ABSTRACT:
Normally, it is not recommended the conditioning of gladiolus stems in water during storage or transport. Hydration of petals may accelerate flower opening, even at a low temperature, which compromises quality at marketing moment. However, for this species, neither the effect of prolonged dry cold storage nor its behavior when transferred to water at room temperature has been evaluated. The present study aimed to evaluate the vase life and the rehydration capacity of gladiolus flowers (Gladiolus grandiflora Hort.) after dry storage at low temperature. Flower stems of cultivars Blue Frost, Gold Field, Traderhorn, and Jester were dry-stored at a temperature of 5 ± 1 ºC and relative humidity of 85% for 12, 24, 36, and 48h. Control stems remained always in deionized water. After storage, they were returned to the water at room temperature and evaluated for vase life (adopting the discard criterion when 50% of the basal flowers displayed loss of color and wilting), fresh weight change (%), water uptake rate and transpiration rate, as well as relative water content of the petals (%). In dry cold storage conditions, for up to 36h, the vase life was not affected although incomplete rehydration of the flowers. Rehydration capacity of the stem is linked to the staggered opening of flowers along the inflorescence.
Key words:
Gladiolus grandiflora (Hort.); dry storage; cold storage; vase life; relative water content
RESUMO:
Normalmente, não é recomendado o condicionamento de hastes de gladíolo em água durante o armazenamento ou transporte. A hidratação das pétalas acelera a abertura das flores, mesmo em baixa temperatura, o que compromete a qualidade no momento da comercialização. No entanto, para essa espécie, ainda não foi estudado o efeito do armazenamento refrigerado prolongado a seco e nem o seu comportamento quando transferida para a água em temperatura ambiente. O presente estudo teve como objetivo avaliar a vida de vaso e a capacidade de reidratação de hastes florais de gladíolo após armazenamento a seco em baixa temperatura (Gladiolus grandiflora Hort.). As hastes das cultivares Blue Frost, Gold Field, Traderhorn e Jester foram armazenadas a seco em temperatura de 5 ± 1 ºC e umidade relativa de 85% por 12, 24, 36 e 48 horas. As hastes controle permaneceram sempre em água deionizada. Após armazenamento, retornaram a água sob condição de temperatura ambiente (22 ± 1 ºC) e foram avaliadas quanto à vida de vaso (adotando o critério de descarte quando 50% das flores basais apresentaram perda de coloração e murcha), variação da massa de matéria fresca (%), taxa de absorção de água e taxa transpiratória, bem como o teor relativo de água das pétalas (%). Em condições de armazenamento a seco em baixa temperatura, por até 36 horas, a vida de vaso não foi afetada, embora tenha ocorrido reidratação incompleta das flores. A capacidade de reidratação da haste está relacionada com a abertura escalonada das flores ao longo da inflorescência.
Palavras-chave:
Gladiolus grandiflora (Hort.); armazenamento a seco; armazenamento refrigerado; vida de vaso; teor relativo de água
INTRODUCTION:
The preservation form of postharvest ornamental quality of cut flowers is specific for each species. In general, producers seek to optimize water relations promoting the maintenance of tissue hydration. Loss of cell turgor, followed by flower senescence during the postharvest life, is the main factor of rejection by consumers (MEMON et al., 2012MEMON, N. et al. Membrane stability and postharvest keeping quality of cut Gladiolus flower spikes. Journal of Agricultural Technology, v.8, n.6, p.2065-2076, 2012. Available from: http://www.ijat-aatsea.com/pdf/v8_n6_12_November/18_IJAT_2012_8(6)_Memon,%20N_Plant%20Science.pdf>. Accessed: Feb. 10, 2015.
http://www.ijat-aatsea.com/pdf/v8_n6_12_...
).
During the transport and commercialization cut flowers are usually not conditioned in water, which may cause permanent wilting, preventing the recovery of tissue hydration when it is returned to the water (VIEIRA et al., 2011VIEIRA, L.M. et al. Water uptake by snapdragon inflorescences cuts after cold dry storage. Ciência Rural, v.41, n.3, p.418-423, 2011. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782011000300009>. Accessed: Jan. 20, 2014. doi: 10.1590/S0103-84782011000300009.
http://www.scielo.br/scielo.php?script=s...
). This can be a result of several factors, among which are the blockage and dehydration of xylem vessels, either by embolism or by development of microorganism (VAN DOORN et al., 2002Van DOORN, W.G. et al. Alkylethoxylate surfactants for rehydration of roses and Bouvardia flowers. Postharvest Biology and Technology , v.24, p.327-333, 2002. Available from: http://www.sciencedirect.com/science/article/pii/S0925521401001521>. Accessed: Feb. 20, 2015. doi: 10.1016/S0925-5214(01)00152-1.
http://www.sciencedirect.com/science/art...
