Abstracts
Guidelines for volume replacement for acutely hemorrhaged and hemodiluted trauma patients have not been well established. Purpose: To evaluate the effects of acute hemodilution on mean arterial pressure (MAP), and responsiveness of acutely hemodiluted and subsequently hemorrhaged rats to different volume therapies. Methods: 180 rats were hemodiluted to simulate hemorrhaged trauma patients with persistent bleeding after high volume replacement with isotonic solutions. Thirty hemodiluted [Anemia (ANE) group] animals received no further treatment. The remaining 150 animals were subjected to hypovolemic shock and randomized into five groups, according to the treatment option employed: Control (CTL) animals did not receive subsequent treatment after hemorrhagic hypovolemia, SAL4 animals received isotonic saline 4 mL/kg, SAL32 animals received isotonic saline 32 mL/kg, HS animals received hypertonic saline 4 mL/kg and BLD animals received re-infusion of drawn blood. Results: Highest mean arterial pressure (MAP) was achieved by BLD, followed by SAL32 and HS. MAP after treatment of BLD, HS, SAL32 and ANE were higher than CTL (p=0.036). At 85 and 95 minutes of experiment, SAL4, SAL32 and HS presented the lowest hematocrit levels (p<0.01). At day 3, ANE, CTL and HS had the highest hematocrit. SAL4 and CTL groups presented the highest mortality rates. Conclusion: Hypertonic saline is an effective and safe initial therapy for hemodiluted rats undergoing hemorrhagic shock, with an overall outcome comparable to blood replacement or high volume isotonic saline administration.
Anemia; hypovolemic shock; hemodilution; volume replacement
A conduta para reposição volêmica em pacientes traumatizados, hemodiluídos não está bem estabelecida. Objetivo: Avaliar o efeito da hemodiluição sobre a pressão arterial média (PAM), bem como a resposta de ratos agudamente hemodiluídos e submetidos a choque hemorrágico, à reanimação com diferentes soluções. Métodos: 180 ratos foram hemodiluídos, simulando pacientes traumatizados com sangramento persistente após reposição com soluções isotônicas. Trinta ratos hemodiluídos (grupo ANE) foram observados, sem tratamento. Os demais 150 foram submetidos a choque hipovolêmico e randomizados em 5 grupos segundo a opção terapêutica: CTL (controle), sem tratamento subseqüente após hipovolemia hemorrágica; SAL4 que recebeu 4 mL/kg de salina isotônica; SAL32 que recebeu 32 mL/kg de salina isotônica ; SH que recebeu 4mL/kg de salina hipertônica; e BLD que recebeu reinfusão do sangue removido. Resultados: A pressão arterial média (PAM) mais alta ocorreu no grupo BLD, seguida pelo SAL32, e SH. A PAM após tratamento nos grupos BLD, SH, SAL32 e ANE foram maiores que no CTL (-=0.036). Aos 85 e 95 minutos do experimento, SAL4, SAL32, e SH apresentaram os menores hematócritos (p<0.01). No 3º dia, ANE, CTL e SH apresentaram os maiores hematócritos. SAL4 e CTL apresentaram as maiores taxas de mortalidade. Em conclusão, nossos resultados indicam que a SH é uma terapia inicial efetiva e segura em ratos hemodiluídos, submetidos a hemorrágico choque, com prognóstico comparável à reposição com sangue ou administração de grande volume de solução isotônica.
Anemia; choque hipovolêmico; hemodiluição; reposição volêmica
ORIGINAL ARTICLE
HYPOVOLEMIC SHOCK
Responsiveness to different volume therapies following hemodilution and hemorrhagic shock. A comparative experimental study in rats1 1 Research performed at Medical Research Laboratory (LIM-62), Hospital das Clínicas, School of Medicine, University of São Paulo (HC, SM-USP), Department of Thoracic Surgery of the Hospital A.C. Camargo (DTC-HACC), São Paulo, Brazil.
