms. It is also used as a base for medicinal and dental preparations, a buffering and dissolution aid for dispersible tablets, a food additive and a calcium supplement. Egg shells are a rich source of mineral salts, mainly calcium carbonate, which corresponds to about 94% of the shell. Layer farms produce large amounts of shells, whose final disposal poses a challenge from the environmental standpoint. This work was designed to evaluate the physicochemical and thermal properties of calcium carbonate obtained from egg shells. The findings indicated that calcium carbonate from egg shells can be used as an alternative pharmaceutical excipient.
Keywords: thermal analysis; X-ray powder diffraction; egg shells; calcium carbonate.
RESUMO
O carbonato de cálcio é um excipiente farmacêutico amplamente utilizado como diluente em formas farmacêuticas sólidas. Também é utilizado para auxiliar na dissolução de comprimidos dispersíveis, como base para preparações dentais e medicinais, e como aditivos em suplementos alimentares. A casca de ovo é rica em sais minerais, principalmente o carbonato de cálcio que corresponde a 94% da casca. As indústrias de ovos produzem grande quantidade de casca e seu destino final é um desafio em termos ambientais. Este trabalho teve como objetivo avaliar as propriedades físico-químicas do carbonato de cálcio obtido a partir da casca de ovo. Os resultados demonstraram que o carbonato de cálcio obtido pode ser uma alternativa como excipiente farmacêutico.
Palavras-chave: análise térmica; difração de Raios-x; casca de ovo; carbonato de cálcio.
1 Introduction
Since ancient times fertile eggs, unfertilized eggs and chicken embryo eggs have been used by humanity as food and also in the treatment of diseases. We must take into account that eggs were not created for human being feed, but generating a new life, therefore, that is the reason why they are so complete and nutritive food4.
The egg shell has the mainly function to protect the chicken embryo acting as physical barrier for inhibiting microorganisms invasion and it also selects gaseous changes1,9.
According to Thapon and Bourgeois, 1994 shell is approximately 11% of the total weight of the egg and it presents contents of: calcium carbonate (94%), calcium phosphate (1%), magnesium carbonate (1%) and organic substances (4%). So, egg shell is a rich source of mineral salts, mainly calcium carbonate. Unfortunately, egg shell is an eggproduct industry residue.
Calcium carbonate, a pharmaceutical excipient, is mainly used as diluent in solid dosage forms. It is also used as a base for medicinal and dental preparations, buffering and dissolution aid in dispersible tablets, as well as food additive and calcium supplement3,6,14.
Nowadays, there is a great interest in finding new pure calcium carbonate sources6. Calcium carbonate obtained from bones flour, does not contain the same bioavailability of calcium obtained from synthetic sources. Calcium carbonate from oyster shells contains lead vestige among the others potential toxical elements such as aluminum, cadmium and mercury15. In the other hand, egg shell has an advantage for not containing toxical elements11.
Eggproducts industries produce great amount of shells and their final destiny is an environmental impact challenge. This work was designed to prepare and evaluate calcium carbonate from egg shell because it has a potencial application in the development of a new option of pharmaceutical excipient. The lack of information on these subjects suggests that there is a promising area to be explored.
In this context, the main objective of this study was to investigate the physical-chemical and thermal behaviour of calcium carbonate obtained from egg shell comparing to industrial calcium carbonate.
2 Experimental
2.1 Production of calcium carbonate from egg shell
The chicken egg shell, raw material (Gallus gallus domesticus, L.), Hy-line white eggs were obtained from Sohovos AB Brasil Sorocaba SP Brazil eggproduct industry. The calcium carbonate was produced after it had gone through stages of: wash, dry, grind and purification. The details on the treatment of the egg shells are suppressed because the Sohovos eggproduct industry is in process of intellectual property submission request.
2.2 Thermogravimetric analysis
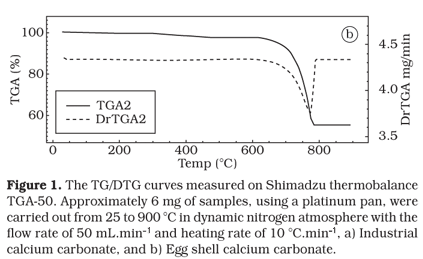
The TG/DTG experiments were measured on Shimadzu thermobalance TGA-50. Approximately 6 mg of samples, using a platinum pan, were carried out from 25 to 900 °C in dynamic nitrogen atmosphere with the flow rate of 50 mL.min1 and heating rate of 10 °C.min1.
