Abstract
PURPOSE:
To evaluate the alterations of two mitogen-activated protein kinases (MAPK)s, extracellular signal regulated kinase (ERK) and c-Jun NH2 terminal kinase (JNK), in the testes of male rats with experimental diabetes.
METHODS:
Twenty males Sprague-Dawley rats were randomly divided into a control group (n=8) and a diabetes group (administration of 40 mg/kg/day streptozotocin (STZ) for five sequential days, n=12). After six weeks, testicular biopsy samples were obtained for light microscopy and immunohistochemical methods.
RESULTS:
The PCNA (proliferating cell nuclear antigen) index was significantly decreased in the diabetes group (p=0.004) when compared to the control group. Both total (t)-ERK and phosphor (p)-ERK immunoreactivities were significantly decreased in the diabetes group (p=0.004, p<0.001, respectively). The t-JNK immunoreactivity was unchanged in both groups (p=0.125), while p-JNK immunoreactivity was significantly increased in the diabetic group (p=0.002).
CONCLUSIONS:
The decrease of androgen levels in the course of diabetes may contribute to the decrease of the immunoreactivities of t-ERK and p-ERK. JNK may be activated due to the changes in various cytokines and chemochines that participate in the oxidative stress process of diabetes. Therefore, testicular apoptosis may occur and lead to infertility associated with diabetes.
Diabetes mellitus; Mitogen-Activated Protein Kinases; Extracellular Signal-Regulated Kinases; JNK Mitogen-Activated Protein Kinases; Streptozocin; Testes; Rats
Introduction
Diabetes mellitus (DM) represents one of the greatest threats to health worldwide. The
growing incidence of DM will inevitably result in an increased prevalence in men of
reproductive age11. Alves MG, Martins AD, Rato L, Moreira PI, Socorro S, Oliveira PF.
Molecular mechanisms beyond glucose transport in diabetes-related male infertility.
Biochim Biophys Acta. 2013 May;1832(5):626-35. doi: 10.1016/j.bbadis.2013.01.011.
https://doi.org/10.1016/j.bbadis.2013.01...
. Male infertility is a common
complication of diabetes. The most frequent effects of DM on the male reproductive
system include decreased levels of testosterone, lower testicular weight, abnormal
spermatogenesis, alterations in sperm count and motility, and histopathological changes
in testis tissue components22. Koh PO. Streptozotocin-induced diabetes increases apoptosis through
JNK phosphorylation and Bax activation in rat testes. J Vet Med Sci. 2007
Sep;69(9):969-71. PMID: 17917385.
3. Cai L, Chen S, Evans T, Deng DX, Mukherjee K, Chakrabarti S.
Apoptotic germ-cell death and testicular damage in experimental diabetes: prevention
by endothelin antagonism. Urol Res. 2000 Oct;28(5):342-7.
PMID:11127715.
4. Aybek H, Aybek Z, Rota S, Sen N, Akbulut M. The effects of diabetes
mellitus, age, and vitamin E on testicular oxidative stress. Fertil Steril. 2008
Sep;90(3):755-60. PMID: 17482598.
-
55. Baccetti B, La Marca A, Piomboni P, Capitani S, Bruni E, Petraglia F,
De Leo V. Insulin-dependent diabetes in men is associated with hypothalamo-pituitary
derangement and with impairment in semen quality. Hum Reprod. 2002 Oct;17(10):2673-7.
PMID: 12351547.. Abnormalities in spermatogenesis due to
increased apoptosis have been demonstrated in certain studies22. Koh PO. Streptozotocin-induced diabetes increases apoptosis through
JNK phosphorylation and Bax activation in rat testes. J Vet Med Sci. 2007
Sep;69(9):969-71. PMID: 17917385.
,
33. Cai L, Chen S, Evans T, Deng DX, Mukherjee K, Chakrabarti S.
Apoptotic germ-cell death and testicular damage in experimental diabetes: prevention
by endothelin antagonism. Urol Res. 2000 Oct;28(5):342-7.
PMID:11127715.
,
66. Kyathanahalli C, Bangalore S, Hanumanthappa K, Muralidhara.
Experimental diabetes-induced testicular damage in prepubertal rats. J Diabetes. 2014
Jan;6(1):48-59. doi: 10.1111/1753-0407.12068.
https://doi.org/10.1111/1753-0407.12068...
7. Koh PO. Streptozotocin-induced diabetes increases the interaction of
Bad/Bcl-XL and decreases the binding of pBad/14-3-3 in rat testis. Life Sci. 2007 Sep
8;81(13):1079-84. PMID: 17870134.
8. Zhao Y, Tan Y, Dai J, Li B, Guo L, Cui J, Wang G, Shi X, Zhang X,
Mellen N, Li W, Cai L. Exacerbation of diabetes-induced testicular apoptosis by zinc
deficiency is most likely associated with oxidative stress, p38 MAPK activation, and
p53 activation in mice. Toxicol Lett. 2011 Jan 15;200(1-2):100-6. doi:
10.1016/j.toxlet.2010.11.001.
https://doi.org/10.1016/j.toxlet.2010.11...