Resposta a diferentes terapias por reposição volêmica após hemodiluição e choque hemorrágico. Estudo experimental comparativo em ratos
Riad Naim YounesI; Fernanda DeutschII; Mario ItinosheIII; Belchor FontesIV; Renato PoggettiV; Dario BiroliniVI
IPhD, FACS Assistant surgeon, HC, SM-USP and DTC-HACC, São Paulo, Brazil
IIMD, Resident, Gynecology and Obstetrics, HC, SM-USP, São Paulo, Brazil
IIIBiomedical Researcher, LIM-62, HC, SM-USP, São Paulo, Brazil
IVPhD, Medical Supervisor, Emergency Surgery Service, HC, SM-USP, São Paulo, Brazil
VDirector, Emergency Surgery Service, HC, SM-USP, São Paulo, Brazil
VIFull Professor, Department of Surgery, Trauma Division, SM-USP, São Paulo, Brazil
Correspondence Correspondence: Belchor Fontes Rua Itacema, 246/11 04530-050 São Paulo - SP Brazil Phone: (55 11)8109-0044 / 3079-3048 belchor@uol.com.br
ABSTRACT
Guidelines for volume replacement for acutely hemorrhaged and hemodiluted trauma patients have not been well established. Purpose: To evaluate the effects of acute hemodilution on mean arterial pressure (MAP), and responsiveness of acutely hemodiluted and subsequently hemorrhaged rats to different volume therapies. Methods: 180 rats were hemodiluted to simulate hemorrhaged trauma patients with persistent bleeding after high volume replacement with isotonic solutions. Thirty hemodiluted [Anemia (ANE) group] animals received no further treatment. The remaining 150 animals were subjected to hypovolemic shock and randomized into five groups, according to the treatment option employed: Control (CTL) animals did not receive subsequent treatment after hemorrhagic hypovolemia, SAL4 animals received isotonic saline 4 mL/kg, SAL32 animals received isotonic saline 32 mL/kg, HS animals received hypertonic saline 4 mL/kg and BLD animals received re-infusion of drawn blood. Results: Highest mean arterial pressure (MAP) was achieved by BLD, followed by SAL32 and HS. MAP after treatment of BLD, HS, SAL32 and ANE were higher than CTL (p=0.036). At 85 and 95 minutes of experiment, SAL4, SAL32 and HS presented the lowest hematocrit levels (p<0.01). At day 3, ANE, CTL and HS had the highest hematocrit. SAL4 and CTL groups presented the highest mortality rates. Conclusion: Hypertonic saline is an effective and safe initial therapy for hemodiluted rats undergoing hemorrhagic shock, with an overall outcome comparable to blood replacement or high volume isotonic saline administration.
Key words: Anemia, hypovolemic shock, hemodilution, volume replacement.
RESUMO
A conduta para reposição volêmica em pacientes traumatizados, hemodiluídos não está bem estabelecida. Objetivo: Avaliar o efeito da hemodiluição sobre a pressão arterial média (PAM), bem como a resposta de ratos agudamente hemodiluídos e submetidos a choque hemorrágico, à reanimação com diferentes soluções. Métodos: 180 ratos foram hemodiluídos, simulando pacientes traumatizados com sangramento persistente após reposição com soluções isotônicas. Trinta ratos hemodiluídos (grupo ANE) foram observados, sem tratamento. Os demais 150 foram submetidos a choque hipovolêmico e randomizados em 5 grupos segundo a opção terapêutica: CTL (controle), sem tratamento subseqüente após hipovolemia hemorrágica; SAL4 que recebeu 4 mL/kg de salina isotônica; SAL32 que recebeu 32 mL/kg de salina isotônica ; SH que recebeu 4mL/kg de salina hipertônica; e BLD que recebeu reinfusão do sangue removido. Resultados: A pressão arterial média (PAM) mais alta ocorreu no grupo BLD, seguida pelo SAL32, e SH. A PAM após tratamento nos grupos BLD, SH, SAL32 e ANE foram maiores que no CTL (-=0.036). Aos 85 e 95 minutos do experimento, SAL4, SAL32, e SH apresentaram os menores hematócritos (p<0.01). No 3º dia, ANE, CTL e SH apresentaram os maiores hematócritos. SAL4 e CTL apresentaram as maiores taxas de mortalidade. Em conclusão, nossos resultados indicam que a SH é uma terapia inicial efetiva e segura em ratos hemodiluídos, submetidos a hemorrágico choque, com prognóstico comparável à reposição com sangue ou administração de grande volume de solução isotônica.