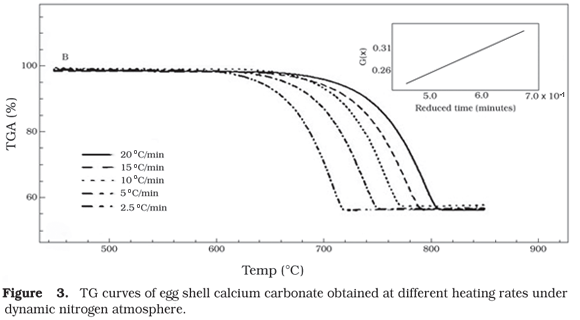
Non-isothermal kinetic study was performed by application of Ozawas method13. The kinetic study of both calcium carbonate, obtained from egg shell and industrial, were evaluated by thermogravimetric curves. The samples were heated at different heating rates (2.5, 5, 10, 15 and 20 °C.min1) to target temperature 900 °C under dynamic nitrogen atmosphere of 50 mL.min1. The equipment was preliminarily calibrated with standard reference of calcium oxalate.
2.3 X- ray powder diffraction (XRPD)
The XRPD patterns were recorded on an X-ray diffractometer (Siemens model D5000). The samples were irradiated with monochromatized Cuka radiation (1.54056 A°) and analyzed between 10-80° (2 theta). The voltage, current and pass time used were 40 kw, 40 mA and 1 s, respectively.
2.4 Morphology of particle
Microscopic observation of both calcium carbonates was performed under a microscope (Zeiss Axiostar Plus, Japan). The samples were mounted on a glass slide, viewed under normal light and pictures taken with a Zeiss MC80DX camera.
3 Results and discussion
The thermal analysis method was used, as well as X-ray powder diffraction and microscopy. These combining techniques provide quick and proper interpretations, offering the thermal stability, molecular organization, particle size and the possibility of analytical quantification of substances7,8,12. The properties of calcium carbonate obtained of the egg shell was compared with the industrial calcium carbonate, purchased from LABSYNTH, batch number 71148 USP-FCC.
The thermogravimetric curve TG/DTG obtained for industrial calcium carbonate (Figure 1a) showed a thermal stability up to 600 °C with a small mass loss (Dm = 1.8%) corresponding to volatile material. The decomposition process occurs in a single step in a defined way, between temperature range from 601 to 770 °C, with a Dm = 41.7% due to carbon dioxide release, given rise to calcium oxide: CaCO3 (s) ¬ heat ® CO2 (g) + CaO(s). The calcium carbonate usually decomposes at temperatures over 600 °C without fusion process10.
The TG/DTG curve obtained for egg shell calcium carbonate is seen in Figure 1b. The curve showed thermal stability up to 630 °C with a mass loss of Dm = 2.6% referring to volatile material. The thermal decomposition process occurs at the temperature range from 636 to 795 °C with mass loss Dm = 42.5%, which corresponds to carbon dioxide release, in a single step, in a defined way.
Through the thermogravimetric analysis it is possible to evidence that industrial calcium carbonate decomposes at temperature about 30 °C less than the calcium carbonate obtained from egg shell, with DTGpeak of 749.9 and 771.5 °C for industrial and egg shell product, respectively.
The kinetic data were calculated from plotting the mass loss vs. temperature of TG curves obtained to different heating rates for both calcium carbonates. Figures 2 and 3, demonstrate the superposition of TG curves obtained to industrial and egg shell calcium carbonate, respectively. The inserted figure demonstrates the linear tendency of reaction mechanism.
The activation energy (Ea) and Arrhenius frequency factor (Z) were calculated by Ozawas method for each five TG curves. The activation energy was obtained from a plot of logarithms of heating rates (A) as a function of the inverse of temperature (1/T) for a constant G(x), in which G(x) is the integrated form of the conversion dependence function, f(x). The Ea values were 47.83 kcal.mol1 ± 0.6% and 49.25 kcal.mol1 ± 0.2% for industrial and egg shell calcium carbonate, respectively. It was evidenced that the activation energy required for the decomposition process of egg shell product is higher than industrial, which pronounces its largest thermal stability. The Arrhenius frequency confirms the stability of egg shell calcium carbonate; the obtained results were 3.469 x 109 for industrial calcium carbonate and 5.011 x 109 for egg shell calcium carbonate. For both products the reaction of degradation was governed by a zero order kinetic.
In order to characterize the crystallinity patterns of industrial and egg shell calcium carbonate, XRPD analysis was performed for both samples. The diffractograms in Figure 4 indicate that diffraction patterns of industrial and egg shell calcium carbonate have similar peaks. The patterns can be put upon being the differences only found in the intensity of the peaks. The sharp peaks of both samples are characteristic of the high crystallinity of samples.