; VIEIRA et al., 2012VIEIRA, L.M. et al. Vascular occlusion and water relations in cut snapdragon flowers. Acta Horticulturae , v.937, p.179-184, 2012. Available from: http://www.actahort.org/books/937/937_21.htm>. Accessed: Oct. 22, 2014.
http://www.actahort.org/books/937/937_21...
). Aiming to maintain the flower hydration, temperature control during storage or transport is among the main techniques utilized by producers, as it reduces the use of carbohydrates by respiration, slows the water loss, inhibits the development of pathogens, and reduces the production and action of ethylene (VIEIRA et al., 2011VIEIRA, L.M. et al. Water uptake by snapdragon inflorescences cuts after cold dry storage. Ciência Rural, v.41, n.3, p.418-423, 2011. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782011000300009>. Accessed: Jan. 20, 2014. doi: 10.1590/S0103-84782011000300009.
http://www.scielo.br/scielo.php?script=s...
).
Optimal storage temperature for cut flowers, in most cases, is based on the temperatures indicated for other species, though it is known that responses can vary even between cultivars (CEVALLOS & REID, 2001CEVALLOS, J.C.; REID, M.S. Effect of dry and wet storage at different temperatures on vase life of cut flowers. Hort Technology, v.11, p.199-202, 2001. Available from: http://horttech.ashspublications.org/content/11/2/199.full.pdf>. Accessed: Nov. 23, 2014.
http://horttech.ashspublications.org/con...
; MAPELI et al., 2011MAPELI, A.M. et al. Influence of storage temperature on Epidendrum ibaguense flowers. Acta Scientiarum. Agronomy, v.33, n.1, p.111-115, 2011. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1807-86212011000100016>. Accessed: Jan. 20, 2015. doi: 10.4025/actasciagron.v33i1.6365.
http://www.scielo.br/scielo.php?script=s...
). For instance, low temperatures are not recommended in the storage of tropical flowers, as they cause physiological damage (COSTA et al., 2010COSTA, A.S. et al. Characterization of symptoms of senescence and chilling injury on inflorescences of Heliconia bihai (L.) cv. Lobster Claw and cv. Halloween. Postharvest Biology and Technology, v.59, p.1-7, 2010. Available from: http://www.sciencedirect.com/science/article/pii/S0925521410001894>. Accessed: Jun. 25, 2015. doi: 1016/j.postharvbio.2010.08.015.
http://www.sciencedirect.com/science/art...
). In Alpinia sp., Strelitzia sp., and Heliconia sp., the recommended temperature without the development chilling-related injuries is between 10 and 13 ºC (FINGER et al., 2003FINGER, F.L. et al. Vase life of bird-of-paradise flowers influenced by "pulsing" and term of cold storage. Acta Horticulturae , v.628, n.1, p.863-867, 2003. Available from: http://www.actahort.org/books/628/628_110.htm>. Accessed: Mar. 10, 2015.
http://www.actahort.org/books/628/628_11...
; JAROENKIT & PAULL, 2003JAROENKIT, T.; PAULL, R.E. Postharvest handling of heliconia, red ginger and bird-of-paradise. HortTechnology , v.13, n.2, p.259-266, 2003. Available from: http://horttech.ashspublications.org/content/13/2/259.full.pdf>. Accessed: Mar. 10, 2015.
http://horttech.ashspublications.org/con...
; COSTA et al., 2010COSTA, A.S. et al. Characterization of symptoms of senescence and chilling injury on inflorescences of Heliconia bihai (L.) cv. Lobster Claw and cv. Halloween. Postharvest Biology and Technology, v.59, p.1-7, 2010. Available from: http://www.sciencedirect.com/science/article/pii/S0925521410001894>. Accessed: Jun. 25, 2015. doi: 1016/j.postharvbio.2010.08.015.
http://www.sciencedirect.com/science/art...
). According to CEVALLOS & REID (2001CEVALLOS, J.C.; REID, M.S. Effect of dry and wet storage at different temperatures on vase life of cut flowers. Hort Technology, v.11, p.199-202, 2001. Available from: http://horttech.ashspublications.org/content/11/2/199.full.pdf>. Accessed: Nov. 23, 2014.
http://horttech.ashspublications.org/con...
), the vase life of cut gillyflowers, narcissuses, irises, chrysanthemums, roses, and tulips is not changed when the plant is conditioned in water or dry, provided that the storage temperature is between 0 and 10 ºC.
Storage in water is not recommended for gladiolus stems, even at low temperatures, since the hydration of petals may accelerate flower opening, causing excess maturation, which compromises their quality at marketing (VEILING, 2009VEILING HOLAMABRA - FLORES E PLANTAS ORNAMENTAIS. Gladíolo. In: _____. Manual de classificação. 2009. Available from: <http://www.veiling.com.br/uploads/padrao_qualidade/criterios/gladiolo-fc.pdf>. Accessed: Sept. 28, 2014.
http://www.veiling.com.br/uploads/padrao...