Palavras chaves: Anemia, choque hipovolêmico, hemodiluição, reposição volêmica.
Introduction
Trauma is the third cause of death in the world,1,2 and the leading cause of death in persons younger than 45 years.3 A significant number of trauma patients presents to the emergency room with hypovolemic shock. Infusion of large volumes (e.g, 32 mL/kg)4 of isotonic saline solution is considered standard therapy worldwide for hemorrhagic hypovolemia. However, there are special situations where availability and efficiency of large volumes of saline is limited. In the pre-hospital setting sufficient volumes of intravenous solutions are not readily available, especially in accidents with multiple trauma patients, and pre-hospital infused volume is usually insufficient. Due to the limited efficiency, different solutions have been evaluated for the past few years in order to provide better plasma volume expansion to hypovolemic patients. In the early 1980´s, a study showed that hypertonic saline (2400 mOsm) significantly improves hemodynamic parameters, even when infused at small volume and as the sole treatment.5 During the following years further studies successfully proved that hypertonic saline significantly increased blood pressure6 and cardiac output, and reversed hypovolemic shock without compromising the cardiopulmonary function or increasing the pulmonary extra-cellular volume.7 Furthermore, hyperosmotic solutions were efficiently used in the treatment of thoraco-abdominal aortic aneurysms, preventing declamping hypotension.8 On the other hand, it is also well known that plasma volume replacement with isotonic solutions may induce hemodilution, which is fairly well tolerated by normal patients. The immediate effects may depend on the duration and intensity of bleeding.´Patients frequently arrive at the Emergency Room acutely hemodiluted due to loss of large volume of blood, and extensive volume replacement with the infusion of isotonic solutions. The possible influence of this hemodilution state on the patients response to treatment or on patients outcome is not determined. Furthermore, continuous bleeding in these patients often requires further treatment. The ideal volume replacement in this situation is not established. The present study evaluates the effects of acute hemodilution on mean arterial pressure, as well as the responsiveness of acutely hemodiluted and subsequently hemorrhaged rats to different volume replacement therapies, including isotonic and hypertonic saline solutions, as well as whole blood replacement.
Methods
Wistar male adult rats (n=180, weight: 250-300 g) were used in this study. The rats were weighed and given intraperitoneal (IP) anesthesia: diazepam (5 mg/kg) and ketamine (40 mg/kg). Following anesthesia and antiseptic preparation, both jugular veins and the right carotid artery were catheterized, with the insertion of silicon catheters (Silastic 602-135). Catheters were filled with a heparin at 12.5 IU/mL 0.9% NaCl solution. The arterial catheter was connected to a transducer to continuously register mean arterial pressure (MAP) (Anamed Mainframe-am200a). The right jugular vein catheter was used for blood removal, and the left jugular vein catheter was used for the various infusions. After this step, 1000 IU of heparin were administered intravenously. Ten minutes were allowed after this procedure for stabilization of MAP. Then the first blood sample was removed for baseline hematocrit measurement (sample A) (Figure 1). To simulate the hemodilution state observed in trauma patients after resuscitation with large volume replacement with isotonic solutions for severe hemorrhagic shock, the hemodilution protocol was then performed, by shedding 0.2 mL of blood and infusion of 0.8 mL of 0.9% NaCl. This maneuver was performed over one minute period, and repeated fifteen times. Hematocrit was determined at 5 min (B), 10 min (C) and 15 min (D). At that point, the 180 rats were randomized (first randomization) into two groups: hemodilution anemia (ANE, n=30) group, and hemodilution followed by hypovolemic shock (SHOCK, n=150). All animals were observed