9. Jiang X, Zhang C, Xin Y, Huang Z, Tan Y, Huang Y, Wang Y, Feng W, Li
X, Li W, Qu Y, Cai L. Protective effect of FGF21 on type 1 diabetes-induced
testicular apoptotic cell death probably via both mitochondrial- and endoplasmic
reticulum stress-dependent pathways in the mouse model. Toxicol Lett. 2013 May
10;219(1):65-76. doi: 10.1016/j.toxlet.2013.
https://doi.org/10.1016/j.toxlet.2013...
-
1010. Roy S, Metya SK, Rahaman N, Sannigrahi S, Ahmed F. Ferulic acid in
the treatment of post-diabetes testicular damage: relevance to the down regulation of
apoptosis correlates with antioxidant status via modulation of TGF-β1, IL-1β and Akt
signalling. Cell Biochem Funct. 2014 Jan;32(1):115-24. doi: 10.1002/cbf.2983.
https://doi.org/10.1002/cbf.2983...
. Testicular apoptosis may be the major
component of the infertility. Infertilityin young men with type 1 or type 2 diabetes is
showing a dramatic increase in recent years.
The nuclear matrix protein known as proliferating cell nuclear antigen (PCNA) is an indicator for DNA synthesis with its maximal release in the S and G2 phases of the cellular cycle; it cannot be detected in the G0 phase. PCNA immunoreactivity should be assessed to understand the effects of DM on spermatogenesis and proliferation1111. Altay B, Cetinkalp S, Doganavşargil B, Hekimgil M, Semerci B. Streptozotocin-induced diabetic effects on spermatogenesis with proliferative cell nuclear antigen immunostaining of adult rat testis. Fertil Steril. 2003 Sep;80 Suppl 2:828-31. PMID: 14505760..
Mitogen-activated protein kinases (MAPK's) regulate various physiological and
pathological cellular actions with their three subgroups; extracellular signal
regulatory kinase (ERK), c-Jun NH2 terminal kinase (JNK)/Stress Activated Protein Kinase
(SAPK), and p38 MAPK. ERK supports cellular proliferation by responding to epidermal
growth factors (EGFs) and other extrinsic mitogenic signals. JNK and p38 MAPK are
identified as "stress activated kinases" that are generally related to inflammation and
apoptosis1212. Kizilay G, Cakmak H, Yen CF, Atabekoglu C, Arici A, Kayisli UA.
Expression and regulation of c-Jun N-terminal kinase (JNK) in endometrial cells in
vivo and in vitro. Histochem Cell Biol. 2008 Oct;130(4):761-71. doi:
10.1007/s00418-008-0421-z.
https://doi.org/10.1007/s00418-008-0421-...
. They participate in the apoptotic
signal transduction as a response to various factors, including inflammatory cytokines,
ultraviolet lights, radiation, heat, hydrogen peroxide, and osmotic shock1313. Wada T, Penninger JM. Mitogen-activated protein kinases in apoptosis
regulation. Oncogene. 2004 Apr 12;23(16):2838-49. PMID: 15077147..
This study compared the immunoreactivities of ERK and JNK to evaluate the effects of DM on male germinal cells.
Methods
After the experimental procedures were approved by Trakya University Animal Experimentations Ethics Committee, twenty male Sprague Dawley rats (twelve-weeks-old, weighing 250 to 350g) were obtained from the Experimental Animal Center of Trakya University. Animals were divided into two groups: control group (n=8) and diabetes group (n=12). All of the subjects were kept in convenient laboratory conditions (temperature 22±1 ºC, 12 hours light/dark cycle), and maintained on a proper diet chow and water adlibitum.
Chemicals
Streptozotocin (STZ) was purchased from Sigma-Aldrich Corporation (St.Louis, MO, USA) dissolved in 0.1M citrate buffer, pH 4.2.
Experimental procedure
The blood glucose levels were checked in all animals before streptozotocin/citrate buffer treatment. Blood glucose levels were no differences in control and diabetes groups (88-111 mg/dl and 90-112 mg/dl, respectively). While control group subjects were administered only 0.1 mL citrate buffer, diabetes group subjects were administered 40mg/kg STZ during five consecutive days. Diabetes was confirmed by weekly measurement of blood glucose concentrations (above 250 mg/dl) with glucometer. After the six weeks of induction of diabetes, all testes tissues were totally removed under the anesthesia with alfamine (Ketamidor, Alfasan, Holland) and rompun (Bayer, Turkey), body and testicular weights were measured and processed for examinations with light microscopy and immunohistochemistry.
Testes tissue samples were fixed with 10% formaldehyde, washed, dehydrated and finally embedded in paraffin (Merck, Darmstadt, Germany). 5μm sections were stained with hematoxylin & eosin(H&E) to evaluate the histological structural features of testes. Using with same slides, mean seminiferous tubules diameters (MSTD) were determined with x200 magnification using ocular micrometer from the transversal sections of ten circular or circular-like random tubules44. Aybek H, Aybek Z, Rota S, Sen N, Akbulut M. The effects of diabetes mellitus, age, and vitamin E on testicular oxidative stress. Fertil Steril. 2008 Sep;90(3):755-60. PMID: 17482598. , 1111. Altay B, Cetinkalp S, Doganavşargil B, Hekimgil M, Semerci B. Streptozotocin-induced diabetic effects on spermatogenesis with proliferative cell nuclear antigen immunostaining of adult rat testis. Fertil Steril. 2003 Sep;80 Suppl 2:828-31. PMID: 14505760..