). Good results in dry and cold storage depends on the knowledge of the ideal temperature and storage time (MACNISH et al., 2009MACNISH, A.J. et al. An alternative postharvest handling strategy for cut flowers - Dry handling after harvest. Acta Horticulturae , v.847, p.215-221, 2009. Available from: http://www.actahort.org/books/847/847_27.htm>. Accessed: Jan. 28, 2015.
http://www.actahort.org/books/847/847_27...
; AHMAD et al., 2012AHMAD, I. et al. Dry storage effects on postharvest performance of selected cut flowers. HortTechnology, v.22, p.463-469, 2012. Available from: http://horttech.ashspublications.org/content/22/4/463.full.pdf>. Accessed: Jan. 12, 2015.
http://horttech.ashspublications.org/con...
). According to REID (2004REID, M.S. Gladiolus. Recommendations for Maintaining Postharvest Quality. 2004. Available from: http://postharvest.ucdavis.edu/pfornamentals/Gladiolus/>. Accessed: Sept. 29, 2014.
http://postharvest.ucdavis.edu/pfornamen...
), for gladiolus, the recommended temperature is 5 ºC, as it is safe regarding to chilling. However, in gladiolus, no studies have investigated the effect of prolonged dry cold storage or its response when transferred to water at room temperature aiming to rehydrate flowers.
Hydration is practiced to restore the water balance of cut flowers transported and dry-stored to reestablish turgidity (SUZUKI et al., 2001SUZUKI, A. et al. Effects of retail hydration on water uptake and quality of 'Madame Delbard" roses after long term transport. Acta Horticulturae , v.543, p.251-256, 2001. Available from: http://www.actahort.org/books/543/543_30.htm>. Accessed: Jan. 28, 2015.
http://www.actahort.org/books/543/543_30...
; VIEIRA et al., 2011VIEIRA, L.M. et al. Water uptake by snapdragon inflorescences cuts after cold dry storage. Ciência Rural, v.41, n.3, p.418-423, 2011. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782011000300009>. Accessed: Jan. 20, 2014. doi: 10.1590/S0103-84782011000300009.
http://www.scielo.br/scielo.php?script=s...
; SANTOS et al., 2012SANTOS, J.S. et al. Capacity of rehydration and influence of cut on the recovery of water flow in inflorescences of Epidendrum ibaguense. Revista Brasileira de Horticultura Ornamental, v.18, n.1, p.15-21, 2012. Available from: http://ornamentalhorticulture.emnuvens.com.br/rbho/article/viewFile/689/499>. Accessed: Jan. 15, 2015. doi: 10.14295/rbho.v18i1.689.
http://ornamentalhorticulture.emnuvens.c...
; AHMAD et al., 2014AHMAD, I. et al. Optimal postharvest handling protocols for cut 'Line Dance' and 'Tap Dance' Eremurus inflorescences. Scientiae Horticulturae, v.179, p.212-220, 2014. Available from: http://www.sciencedirect.com/science/article/pii/S0304423814005093> Accessed: July 15, 2015. doi: 10.1016/j.scienta.2014.09.031.
http://www.sciencedirect.com/science/art...
). Water deficit occurs when the water uptake rate is lower than the flower's transpiration rate. A high transpiration rate coupled with limited absorption, due to the composition of the solution or due to high resistance and low hydraulic conductance of the xylem vessels, are the main factors negatively affecting the maintenance of postharvest quality of cut flowers (VIEIRA et al., 2012VIEIRA, L.M. et al. Vascular occlusion and water relations in cut snapdragon flowers. Acta Horticulturae , v.937, p.179-184, 2012. Available from: http://www.actahort.org/books/937/937_21.htm>. Accessed: Oct. 22, 2014.
http://www.actahort.org/books/937/937_21...
; SALEEM et al., 2014SALEEM, M. et al. Vase water effects on postharvest longevity and water relations of Gladiolus grandiflorus 'White Prosperity'. Pakistan Journal of Agricultural Sciences, v.51, n.1, p.137-141, 2014. Available from: http://pakjas.com.pk/papers%5C2252.pdf>. Accessed: Feb. 10, 2015.
http://pakjas.com.pk/papers%5C2252.pdf...
; COSTA et al., 2015COSTA, L.C. et al. Postharvest longevity of Heliconia wagneriana. Acta Horticulturae, v.1060, p.93-199, 2015. Available from: http://www.actahort.org/books/1060/1060_28.htm>. Accessed: Feb. 10, 2015.
http://www.actahort.org/books/1060/1060_...
).
The objective of the present study was to evaluate the vase life and the rehydration capacity of gladiolus flowers after dry storage at low temperature.