for ten minutes after hemodilution and another blood sample was drawn (E). Following this observation period, in animals of SHOCK group, blood was shed gradually in order to reach a MAP of 50 mmHg that was continuously maintained till the end of a 30 min period by means of blood shedding or reinfusion as necesssary. Then, the rats of SHOCK group were further randomized to treatment groups (n=30 for each group): CTL, BLD, SAL4, SAL32, HS. After a 25 minutes of observation, shock treatment was started at 80 minutes of the experiment, and applied over 10 minutes to the different groups as follows (Figure 1): CTL group animals received no subsequent treatment, BLD group animals received the infusion of the blood shed during the shock induction and shock maintenance periods; SAL4 group animals received the infusion of 4 mL/kg 0.9% NaCl isotonic solution (NaCl) at 0.9%, SAL32 group animals received 32 mL/kg 0.9% NaCl; and HS group animals received 4 mL/kg of hypertonic 7.5% NaCl solution. Thereafter, all the animals of shock groups were observed till the end of the experiment, at 110 minutes, with sample collection for hematocrit at 85 (F), 95 (G), and 110 minutes (H). ANE group animals were not subjected to shock, received no treatment, and were observed till the end of the experiment, with sample collections for hematocrit at the same time points as in the shock groups. At the end of the experiment, all catheters were removed and jugular veins and right carotid artery ligated. All incisions were sutured and the animals were kept in individual cages. All animals (ANE an SHOCK groups) were observed for three days after completion of the study protocol. At the third post-operative day, the animals were anesthetized, a median laparotomy was performed and the aorta was punctured for another hematocrit (I). All animals were then sacrificed by inhaling saturated ethyl ether.
Statistical analysis
Baseline mean arterial pressure, the bled volume and the different hematocrit measurements were compared in the different groups, using the variance analysis test (ANOVA). The survival after the three-day period of observation was analyzed through the exact Fisher test. MAP recovery after the shock period (whether or not treated) was analyzed through the linear regression of the ascending MAP curve of each rat, and the tangent of the curve was considered as the individual coefficient.9 These coefficients were compared in the different groups, using the ANOVA. All differences were considered significant for p<0.05.
Results
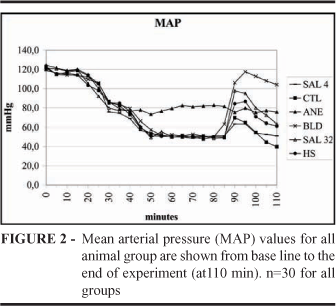
Baseline values for MAP (N=30 for all groups) was: CTL=123.0 ± 9.5; HS=120.9 ± 7.4; SAL4=118.9 ± 12.2; SAL32=121.6 ± 7.4; ANE=124.3 ± 11.5; BLD=122.3 ± 8.1. Statistical analysis showed no significant difference among the groups (p=0.345). All rats presented a similar decrease of MAP during the first 45 minutes of experiment. Thereafter, MAP of group ANE remained stable; MAP in all other groups showed a continuous trend to decrease till 55 minutes, and remained stable thereafter till 85 minutes. Fifteen minutes after the beginning of treatment, at 95 minutes of the experiment, MAP of all groups, except ANE, had significantly increased (p=0.027), being the highest MAP level achieved by the BLD group, followed by SAL32 and HS. MAP after treatment of BLD, ANE, HS and SAL32 groups were significantly higher than that of CTL group (p=0.036) (Figure 2).
Hematocrit of all groups (Figure 3) showed no significant difference at zero, 5, 10, 15, 25 minutes of experiment (p=0.611). At 85 minutes, SAL 4 and SAL32 presented the lowest hematocrit levels, followed by HS group (p<0.01). At 95 minutes of experiment, these three groups reached their lowest levels of hematocrit (p<0.01). On the other hand, at the end of experiment, BLD and ANE presented with the highest hematocrit levels (p<0.01). Hematocrit levels were analysed also on day 3 (Figure 3). BLD had the lowest hematocrit level, followed by SAL 32 group; ANE, CTL and HS groups had the highest hematocrit levels. There were significant differences between BLD and ANE groups (p=0.022).