Immunohistochemistry
Formaldehyde-fixed, paraffin-embedded tissues were cut into 5μm sections. Slides were deparaffinized, rehydrated in a graded series of ethanol, and then boiled in citrate buffer (10mM; pH 6.0) for 20 min for antigen retrieval. Following washing in Tris-buffered saline (TBS), the sections were immersed in 3% hydrogen peroxide for 5 min. Slides were incubated with 5% normal horse serum (VectorLaboratories, Burlingame, CA, USA) for PCNA and with 5% normal goat serum (VectorLab.) for ERK and JNK in TBS for 30 min at room temperature in a humidified chamber. Excess serum was drained, and incubated in primary antibodies [monoclonal mouse, PCNA (F-2): sc-25280 (1:200 dilution in TBS; Santa Cruz BioTechnology, CA, USA); monoclonal rabbit p44/42 MAPK (ERK 1/2) and phospho-p44/42 MAPK (ERK 1/2) (Thr 202/Tyr 204) (1:100 dilution in TBS; Cell Signaling Technology-CST, Beverly, MA, USA), polyclonal rabbit SAPK/JNK and phospho-SAPK/JNK (Thr183/Tyr185) (1:100 dilution in TBS; CST), overnight at 4ºC in a humidified chamber. For negative controls, primary antibodies were replaced by their non-immune isotypes. Following washing three times for 5 min with TBS, sections were incubated with biotinylated horse anti-mouse and goat anti-rabbit antibodies (Vector Lab.) added at a 1:400 dilution for 30 min at room temperature. After washing three times with TBS, sections were incubated with a streptavidin-biotin-peroxidase kit (Vector Lab.) for an additional 30 min and the immunoreactivity was developed using DAB (3, 3-diaminobenzidine tetrahydrochloride dihydrate; Vector Lab.). Slides were slightly stained with hematoxylin and mounted with permount mounting medium (Fisher Chemicals, Spring Weld, NJ, USA).
In order to estimate the PCNA-index, PCNA-positive cells in seminiferous tubules 100 cell in three randomly chosen fields were counted in each slide. The cells with brown nuclear staining were considered positive. Both stained and nonstained germ cells were counted and the ratio of stained cells to the total number of germ cells, "PCNA index," was calculated for each seminiferous tubule. The average PCNA index in each case was obtained by dividing the sum of all PCNA indices by the number of seminiferous tubules in which the calculation was carried out1111. Altay B, Cetinkalp S, Doganavşargil B, Hekimgil M, Semerci B. Streptozotocin-induced diabetic effects on spermatogenesis with proliferative cell nuclear antigen immunostaining of adult rat testis. Fertil Steril. 2003 Sep;80 Suppl 2:828-31. PMID: 14505760..
Total (t)/phospho (p)-ERK and t/p-JNK were semiquantitatively evaluated using a
histological score (HSCORE) value, average score was used for statistical analysis,
described previosuly1212. Kizilay G, Cakmak H, Yen CF, Atabekoglu C, Arici A, Kayisli UA.
Expression and regulation of c-Jun N-terminal kinase (JNK) in endometrial cells in
vivo and in vitro. Histochem Cell Biol. 2008 Oct;130(4):761-71. doi:
10.1007/s00418-008-0421-z.
https://doi.org/10.1007/s00418-008-0421-...
.
Statistical analysis
S0064 Minitab Release 13 programme (License Number: WCP1331.00197) of Trakya University, Faculty of Medicine, Center of Information Technologies has been used for statistical analysis and values were stated as median ± standard deviation (SD). P values less than 0.005 were regarded as significant. Blood glucose levels (first, seventh and the last day), initial and final body weights, testis weights and the measurements of mean seminifer tubule diameters (MSTD) using with ocular micrometer in both groups were compared with Mann-Whitney U test.
The immunoreactivity data obtained by HSCORE method and PCNA index were analyzed by Mann-Whitney U test and p values less than 0.005 were regarded as significant while the differences between both groups were compared with Wilcoxon test.The differences of the immunoreactivity data in terms of total and phospho were compared as two dependent variables.
Results
In our study, blood glucoselevels were measured two days after the STZ administration and values over 250 mg/dl were accepted as diabetes. Blood glucose levels after STZ administration were significantly higher in the diabetes group than in controls (>250 mg/dl; p=0.001; Table 1).
At the beginning of the study, rats with higher body weights were placed in the diabetes group, although the body weights of the diabetes group were observed to decrease notably more than controls before they were sacrificed (p=0.001; Table 1).
Additionally, the weights of both testes were observed to decrease more in the diabetes group than the control group (p=0.001; Table 1).
Mean seminiferous tubuli diameter (MSTD) values of the diabetes group were significantly lower than controls (p=0.001; Table1).
Immunohistochemical findings
Testicular injury and spermatogenesis were evaluated histopathologically using H&E slides (Figure 1A and 1C). When we compared findings for the two groups, the diabetes group's testis sections reflected all expected findings such as decrease in MSTD, vacuolization, and degeneration in spermatogenetic series cells.In diabetic rats, atrophy of the seminiferous tubules with varying degrees of spermatogenetic arrest was also observed (Figure 1C).