MATERIALS AND METHODS:
The experiment was conducted at the Universidade Federal de Viçosa, located in Viçosa-MG, Brazil (Latitude: 20º45'14" S, Longitude: 42º52'55" W, Altitude: 648 m). Stems of Gladiolus grandiflora (Hort.) cultivars Blue Frost, Gold Field, Green Star, and Jester were used. The commercial harvest point of the species was adopted; i.e., flowers were harvested when the flower buds showed the color of the cultivar. After the harvest, stems were placed in containers with water and then taken to the laboratory, where the base of the stem in water was cut and stems were standardized to a length of 70cm. Afterwards, flower stems were distributed at random across the treatments.
After the standardization, stems were wrapped in Kraft paper and packed in perforated plastic bags. Next, they were dry-stored in a cold room at 5 ºC in the vertical position at a temperature of 5 ± 1 ºC, relative humidity (RH) 85%, for 12, 24, and 48 h. Control stems remained in deionized water at all times. At the end of each period, inflorescences were returned to containers with 100mL water maintained at room temperature (± 22 ºC). After 0, 1, 2, 3, 4, 5, 6, 12, and 24h in water, the following variables were evaluated: fresh weight change, water uptake rate, transpiration rate, relative water content, and vase life of inflorescences (number of days between the onset of return to water and the appearance of wilting in at least 50% of the flowers).
Initial fresh weight was assigned the value of 100%. Fresh weight change was estimated as a percentage in relation to the initial weight of the stems, according to the following formula:
FWC = (FWST × 100)/IWST
FWC: fresh weight change, %;
FWST: final fresh weight of stem;
IWST: initial fresh weight of stem.
Water uptake rate was determined according to methodology described by VAN DOORN et al. (2002Van DOORN, W.G. et al. Alkylethoxylate surfactants for rehydration of roses and Bouvardia flowers. Postharvest Biology and Technology , v.24, p.327-333, 2002. Available from: http://www.sciencedirect.com/science/article/pii/S0925521401001521>. Accessed: Feb. 20, 2015. doi: 10.1016/S0925-5214(01)00152-1.
http://www.sciencedirect.com/science/art...
) and VIEIRA et al. (2012VIEIRA, L.M. et al. Vascular occlusion and water relations in cut snapdragon flowers. Acta Horticulturae , v.937, p.179-184, 2012. Available from: http://www.actahort.org/books/937/937_21.htm>. Accessed: Oct. 22, 2014.
http://www.actahort.org/books/937/937_21...
). After storage, stems were placed in individual tubes, initially weighed, containing 100mL deionized water. Tubes were weighed daily with and without the stems. To negate the effects of evaporation, the upper extremity of the tubes was coated with four layers of polyvinyl chloride (PVC) film. Water uptake rate of each solution was estimated as the volume of solution absorbed, in mg g−1 of fresh weight (FW), calculated by the following formula:
V = (IWSo - FWSo)/FWST
V: volume of solution absorbed;
IWSo: initial weight of solution;
FWSo: final weight of solution;
FWST: final fresh weight of stem.
The transpiration rate was estimated, in mg g−1 FW, by subtracting the stem fresh weight change from the volume of solution consumed, by the formula described below:
T=Vc-(FWST-IWST)
T: transpiration rate
Vc: volume of solution consumed;
FWST: final fresh weight of stem;
IWST: initial fresh weight of stem.
The relative water content (RWC) of the petals was determined according to the method described by CATSKY (1974CÁTSKY, J. Water content. In: SLAVIK, B. Methods of studying plant water relations. Berlin: Springer-Verlag, 1974. p.121-131.), with adaptations, at the end of the dry storage period and 4, 8, 12, 16, 20, and 24h after they were returned to water. Petals remained in the moistened foam until complete saturation for 24h; next, the material was weighed again to obtain the turgid weight. After the turgid weight was obtained, petals were dried in an oven at 70ºC for 72h to obtain the constant dry weight. The relative water content was calculated according to the following equation:
RWC=100 (FW-DW)/(TW-DW)
RWC: relative water content, expressed in %;
FW, DW, and TW: fresh weight, dry weight, and turgid weight, respectively.
Analysis of variance (Anova) with 1% significance, Tukey's test, and a descriptive statistics based on the mean values and standard error were performed.
RESULTS AND DISCUSSION:
There was no significant interaction between cultivars and dry storage time, or any isolated effect of cultivar, on any variable. There was an isolated effect of treatments on fresh weight change after 24h of rehydration and on vase life. Thus, the study continued using only the effect of treatments.