Mortality
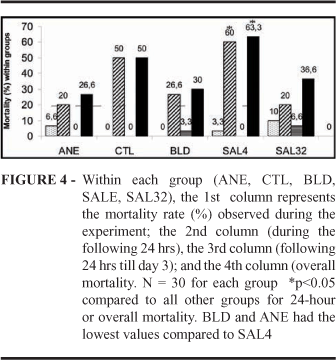
Animals in SAL4 group presented the highest 24-hour mortality rate (Figure 4) compared to all other groups (p=0.011), followed by the CTL group. The lowest mortality rates were found in ANE and BLD groups compared to SAL4 (p<0.01). Almost all deaths (68/72) occurred in the first 24 hours of the protocol. Only four (2.2%) of the rats surviving the first day of the study died subsequently during the following two days.
Discussion
Rapid removal from the trauma scene to a medical center has proved to be of the utmost importance for severely injured patients.10 Isotonic fluids are often administered to the patients in the pre-hospital setting. However, the volume infused is usually unable to compensate for all blood losses.10 The attempt to restore MAP with crystalloid solutions alone may worsen blood loss, decrease oxygen delivery, and increase mortality.11,12 The use of small volumes of hypertonic solutions has been proposed to minimize such problem and treat hypovolemic shock. Hypertonic saline solutions were shown to increase blood pressure, cardiac output, myocardial contractility and re-distribution of arterial blood flow.7,9 On the other hand, blood derivatives are rarely necessary or suitable as the initial fluid choice to restore MAP during resuscitation from hemorrhagic shock. Besides limited availability, they are associated with significant risks, and immediate as well as long-term complications.13 The results of the present study show that before treatment, MAP of the ANE group remained stable since the end of anemia induction till the end of the experiment. Burris et al found similar results.14 However, MAP presented a constant decrease in rats with no further treatment after 1 hour, leading to death.14 Our data also indicate that after the treatment with solutions infusion, MAP of all treatment groups increased. The highest peak was reached by BLD group, followed by SAL32 and HS. Meanwhile, CTL group animals presented a higher MAP increase than SAL4, and decreased thereafter to the end of the experiment. Previous clinical and experimental studies have compared hypertonic and isotonic fluid resuscitations for hypovolemic shock. Such studies showed that hypertonic solutions significantly increased MAP when compared to isotonic solutions.8.10,15-18 In all treated groups of the present study, the decreased MAP did not restore to initial levels. Several authors have demonstrated that restoring MAP may increase bleeding and worsen patients' conditions.11 On the other hand, Younes and Holcroft did not observe any major problems of re-bleeding after restoration of blood pressure in patients in the emergency room.10 However Krausz et al detected a drop in MAP after infusion of hypertonic solutions during the first 15 minutes of uncontrolled hemorrhagic shock.19 Moreover, other groups have not demonstrated differences in hemodynamic parameters between hypertonic and isotonic solutions.20 Our data indicated that hypertonic solutions, in comparison to isotonic solutions at small volumes, significantly increased MAP. As for blood resuscitation, Singh et al found similar results when comparing blood and isotonic saline solutions. Animals resuscitated with blood presented MAP significantly higher than those animals resuscitated with saline solutions.13 As for hematocrit, SAL4, SAL32 and HS groups presented the lowest levels during treatment. Meanwhile, BLD and ANE presented the highest levels at this moment. Similarly to our results, Singh et al and Marshall et al concluded that hematocrit levels in experimental groups that recovered from hypovolemic shock with blood were significantly higher than all groups of isotonic saline.11,13 Singh et al advocate that the benefit of increased oxygen-carrying capacity outweighed the risk of increased blood viscosity.13 However, Burris et al14 demonstrated that resuscitation with no fluids or with lactated Ringer´s solution presented the lowest hematocrit levels compared to hypertonic solutions and the hematocrit of the hypertonic solution groups decreased for