Counting spermatogenic cells for each group revealed that PCNA-positive cells were detected at a higher level in the control group (Figure 1B). The number of PCNA-positive cells was lower in the diabetic group (Figure 1D). The PCNA indexwas significantly decreased in the diabetes group compared with the control group (p=0.004, Table 2; Figure 1D and 1B, respectively).
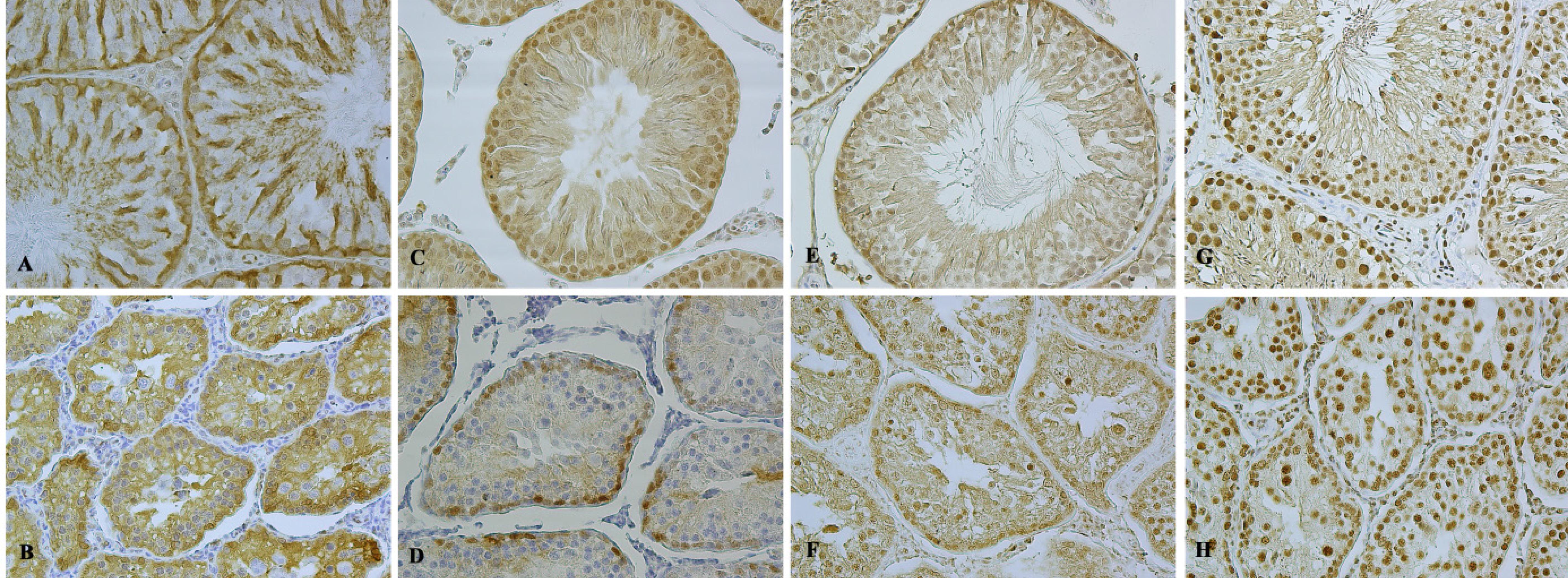
Regarding t-ERK immunoreactivity, the values were significantly lower in the diabetes group than in controls (p=0.004, Table2; Figure 2B and 2A, respectively), while the decrease in p-ERK immunoreactivity was even more significant between the control and diabetes groups (p<0.001, Table 2; Figure 2C and 2D, respectively).
Immunoreactivities in control and diabetic group for t/p-ERK and t/p-JNK. t-ERK and p-ERK immunoreactivity was both cytoplasmic and nuclear, with a higher intensity in control (A and C, respectively) compared to diabetic group (B and D, respectively). t-JNK immunreactivities were no differences in control (E) and diabetic group (F) but p-JNK immunoreactivities increased in diabetes group (H) than controls (G). Original magnification: x400.
While p-JNK immunoreactivity was higher in the diabetes group than in controls (p=0.002, Table 2; Figures 2H and 2G, respectively), the t-JNK immunoreactivity showed no change between the control and diabetes groups (p=0.125, Table2; Figures 2E and 2F, respectively).
Discussion
Diabetes mellitus affects almost all organs and systems with either micro- or macrovascular complications such as retinopathy, nephropathy, neuropathy, etc.44. Aybek H, Aybek Z, Rota S, Sen N, Akbulut M. The effects of diabetes mellitus, age, and vitamin E on testicular oxidative stress. Fertil Steril. 2008 Sep;90(3):755-60. PMID: 17482598.. The pathophysiology of these complications are well known but the effects of diabetes on the male reproductive system remains obscure.
Our study's body and testis weightfindings have been supported by several authors like Altay et al. 1111. Altay B, Cetinkalp S, Doganavşargil B, Hekimgil M, Semerci B. Streptozotocin-induced diabetic effects on spermatogenesis with proliferative cell nuclear antigen immunostaining of adult rat testis. Fertil Steril. 2003 Sep;80 Suppl 2:828-31. PMID: 14505760.. Aybek et al.44. Aybek H, Aybek Z, Rota S, Sen N, Akbulut M. The effects of diabetes mellitus, age, and vitamin E on testicular oxidative stress. Fertil Steril. 2008 Sep;90(3):755-60. PMID: 17482598. suggested that the body weight of diabetic men increased and Cai et al.33. Cai L, Chen S, Evans T, Deng DX, Mukherjee K, Chakrabarti S. Apoptotic germ-cell death and testicular damage in experimental diabetes: prevention by endothelin antagonism. Urol Res. 2000 Oct;28(5):342-7. PMID:11127715. suggested that testis weight did not show any change at one month of follow-up but decreased after six months of diabetes.