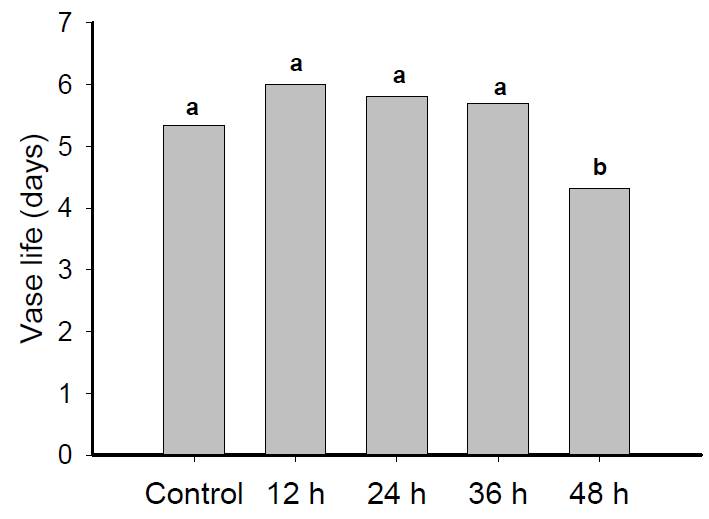
Gladiolus flower vase life was reduced in the treatment with 48h (Figure 1). The vase life of the inflorescences ranged from three to five days, totaling a difference of approximately 26% between control and the worst treatment (48h). Stems dry-stored for 36h at 5ºC showed little variation in vase life. It was thus demonstrated that vase life implies quality of gladiolus inflorescences, as it allows a longer time under marketing conditions, especially during transport. Contrasting the results found by SANTOS et al. (2012SANTOS, J.S. et al. Capacity of rehydration and influence of cut on the recovery of water flow in inflorescences of Epidendrum ibaguense. Revista Brasileira de Horticultura Ornamental, v.18, n.1, p.15-21, 2012. Available from: http://ornamentalhorticulture.emnuvens.com.br/rbho/article/viewFile/689/499>. Accessed: Jan. 15, 2015. doi: 10.14295/rbho.v18i1.689.
http://ornamentalhorticulture.emnuvens.c...
) with Epidendrum ibaguense , in which, irrespective of the dry storage time, there was a reduction in vase life of the inflorescences. In their study, dry storage of stems for only 12h was sufficient to reduce vase life. VIEIRA et al. (2011VIEIRA, L.M. et al. Water uptake by snapdragon inflorescences cuts after cold dry storage. Ciência Rural, v.41, n.3, p.418-423, 2011. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782011000300009>. Accessed: Jan. 20, 2014. doi: 10.1590/S0103-84782011000300009.
http://www.scielo.br/scielo.php?script=s...
); however, reported that cold and dry storage for two days retarded the senescence of cut inflorescences of snapdragon (Antirrhinum majus ) and prolonged the marketable period, provided that it was associated with the use of preservative solutions such as 8-HQC + sucrose + citric acid post-storage. In an experiment with dry storage and with water in several species, AHMAD et al. (2012AHMAD, I. et al. Dry storage effects on postharvest performance of selected cut flowers. HortTechnology, v.22, p.463-469, 2012. Available from: http://horttech.ashspublications.org/content/22/4/463.full.pdf>. Accessed: Jan. 12, 2015.
http://horttech.ashspublications.org/con...
) observed that cut roses of cultivar Angelique had a longer vase life when dry-stored for two weeks than those stored in water for the same period.
Vase life of gladiolus flowers subjected to dry storage for 0, 12, 24, 36, and 48h at 5°C. Lines represent mean values of cultivars Blue Frost, Gold Field, Green Star, and Jester in each treatment. Different letters indicate significant differences by Tukey's test (P<0.05).
As shown in figure 2A, there was a decrease in stem fresh weight during storage when compared with control. This reduction was proportional to the storage time. All flower stems gained weight in the first 12h, which remained constant for 24h. Differently, plants from control treatments increased in weight in the first 6h, with little variation after the 12h, maintaining fresh weight below that obtained in the treatments with 12, 24, and 36h.
Fresh weight change (A), water uptake rate (B), transpiration rate (C) and relative water content (D) of gladiolus flowers dry-stored for 0, 12, 24, 36, and 48h. at 5°C Lines represent mean values of cultivars Blue Frost, Gold Field, Green Star, and Jester in each treatment. Vertical bars represent the standard error of the mean.
The greater fresh weight accumulation capacity after the treatments of 12, 24, and 36h in relation to control may be related to the flower opening induced by the presence of water in control, which increases the exposure of tissues and consequently transpiration. Another factor that may be related to the divergence among treatments is the difference between room temperature (22ºC) and storage temperature (5ºC), which favors the concentration of humidity on the tissue surface.
The only time at which rehydration was not established was 48h of dry storage. Greater damage was reported in the flowers under water stress prolonged for more than 36h. A similar result was observed by VAN MEETEREN et al. (2006Van MEETEREN, U. et al. Inhibition of water uptake after dry storage of cut flowers: Role of aspired air and wound-induced processes in Chrysanthemum. Postharvest Biology and Technology , v.41, p.70-77, 2006. Available from: http://www.sciencedirect.com/science/article/pii/S0925521406000846>. Accessed: Jun. 10, 2015. doi: 10.1016/j.postharvbio.2006.03.005.
http://www.sciencedirect.com/science/art...