Table 1 shows the reflections, d-distances and relative intensities (I/I0) of the observed peaks in the patterns. The variation of samples reflections is small being larger in the relative intensity. Studies have been considering that the similarity of patterns is established if the scattering angles of the strongest reflections obtained from an sample agree to about ± 0.2 degrees with that of the reference material; and if the relative intensities of these reflections do not vary more than 20%2,5. The d-distance that considers the distance among the crystal plans is similar between carbonates, however, the values of the egg shell calcium carbonate is slightly smaller.
The morphology of particles was investigated by optical microscopy for both calcium carbonates. The photomicrographs of products are show in Figure 5. The images of industrial product (Figure 5a) and egg shell product (Figure 5b) indicate that these powders are preferably crystalline, and the size of the egg shell product is larger than industrial calcium carbonate crystals. The difference found between the crystals size can be related with the thermal stability of egg shell product, once the structure and size of crystals are directly correlated with the stability.
4 Conclusions
Through the thermogravimetric analysis it was evidenced that industrial calcium carbonate decomposes at a temperature of about 30 °C less than the calcium carbonate obtained from egg shell. The obtained kinetics parameters (Ea) and (Z) confirm that egg shell calcium carbonate is more stable comparing with the industrial. The XRPD analysis demonstrates that both products have similar cristallinity and the microscopy indicates that egg shell carbonate has largest particle size. The results showed that both products have similar physical chemical parameters, indicating that egg shell product should be an alternative of pharmaceutical excipient. Furthermore, studies have been accomplished about the influence of the egg shell calcium carbonate in the quality of pharmaceutical products like cosmetics.
Recebido para publicação em 14/11/2006
Aceito para publicação em 21/5/2007 (002062)
- 1. BARON, F.; GAUTIER, M.; BRULE, G. Factors involved in the inhibition of growth of Salmonella enteritidis in liquid egg white. J. Food Protect, v. 60, n. 11, p. 1318-1323, 1997.
- 2. BRITTAIN, H. G. Effects of pharmaceutical processing on drug polymorphs and solvates: In Polymorphism in Pharmaceutical Solids, Vol. II. New York: Marcel Dekker Inc., 1999, p. 227-278.
- 3. BROSTOW, W. et al. Synthesis of hydroxyapatite from eggshells. Mater. Lett, v. 41, n. 3, p. 128-134, 1999.
- 4. CAMPOS, C. M. T. et al. J. Composition of w-3 and w-6 fatty acids in freeze-dried chicken embryo eggs with different days of development. Braz. Arch. Biol. Tech, v. 47, n. 2, p. 219-224, 2004.
- 5. CARDOSO, T. M. et al. Physical-Chemical characterization and polymorphism determination of two Nimodipine samples deriving from distinct laboratories. Drug Dev. Ind. Pharm, v. 31, n. 7, p. 631-637, 2005.
- 6. DAENGPROK, W.; MINE, Y.; GARNJANAGOONCHORN W. Fermented pork sausage fortified with commercial or hen eggshell calcium lactate. Meat Sci, v. 62, n. 2, p. 199-204, 2000.
- 7. GIRON, D. Applications of thermal analysis and coupled techniques in pharmaceutical Industry. J. Therm. Anal. Cal, v. 68, n. 2, p. 335-357, 2002.
- 8. GIRON, D. Contribution of thermal methods and related techniques to the rational development of pharmaceuticals. PSTT, v. 1, n. 5, p. 191-199, 1998.
- 9. JAY, J. M. Modern food microbiology, 6th edition. Maryland: An Aspen Publication, 2000.
- 10. KIBBE, A. H.; WADE, A.; WELLER, P. J. Handbook of Pharmaceutical Excipients, 3th editon. London: Pharmaceutical Press, 2000.
- 11. MACNEIL, J. Composition of egg shell. Egg industry, v. 102, p. 15-17, 1997.
- 12. MEDEIROS, A. C. D. et al. Thermal stability of prednisone drug and tablets. J. Therm. Anal. Cal, v. 64, n. 2, p. 745-750, 2001.
- 13. OZAWA, T. Non-isothermal kinetics of consecutive reactions. J. Therm. Anal. Cal, v. 60, n. 3, p. 887-894, 2000.
- 14. SCHAAFSMA, A. et al. Mineral, amino acid, and hormonal composition of chicken eggshell powder and the evolution of its use in humam nutrition. Poultry Sci, v. 79, p. 1833-1838, 2000.
- 15. SCHAAFSMA, G. Bioavailability of calcium and magnesium. Eur. J. Clin. Nutr, v. 51, p. S13-S16, 1997.
- 16. THAPON, J. L.; BOURGEOIS, C. M. LOeuf et les ovoproduits. Paris : Lavousier Technique et Documentation, p. 344, 1994.
Publication Dates
-
Publication in this collection
29 Oct 2007 -
Date of issue
Sept 2007
History
-
Received
14 Nov 2006 -
Accepted
21 May 2007