Our findings about MSTD are similar to other studies1111. Altay B, Cetinkalp S, Doganavşargil B, Hekimgil M, Semerci B. Streptozotocin-induced diabetic effects on spermatogenesis with proliferative cell nuclear antigen immunostaining of adult rat testis. Fertil Steril. 2003 Sep;80 Suppl 2:828-31. PMID: 14505760. , 1414. Trindade AA, Simões AC, Silva RJ, Macedo CS, Spadella CT. Long term evaluation of morphometric and ultrastructural changes of testes of alloxan-induced diabetic rats. Acta Cir Bras. 2013 Apr;28(4):256-65. PMID: 23568233..
Histopathological changes have been observed in the germinal epithelium and the interstitial tissue due to diabetes in testis tissues of diabetic animals. In our study, damage to the spermatogenic cell line and detachment, germ cell loss in certain tubules, vacuolization in Sertoli cells, multinuclear giant cells, decreased sperm concentration in the tubular lumen, tubular atrophy, and invaginations from the walls of the tubules have been observed.
Cai et al.33. Cai L, Chen S, Evans T, Deng DX, Mukherjee K, Chakrabarti S. Apoptotic germ-cell death and testicular damage in experimental diabetes: prevention by endothelin antagonism. Urol Res. 2000 Oct;28(5):342-7. PMID:11127715. have observed atrophy in the seminiferous tubules. Koh et al.77. Koh PO. Streptozotocin-induced diabetes increases the interaction of Bad/Bcl-XL and decreases the binding of pBad/14-3-3 in rat testis. Life Sci. 2007 Sep 8;81(13):1079-84. PMID: 17870134. have reported decrease in sperm counts, differentiation of Leydig cells, increase in the apoptosis of spermatogonium and spermatocytes, and abnormalities of the pituitary-testis axis. They suggested that decreases in testosterone levels, abnormal spermatogenesis, and germ cell apoptosis might be related to these changes.
In the diabetic group, the results of the PCNA index was in agreement with previous studies1111. Altay B, Cetinkalp S, Doganavşargil B, Hekimgil M, Semerci B. Streptozotocin-induced diabetic effects on spermatogenesis with proliferative cell nuclear antigen immunostaining of adult rat testis. Fertil Steril. 2003 Sep;80 Suppl 2:828-31. PMID: 14505760.. In our study, PCNA immunoreactivity was observed to be strong in the seminiferous tubules of the control group and especially spermatogonia, while moderate reactivity was observed in spermatocyte-I and spermatocyte-II. PCNA was nonreactive in Sertoli cells. In the diabetes group, moderate immunoreactivity was observed in a few germ cells and generally mild reactivity was observed. Altay et al.1111. Altay B, Cetinkalp S, Doganavşargil B, Hekimgil M, Semerci B. Streptozotocin-induced diabetic effects on spermatogenesis with proliferative cell nuclear antigen immunostaining of adult rat testis. Fertil Steril. 2003 Sep;80 Suppl 2:828-31. PMID: 14505760. reported similar results in their study conducted with type I diabetic rats in 2003.
Oxidative stress may develop with a decrease in insulin responsiveness, diabetes, and
cardiovascular diseases. Reactive oxygen species (ROS) production may increase and
insulin resistance may occur as a result of increased blood glucose and fatty acid
levels1515. Berdichevsky A, Guarente L, Bose A. Acute oxidative stress can
reverse insulin resistance by inactivation of cytoplasmic JNK. J Biol Chem. 2010 Jul
9;285(28):21581-9. doi: 10.1074/jbc.M109.093633.
https://doi.org/10.1074/jbc.M109.093633...
. There is a fine balance in the
reproductive system between ROS production and the antioxidant protective system, and
normally the level of ROS is quite low to regulate normal sperm functions44. Aybek H, Aybek Z, Rota S, Sen N, Akbulut M. The effects of diabetes
mellitus, age, and vitamin E on testicular oxidative stress. Fertil Steril. 2008
Sep;90(3):755-60. PMID: 17482598.. The increase in ROS levels results in testes and
sperm with oxidative damage and may lead to infertility. ROS levels have been observed
to decrease in 40% of the infertile couples44. Aybek H, Aybek Z, Rota S, Sen N, Akbulut M. The effects of diabetes
mellitus, age, and vitamin E on testicular oxidative stress. Fertil Steril. 2008
Sep;90(3):755-60. PMID: 17482598.