) in chrysanthemum, with no recovery of turgescence of flowers, in which the dry storage of stems at 20ºC for one hour led to a reduction in fresh weight in the 23h during which stems remained in water. In a study conducted by AHMAD et al. (2012AHMAD, I. et al. Dry storage effects on postharvest performance of selected cut flowers. HortTechnology, v.22, p.463-469, 2012. Available from: http://horttech.ashspublications.org/content/22/4/463.full.pdf>. Accessed: Jan. 12, 2015.
http://horttech.ashspublications.org/con...
), stems of roses stored in water lost more weight during vase life than those kept in dry storage.
Water uptake rate in the first hour was higher in control, compared with the other treatments. In dry-stored stems, the water uptake rate in the first hour decreased as the storage time was decreased (Figure 2B). After five hours of rehydration, there was a dramatic increase in water uptake rate in all treatments, including control. This increase coincides with the period when the basal flower from the stem start to open and with the peak of transpiration, as can be seen in figure 2C. This fact demonstrated that the room temperature and the induction of staggered opening of flowers in the first hours amplified the water uptake rate. In the dry-stored stems, the water uptake rate had a similar behavior to that shown by fresh weight change (Figure 2A), in which the rate was directly proportional to the duration of water stress. This, in turn, might have generated a greater water uptake potential brought about by tissue dehydration.
Control and flowers stored for 12h revealed a similar response in water uptake rate and in transpiration rate after 12h of rehydration, whereas in the other treatments there was a decrease in both variables. This decline demonstrates that the tissue was not able to rehydrate possibly due to irreversible damage caused by lack of cell hydration. In an experiment with Epidendrum ibaguense , SANTOS et al. (2012SANTOS, J.S. et al. Capacity of rehydration and influence of cut on the recovery of water flow in inflorescences of Epidendrum ibaguense. Revista Brasileira de Horticultura Ornamental, v.18, n.1, p.15-21, 2012. Available from: http://ornamentalhorticulture.emnuvens.com.br/rbho/article/viewFile/689/499>. Accessed: Jan. 15, 2015. doi: 10.14295/rbho.v18i1.689.
http://ornamentalhorticulture.emnuvens.c...
) reported that after 24h of rehydration, the transpiration rate surpassed the water uptake rate in all treatments. Flowers wilted in the period between 24 and 48h and after 96h in water. Inflorescences of Epidendrum ibaguense did not demonstrate high sensitivity to water stress after harvest, and did not require additional treatments with cutting of the stem base (FINGER et al., 2008FINGER, F.L. et al. Longevity of Epidendrum ibaguense flowers as affected by pre-loading treatments and vase solution. Journal of Horticultural Science and Biotechnology, v.83, p.144-147, 2008. Available from: http://www.tandfonline.com/doi/abs/10.1080/14620316.2008.11512361>. Accessed: Jan. 20, 2015. doi: 10.1080/14620316.2008.11512361.
http://www.tandfonline.com/doi/abs/10.10...
).
The RWC of the petals differed as a function of storage time and rehydration utilized (Figure 2D). Stems dry-stored for 12 and 24h, together with control, did not show losses in content after storage, but rather an increase, differently from those under water stress for 36 and 48h. This response can be explained by the reduced temperature in the storage chamber in comparison with the field. Coupled with the high RH of the chamber (85%), this reduction might have caused a concentration of the water content in the flower, especially on the petal surface, which contributed as a barrier against water loss. However, as this storage time was extended (36 and 48h), this loss became sharper, decreasing by 87 and 82%, respectively.
The relative water content of the petals increased up to the twelve hours of rehydration, regardless of the dry storage time. In flowers under dry storage for 36 and 48h, the relative water content was kept low throughout the rehydration period. Inflorescences that remained for 12 or 24h, when subjected to rehydration, reached values near 130% in relation to the initial RWC of the stems (100%). A higher relative water content was observed in the flowers subjected to 12h of dry storage, compared with the other treatments and also with control. In control stems, immediate hydration caused an increase in the water content of petals in the first 4h, followed by a stabilization period, with a decrease after 12h in water. Only the stems with 12 and 24h of storage had full and superior recovery of the water content in the petals, surpassing the level achieved by control after 12h in water. In the stems dry-stored for 36 and 48h, petal rehydration was partial, lower than that of control flowers, even after 24h of rehydration.
SANTOS et al. (2102SANTOS, J.S. et al. Capacity of rehydration and influence of cut on the recovery of water flow in inflorescences of Epidendrum ibaguense. Revista Brasileira de Horticultura Ornamental, v.18, n.1, p.15-21, 2012. Available from: http://ornamentalhorticulture.emnuvens.com.br/rbho/article/viewFile/689/499>. Accessed: Jan. 15, 2015. doi: 10.14295/rbho.v18i1.689.
http://ornamentalhorticulture.emnuvens.c...