the first hour and then stabilized. Guidelines for blood transfusion commonly used the "transfusion trigger" of hematocrit at 30%.21 Recent experimental studies have concluded that there is complete compensation of all organs to a hematocrit of 20%, incomplete compensation to a hematocrit ranging from 15%-20%, reversible compensation to 10%-15% and irreversible compensation to a hematocrit lower than 10%, the latter considered not compatible with survival.11,13 At day 3, hematocrit measurements in our study showed that ANE, CTL and HS presented the highest levels and BLD the lowest, followed by SAL32. All mean hematocrit levels were higher than 20%. SAL4 and CTL presented the highest mortality rates. ANE, BLD and HS were the groups with best survival rates. Most deaths occurred within 24 hours of procedure. Experimental studies demonstrated that some fluid resuscitation should be provided in early treatment to avoid early deaths.11,14 Comparable to the present results, Younes et al15 concluded in a prospective randomized study that survival rates were better for patients who received hypertonic solutions, with MAP at admission lower than 70 mmHg, than patients who received isotonic saline. However, Krausz19 found no significant differences in mortality rates between groups that received hypertonic saline after 30 minutes of injury or groups that received no treatment. Moreover, one experimental study showed no significant differences in survival rates between groups that received crystalloid alone or combined with blood.13 Another experimental study showed no significant differences in long-term survival between groups treated with hypertonic saline or crystalloid solutions. However, survival rates were significantly better in the treated groups, when compared to groups that received no further treatment.14 In summary, this study showed that hemodilution decreases mean arterial pressure in all experimental groups and in the abscence of hemorrhage or treatment (ANE group) was associated with an overal mortality of 26.6%. Whether one should restore baseline MAP or not, remains a controversial issue. When followed by hemorrhagic shock, in the abscence of treatment (CTL group), the overall mortality rate was 50%. When hemodilution was followed by hemorrhagic shock, the treatment with blood (BLD), hipertonic saline (HS) or high volume isotonic saline (SAL32) induced a sharply increased MAP, and an overall mortality rate of, respectively 30%, 33.3%, and 36.6%, was observed. The treatment with small volume isotonic saline (SAL4) resulted in the highest overall mortality rate (63.3%). Although presenting the lowest hematocrit level at day 3, the experimental group that received blood had a mortality rate of 30%
Conclusion
Hypertonic saline is an effective and safe initial therapy for hemodiluted rats undergoing hemorrhagic shock, with an overall outcome comparable to blood replacement or high volume isotonic saline administration.
Received: March 14, 2007
Review: May 15, 2007
Accepted: June 14, 2007
Conflict of interest: none
Financial source: none
- 1. Compton C, Rhee R. Peripheral vascular trauma. Perspect Vasc Surg Endovasc Ther. 2005;17:297-307.
- 2. Macho JR, Krupski WC, Lewis Jr FR. Management of the injured patient. In: Way LW, Dowerty GM. Current surgical diagnosis and treatment. 11ed. New York: McGraw-Hill; 2003. p 230-66.
- 3. Melniker LA, Leibner E, McKenney MG, Lopez P, Briggs WM, Mancuso CA. Randomized controlled clinical trial of point-of-care, limited ultrasonography for trauma in the emergency department: the first sonography outcomes assessment program trial. Ann Emerg Med. 2006;48(3):227-39.
- 4. Rocha-e-Silva M, Poli de Figueiredo LF. Small volume hypertonic resuscitation of circulatory shock. Clinics. 2005;60:159-72.
- 5. Velasco IT, Pontieri V, Rocha e Silva M, Lopes OU. Hyperosmotic NaCl and severe hemorrhagic shock. Am J Physiol. 1980;239:H664-73.
- 6. Nakayama S, Sibley L, Gunther RA, Holcroft JW, Kramer GC. Small volume resuscitation with hypertonic saline (2400 mOsm/litter) during hemorrhagic shock. Circ Shock 1984;13:149-59.