,
1616. Ranawat P, Bansal MP. Decreased glutathione levels potentiate the
apoptotic efficacy of selenium: possible involvement of p38 and JNK MAPKs--in vitro
studies. Mol Cell Biochem. 2008 Feb;309(1-2):21-32. PMID: 17989939.. The relation between increased ROS levels and
insulin resistance may be activated by some certain signal pathways. The DNA damage
caused by ROS accelerates apoptosis and shows disadvantageous effects by decreasing
sperm counts and results in infertility. The mRNA levels of stress-sensitive kinases
such as JNK, p38, MAPK, and kappa B kinase inhibitor increase with the proinflammatory
cytokines, including tumor necrosis factor- α (TNF-α), interleukin-6 (IL-6), and
monocyte chemoattractant protein (MCP-1) by means of oxidative stress and ROS
production. Systemic or local activation of TNF-α, IL-6, and MCP-1 induces the
production of ROS and a positive feedback mechanism takes place1616. Ranawat P, Bansal MP. Decreased glutathione levels potentiate the
apoptotic efficacy of selenium: possible involvement of p38 and JNK MAPKs--in vitro
studies. Mol Cell Biochem. 2008 Feb;309(1-2):21-32. PMID: 17989939..
The first member of the MAPK family that we have evaluated in our study is ERK, which is an important protein for tubule germinal epithelium proliferation. ERK is supposed to regulate the cellular proliferation by effecting the transcription of genes and phosphorylation of substrates. The significant decrease of p-ERK in diabetic subjects compared to t-ERK immunoreactivitymay be related to the decrease in germ cell proliferation. Moreover, the phosphorylation of ERK, which is produced by the interactions between spermatids and Sertoli cells, weakens communication between the adjacent proteins, thus facilitating the transport of mature sperm into the lumen1717. Cheng J, Watkins SC, Walker WH. Testosterone activates mitogen-activated protein kinase via Src kinase and the epidermal growth factor receptor in sertoli cells. Endocrinology. 2007 May;148(5):2066-74. PMID: 17272394.. The decreased number of sperm in the lumens of the diabetic testis samples may be related to the decreased levels of p-ERK caused by steroid hormones. The immunoreactivity evaluations of ERK, which is the member of the MAPK family responsible for cell proliferation, are consistent with our PCNA findings.
Zhang et al.1818. Zhang H, Yin Y, Wang G, Liu Z, Liu L, Sun F. Interleukin-6 disrupts
blood-testis barrier through inhibiting protein degradation or activating
phosphorylated ERK in Sertoli cells. Sci Rep. 2014 Mar 3;4:4260. doi:
10.1038/srep04260.
https://doi.org/10.1038/srep04260...
suggested that
IL-6 disrupts the blood-testis barrier (BTB) integrity and changes the normal level and
the regular localization of the BTB integral membrane proteins. It has also been shown
that IL-6 has a role in inhibiting the degradation of BTB constitutive proteins and
activating ERK-MAPK pathways. The results introduce a mechanistic understanding forthe
function of IL-6 in regulation of BTB dynamics.
The JNK pathway is activated by cellular stress conditions such as oxidative stress and
plays an important role in apoptosis, resistance to stress, and immune response.
Phosphorylation of this pathway within the nucleus may activate stress response and
apoptotic pathways. Acute oxidative stress leads to an increase of p-JNK within the
nucleus while chronic oxidative stress leads to an increase of p-JNK within the
cytoplasmic space. Oxidative stress and activated JNK may alter the expression of genes
associated with diabetes1515. Berdichevsky A, Guarente L, Bose A. Acute oxidative stress can
reverse insulin resistance by inactivation of cytoplasmic JNK. J Biol Chem. 2010 Jul
9;285(28):21581-9. doi: 10.1074/jbc.M109.093633.
https://doi.org/10.1074/jbc.M109.093633...
.
Koh suggested that western blotting analysis predicts a rise in p-JNK expression in testes of diabetic rats22. Koh PO. Streptozotocin-induced diabetes increases apoptosis through JNK phosphorylation and Bax activation in rat testes. J Vet Med Sci. 2007 Sep;69(9):969-71. PMID: 17917385.. In another study, Koh demonstrated hat there is a decrease in Bcl-2 and Bcl-XL levels and increase in Bax and Bad levelsin the testes of diabetic animals77. Koh PO. Streptozotocin-induced diabetes increases the interaction of Bad/Bcl-XL and decreases the binding of pBad/14-3-3 in rat testis. Life Sci. 2007 Sep 8;81(13):1079-84. PMID: 17870134. .
Prause et al.1919. Prause M, Christensen DP, Billestrup N, Mandrup-Poulsen T. JNK1
protects against glucolipotoxicity-mediated beta-cell apoptosis. PLoS One. 2014 Jan
24;9(1):e87067. doi: 10.1371/journal.pone.0087067.
https://doi.org/10.1371/journal.pone.008...
showed that JNK
functions as antiapoptotic in palmitate and high glucose-induced β-cell death. They also
concluded that ROS and endoplasmic reticulum stress are associated with higher
expression of proapoptotic Bcl-2 proteins in palmitate and high glucose-induced
oxidative stress. To sum up, they propose that JNK1 prevents palmitate and high
glucose-induced β-cell apoptosis, expression of puma, and formation of ROS.
Ranawat and Bansal suggested that a reduction of endogenous GSH as a result of selenite treatment in testicular cells is correlated with decreased Bcl-2 expression, increased apoptosis, increased expression of p38 and JNK MAPK, and an increase in caspase-3 expression1616. Ranawat P, Bansal MP. Decreased glutathione levels potentiate the apoptotic efficacy of selenium: possible involvement of p38 and JNK MAPKs--in vitro studies. Mol Cell Biochem. 2008 Feb;309(1-2):21-32. PMID: 17989939..