) found that dry storage reduced the water content of petals of Epidendrum ibaguense , and, likewise, the decline was proportional to the duration of water stress. However, only the stems with 12h of stress had full recovery of petal water content after 24h. VIEIRA et al. (2011VIEIRA, L.M. et al. Water uptake by snapdragon inflorescences cuts after cold dry storage. Ciência Rural, v.41, n.3, p.418-423, 2011. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782011000300009>. Accessed: Jan. 20, 2014. doi: 10.1590/S0103-84782011000300009.
http://www.scielo.br/scielo.php?script=s...
) observed that, regardless of storage time, after the same period, the water content of snapdragon was lower than stems were placed in deionized water, with a reduction of 71 and 70% from zero time to 55 and 56% when stored for two and four days, respectively.
CONCLUSION:
Vase life of gladiolus flowers is not affected by the partial recuperation of the relative water content, when dry-stored for up to 36h at 5°C. Furthermore, the staggered opening of flowers is a major event that contributes with rehydration capacity of gladiolus stems.
ACKNOWLEDGMENTS
The authors are grateful to Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support.
REFERENCES:
- AHMAD, I. et al. Optimal postharvest handling protocols for cut 'Line Dance' and 'Tap Dance' Eremurus inflorescences. Scientiae Horticulturae, v.179, p.212-220, 2014. Available from: http://www.sciencedirect.com/science/article/pii/S0304423814005093> Accessed: July 15, 2015. doi: 10.1016/j.scienta.2014.09.031.
» https://doi.org/10.1016/j.scienta.2014.09.031.» http://www.sciencedirect.com/science/article/pii/S0304423814005093 - AHMAD, I. et al. Dry storage effects on postharvest performance of selected cut flowers. HortTechnology, v.22, p.463-469, 2012. Available from: http://horttech.ashspublications.org/content/22/4/463.full.pdf>. Accessed: Jan. 12, 2015.
» http://horttech.ashspublications.org/content/22/4/463.full.pdf - CÁTSKY, J. Water content. In: SLAVIK, B. Methods of studying plant water relations. Berlin: Springer-Verlag, 1974. p.121-131.
- CEVALLOS, J.C.; REID, M.S. Effect of dry and wet storage at different temperatures on vase life of cut flowers. Hort Technology, v.11, p.199-202, 2001. Available from: http://horttech.ashspublications.org/content/11/2/199.full.pdf>. Accessed: Nov. 23, 2014.
» http://horttech.ashspublications.org/content/11/2/199.full.pdf - COSTA, A.S. et al. Characterization of symptoms of senescence and chilling injury on inflorescences of Heliconia bihai (L.) cv. Lobster Claw and cv. Halloween. Postharvest Biology and Technology, v.59, p.1-7, 2010. Available from: http://www.sciencedirect.com/science/article/pii/S0925521410001894>. Accessed: Jun. 25, 2015. doi: 1016/j.postharvbio.2010.08.015.
» http://www.sciencedirect.com/science/article/pii/S0925521410001894 - COSTA, L.C. et al. Postharvest longevity of Heliconia wagneriana. Acta Horticulturae, v.1060, p.93-199, 2015. Available from: http://www.actahort.org/books/1060/1060_28.htm>. Accessed: Feb. 10, 2015.
» http://www.actahort.org/books/1060/1060_28.htm - FINGER, F.L. et al. Vase life of bird-of-paradise flowers influenced by "pulsing" and term of cold storage. Acta Horticulturae , v.628, n.1, p.863-867, 2003. Available from: http://www.actahort.org/books/628/628_110.htm>. Accessed: Mar. 10, 2015.
» http://www.actahort.org/books/628/628_110.htm - FINGER, F.L. et al. Longevity of Epidendrum ibaguense flowers as affected by pre-loading treatments and vase solution. Journal of Horticultural Science and Biotechnology, v.83, p.144-147, 2008. Available from: http://www.tandfonline.com/doi/abs/10.1080/14620316.2008.11512361>. Accessed: Jan. 20, 2015. doi: 10.1080/14620316.2008.11512361.
» https://doi.org/10.1080/14620316.2008.11512361.» http://www.tandfonline.com/doi/abs/10.1080/14620316.2008.11512361 - JAROENKIT, T.; PAULL, R.E. Postharvest handling of heliconia, red ginger and bird-of-paradise. HortTechnology , v.13, n.2, p.259-266, 2003. Available from: http://horttech.ashspublications.org/content/13/2/259.full.pdf>. Accessed: Mar. 10, 2015.
» http://horttech.ashspublications.org/content/13/2/259.full.pdf - MACNISH, A.J. et al. An alternative postharvest handling strategy for cut flowers - Dry handling after harvest. Acta Horticulturae , v.847, p.215-221, 2009. Available from: http://www.actahort.org/books/847/847_27.htm>. Accessed: Jan. 28, 2015.