- 7. Layon J, Duncan D, Gallaghar TJ, Banner MJ. Hypertonic saline as a resuscitation solution in hemorrhagic shock: effect on extravascular lung water and cardiopulmonary function. Anesth Analg 1987;66:154-8.
- 8. Younes RN, Bechara MJ, Langer B, Aun F, Birolini D, Kusniek S, Guimarães PCM, Puech Leão LE. Prevention of aortic declamping shock with hypertonic 7.5% NaCl solution in abdominal aneurysmectomy of the aorta. Braz J Med Biol Res. 1986;19:141.
- 9. Younes RN, Rogatko A, Brennan MF. The hemodynamic response to hemorrhage in tumor-bearing animals. Surgery. 1991;110:508-13.
- 10. Younes RN, Aun F, Accioly CQ, Casale LP, Szajnbok I, Birolini D. Hypertonic solutions in the treatment of hypovolemic shock: a prospective, randomized study in patients admitted to the emergency room. Surgery. 1992;111:380-5.
- 11. Marshall Jr HP, Capone A, Courcoulas AP, Harbrecht BG, Billiar TR, Udekwu AO, Peitzman AB. Effects of hemodilution on long-term survival in an uncontrolled hemorrhagic shock model in rats. J Trauma. 1997;43:673.
- 12. Bickell WH, Bruttig SP, Millnamow GA, O'Benar J, Wade CE. The detrimental effects of intravenous crystalloid after aortotomy in swine. Surgery. 1991;110:529-36.
- 13. Singh G, Chaudry KI, Chaudry IH. Crystalloid is as effective as blood in the resuscitation of hemorrhagic shock. Ann Surg. 1992;215:377.
- 14. Burris D, Rhee P, Kaufmann C, Pikoulis E, Austin B, Eror A, Debraux S, Guzzi L, Leppaniemi. Controlled resuscitation for uncontrolled hemorrhagic shock. J Trauma. 1999;46:216.
- 15. Younes RN, Aun F, Ching CT, Goldenberg DC, Franco MH, Miura FK, Santos SS, Sequeiros IM, Silva MR, Fujimura I, Birolini D. Prognostic factors to predict outcome following the administration of hypertonic/hyperoncotic solution in hypovolemic patients. Shock. 1997;7:79.
- 16. Lopes OU, Velasco IT, Guetzenstein PG, Rocha e Silva M, Pontieri V. Hypertonic NaCl restores mean circulatory filling pressure in severely hypovolemic dogs. Hypertension 1986;8:195.
- 17. Younes RN, Aun F, Birolini D. Kawahara NT, Takeuti MM, Casali LL, Accioli CQ, Szajnbok I, Mori ND, Brito PL The initial treatment of hypovolemic patients: use of hypertonic 7.5% NaCl hypertonic solution. Rev Hosp Clin Fac Med São Paulo. 1988;43:138-4.
- 18. Vassar MJ, Perry CA, Holcroft JW. Prehospital resuscitation of hypotensive trauma patients with 7.5% NaCl vs. 7.5% NaCl with added dextran: a controlled trial. J Trauma. 1993;34:529.
- 19. Krausz MM, Landau EH, Klin B, Gross D. Hypertonic saline treatment of uncontrolled hemorrhagic shock at different periods from bleeding. Arch Surg. 1992;127:93.
- 20. Tobias TA, Schertel ER, Schmall LM, Wilbur N, Muir WW. Comparative effects of 7.5% NaCl in 6% dextran 70 and 0.9% NaCl on cardiorespiratory parameters after cardiac output-controlled resuscitation from canine hemorrhagic shock. Circ Shock. 1993;398:139.
- 21. Messmer KF. Acceptable hematocrit levels in surgical patients. World J Surg. 1987;11:41.
Publication Dates
-
Publication in this collection
27 Sept 2007 -
Date of issue
Oct 2007
History
-
Accepted
14 June 2007 -
Reviewed
15 May 2007 -
Received
14 Mar 2007