This is the first study that shows the level of JNK phosphorylation in diabetic male rats by immunohistochemistry. In our study, we observed that levels of t-JNK and f-JNK were noticeably higher in the diabetic group than in the control group and the immunoreactivity of t-JNK was also higher in the diabetic group,though it was statistically insignificant (p=0.125; Table 2), while f-JNK immunoreactivity was significantly increased in diabetic subjects (p=0.002; Table 2).
The JNK interference in cytokine-induced β-cell death was used to help understand the
differential biological functions of JNK subtypes. It was found that JNK3 has
antiapoptotic properties in cytokine-induced β-cell apoptosis. In addition, JNK 1 and
JNK2 were shown to have proapoptotic functions in response to cytokines1919. Prause M, Christensen DP, Billestrup N, Mandrup-Poulsen T. JNK1
protects against glucolipotoxicity-mediated beta-cell apoptosis. PLoS One. 2014 Jan
24;9(1):e87067. doi: 10.1371/journal.pone.0087067.
https://doi.org/10.1371/journal.pone.008...
.
Inflammatory issues play important roles in the pathogenesis of both type I and II
diabetes. TNF-α, IL-1β, certain cytokines within the IL-6 family, IL-8 and some
chemokines are active in the inflammatory process in both types of diabetes1919. Prause M, Christensen DP, Billestrup N, Mandrup-Poulsen T. JNK1
protects against glucolipotoxicity-mediated beta-cell apoptosis. PLoS One. 2014 Jan
24;9(1):e87067. doi: 10.1371/journal.pone.0087067.
https://doi.org/10.1371/journal.pone.008...
. The plasma levels of IL-6 are assumed to
increase the risk of diabetes with regard to insulin resistance, regardless of body
weight. Insulin resistancemay develop in healthy humans after the administration of high
doses of IL-6. TNF-α inhibits insulin secretion induced by glucosis, which may damage
the DNA of insulin and may induce β-cell apoptosis by means of Bcl-2. TNF-αhas been
demonstrated to be highly secreted in the adipose tissue and to play an important role
in the development of insulin resistance. Adipocytokines such as leptin, TNF-α, and IL-6
have been identified as increasing the risk of diabetes2020. Hajer GR, van Haeften TW, Visseren FL. Adipose tissue dysfunction in
obesity, diabetes, and vascular diseases. Eur Heart J. 2008 Dec;29(24):2959-71. doi:
10.1093/eurheartj/ehn387.
https://doi.org/10.1093/eurheartj/ehn387...
.
The increase of JNK levels in our diabetic subjects suggested that various cytokines such as TNF-α, IL-1β, and IL-6 and chemokines are released as a result of alterations in the expression of certain genes and proteins and therebyinduce apoptosis within the testis tissue. Furthermore, the decrease in the immunoreactivity of PCNA in the diabetes group indicated a lapse of proliferation in testis tissue while the increase in JNK may indicate the possible activation of the apoptotic pathway. The damage to the germ epithelium and the loss of cells may be attributed to these alterations.
Our findings may be utilized in the treatment of the patients with infertility due to diabetes; however, further and more advanced detailed studies are needed.
Conclusion
The decrease of androgen levels accompanying diabetes may contribute to the decrease of immunoreactivities of t-ERK and p-ERK. JNK may be activated by modification of the levels of certain genes and proteins by various cytokines and chemokines such as TNF-α, IL-1β, and IL-6 that play roles in the regulation of diabetes, the process of oxidative stress, and apoptosis resulting in infertility.
References
-
1Alves MG, Martins AD, Rato L, Moreira PI, Socorro S, Oliveira PF. Molecular mechanisms beyond glucose transport in diabetes-related male infertility. Biochim Biophys Acta. 2013 May;1832(5):626-35. doi: 10.1016/j.bbadis.2013.01.011.
» https://doi.org/10.1016/j.bbadis.2013.01.011 -
2Koh PO. Streptozotocin-induced diabetes increases apoptosis through JNK phosphorylation and Bax activation in rat testes. J Vet Med Sci. 2007 Sep;69(9):969-71. PMID: 17917385.
-
3Cai L, Chen S, Evans T, Deng DX, Mukherjee K, Chakrabarti S. Apoptotic germ-cell death and testicular damage in experimental diabetes: prevention by endothelin antagonism. Urol Res. 2000 Oct;28(5):342-7. PMID:11127715.
-
4Aybek H, Aybek Z, Rota S, Sen N, Akbulut M. The effects of diabetes mellitus, age, and vitamin E on testicular oxidative stress. Fertil Steril. 2008 Sep;90(3):755-60. PMID: 17482598.
-
5Baccetti B, La Marca A, Piomboni P, Capitani S, Bruni E, Petraglia F, De Leo V. Insulin-dependent diabetes in men is associated with hypothalamo-pituitary derangement and with impairment in semen quality. Hum Reprod. 2002 Oct;17(10):2673-7. PMID: 12351547.
-
6Kyathanahalli C, Bangalore S, Hanumanthappa K, Muralidhara. Experimental diabetes-induced testicular damage in prepubertal rats. J Diabetes. 2014 Jan;6(1):48-59. doi: 10.1111/1753-0407.12068.
» https://doi.org/10.1111/1753-0407.12068 -
7Koh PO. Streptozotocin-induced diabetes increases the interaction of Bad/Bcl-XL and decreases the binding of pBad/14-3-3 in rat testis. Life Sci. 2007 Sep 8;81(13):1079-84. PMID: 17870134.
-
8Zhao Y, Tan Y, Dai J, Li B, Guo L, Cui J, Wang G, Shi X, Zhang X, Mellen N, Li W, Cai L. Exacerbation of diabetes-induced testicular apoptosis by zinc deficiency is most likely associated with oxidative stress, p38 MAPK activation, and p53 activation in mice. Toxicol Lett. 2011 Jan 15;200(1-2):100-6. doi: 10.1016/j.toxlet.2010.11.001.
» https://doi.org/10.1016/j.toxlet.2010.11.001 -
9Jiang X, Zhang C, Xin Y, Huang Z, Tan Y, Huang Y, Wang Y, Feng W, Li X, Li W, Qu Y, Cai L. Protective effect of FGF21 on type 1 diabetes-induced testicular apoptotic cell death probably via both mitochondrial- and endoplasmic reticulum stress-dependent pathways in the mouse model. Toxicol Lett. 2013 May 10;219(1):65-76. doi: 10.1016/j.toxlet.2013.
» https://doi.org/10.1016/j.toxlet.2013 -
10Roy S, Metya SK, Rahaman N, Sannigrahi S, Ahmed F. Ferulic acid in the treatment of post-diabetes testicular damage: relevance to the down regulation of apoptosis correlates with antioxidant status via modulation of TGF-β1, IL-1β and Akt signalling. Cell Biochem Funct. 2014 Jan;32(1):115-24. doi: 10.1002/cbf.2983.
» https://doi.org/10.1002/cbf.2983 -
11Altay B, Cetinkalp S, Doganavşargil B, Hekimgil M, Semerci B. Streptozotocin-induced diabetic effects on spermatogenesis with proliferative cell nuclear antigen immunostaining of adult rat testis. Fertil Steril. 2003 Sep;80 Suppl 2:828-31. PMID: 14505760.
-
12Kizilay G, Cakmak H, Yen CF, Atabekoglu C, Arici A, Kayisli UA. Expression and regulation of c-Jun N-terminal kinase (JNK) in endometrial cells in vivo and in vitro. Histochem Cell Biol. 2008 Oct;130(4):761-71. doi: 10.1007/s00418-008-0421-z.
» https://doi.org/10.1007/s00418-008-0421-z -
13Wada T, Penninger JM. Mitogen-activated protein kinases in apoptosis regulation. Oncogene. 2004 Apr 12;23(16):2838-49. PMID: 15077147.
-
14Trindade AA, Simões AC, Silva RJ, Macedo CS, Spadella CT. Long term evaluation of morphometric and ultrastructural changes of testes of alloxan-induced diabetic rats. Acta Cir Bras. 2013 Apr;28(4):256-65. PMID: 23568233.
-
15Berdichevsky A, Guarente L, Bose A. Acute oxidative stress can reverse insulin resistance by inactivation of cytoplasmic JNK. J Biol Chem. 2010 Jul 9;285(28):21581-9. doi: 10.1074/jbc.M109.093633.
» https://doi.org/10.1074/jbc.M109.093633 -
16Ranawat P, Bansal MP. Decreased glutathione levels potentiate the apoptotic efficacy of selenium: possible involvement of p38 and JNK MAPKs--in vitro studies. Mol Cell Biochem. 2008 Feb;309(1-2):21-32. PMID: 17989939.
-
17Cheng J, Watkins SC, Walker WH. Testosterone activates mitogen-activated protein kinase via Src kinase and the epidermal growth factor receptor in sertoli cells. Endocrinology. 2007 May;148(5):2066-74. PMID: 17272394.
-
18Zhang H, Yin Y, Wang G, Liu Z, Liu L, Sun F. Interleukin-6 disrupts blood-testis barrier through inhibiting protein degradation or activating phosphorylated ERK in Sertoli cells. Sci Rep. 2014 Mar 3;4:4260. doi: 10.1038/srep04260.
» https://doi.org/10.1038/srep04260 -
19Prause M, Christensen DP, Billestrup N, Mandrup-Poulsen T. JNK1 protects against glucolipotoxicity-mediated beta-cell apoptosis. PLoS One. 2014 Jan 24;9(1):e87067. doi: 10.1371/journal.pone.0087067.
» https://doi.org/10.1371/journal.pone.0087067 -
20Hajer GR, van Haeften TW, Visseren FL. Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur Heart J. 2008 Dec;29(24):2959-71. doi: 10.1093/eurheartj/ehn387.
» https://doi.org/10.1093/eurheartj/ehn387
-
Financial source: Trakya University Research Center (Project nº: 2008/116)
-
1
Research performed at Department of Histology and Embryology, Faculty of Medicine, Trakya University, Edirne, Turkey.
Publication Dates
-
Publication in this collection
Oct 2014
History
-
Received
19 May 2014 -
Reviewed
18 July 2014 -
Accepted
22 Aug 2014