» http://www.actahort.org/books/847/847_27.htm - MAPELI, A.M. et al. Influence of storage temperature on Epidendrum ibaguense flowers. Acta Scientiarum. Agronomy, v.33, n.1, p.111-115, 2011. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1807-86212011000100016>. Accessed: Jan. 20, 2015. doi: 10.4025/actasciagron.v33i1.6365.
» https://doi.org/10.4025/actasciagron.v33i1.6365.» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1807-86212011000100016 - MEMON, N. et al. Membrane stability and postharvest keeping quality of cut Gladiolus flower spikes. Journal of Agricultural Technology, v.8, n.6, p.2065-2076, 2012. Available from: http://www.ijat-aatsea.com/pdf/v8_n6_12_November/18_IJAT_2012_8(6)_Memon,%20N_Plant%20Science.pdf>. Accessed: Feb. 10, 2015.
» http://www.ijat-aatsea.com/pdf/v8_n6_12_November/18_IJAT_2012_8(6)_Memon,%20N_Plant%20Science.pdf - REID, M.S. Gladiolus. Recommendations for Maintaining Postharvest Quality. 2004. Available from: http://postharvest.ucdavis.edu/pfornamentals/Gladiolus/>. Accessed: Sept. 29, 2014.
» http://postharvest.ucdavis.edu/pfornamentals/Gladiolus/ - SALEEM, M. et al. Vase water effects on postharvest longevity and water relations of Gladiolus grandiflorus 'White Prosperity'. Pakistan Journal of Agricultural Sciences, v.51, n.1, p.137-141, 2014. Available from: http://pakjas.com.pk/papers%5C2252.pdf>. Accessed: Feb. 10, 2015.
» http://pakjas.com.pk/papers%5C2252.pdf - SANTOS, J.S. et al. Capacity of rehydration and influence of cut on the recovery of water flow in inflorescences of Epidendrum ibaguense. Revista Brasileira de Horticultura Ornamental, v.18, n.1, p.15-21, 2012. Available from: http://ornamentalhorticulture.emnuvens.com.br/rbho/article/viewFile/689/499>. Accessed: Jan. 15, 2015. doi: 10.14295/rbho.v18i1.689.
» https://doi.org/10.14295/rbho.v18i1.689.» http://ornamentalhorticulture.emnuvens.com.br/rbho/article/viewFile/689/499 - SUZUKI, A. et al. Effects of retail hydration on water uptake and quality of 'Madame Delbard" roses after long term transport. Acta Horticulturae , v.543, p.251-256, 2001. Available from: http://www.actahort.org/books/543/543_30.htm>. Accessed: Jan. 28, 2015.
» http://www.actahort.org/books/543/543_30.htm - Van DOORN, W.G. et al. Alkylethoxylate surfactants for rehydration of roses and Bouvardia flowers. Postharvest Biology and Technology , v.24, p.327-333, 2002. Available from: http://www.sciencedirect.com/science/article/pii/S0925521401001521>. Accessed: Feb. 20, 2015. doi: 10.1016/S0925-5214(01)00152-1.
» https://doi.org/10.1016/S0925-5214(01)00152-1.» http://www.sciencedirect.com/science/article/pii/S0925521401001521 - Van MEETEREN, U. et al. Inhibition of water uptake after dry storage of cut flowers: Role of aspired air and wound-induced processes in Chrysanthemum. Postharvest Biology and Technology , v.41, p.70-77, 2006. Available from: http://www.sciencedirect.com/science/article/pii/S0925521406000846>. Accessed: Jun. 10, 2015. doi: 10.1016/j.postharvbio.2006.03.005.
» https://doi.org/10.1016/j.postharvbio.2006.03.005.» http://www.sciencedirect.com/science/article/pii/S0925521406000846 - VEILING HOLAMABRA - FLORES E PLANTAS ORNAMENTAIS. Gladíolo. In: _____. Manual de classificação. 2009. Available from: <http://www.veiling.com.br/uploads/padrao_qualidade/criterios/gladiolo-fc.pdf>. Accessed: Sept. 28, 2014.
» http://www.veiling.com.br/uploads/padrao_qualidade/criterios/gladiolo-fc.pdf - VIEIRA, L.M. et al. Vascular occlusion and water relations in cut snapdragon flowers. Acta Horticulturae , v.937, p.179-184, 2012. Available from: http://www.actahort.org/books/937/937_21.htm>. Accessed: Oct. 22, 2014.
» http://www.actahort.org/books/937/937_21.htm - VIEIRA, L.M. et al. Water uptake by snapdragon inflorescences cuts after cold dry storage. Ciência Rural, v.41, n.3, p.418-423, 2011. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782011000300009>. Accessed: Jan. 20, 2014. doi: 10.1590/S0103-84782011000300009.
» https://doi.org/10.1590/S0103-84782011000300009.» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782011000300009
-
1
CR-2016-0139.R1
Publication Dates
-
Publication in this collection
12 Dec 2016 -
Date of issue
2017
History
-
Received
12 Feb 2016 -
Accepted
27 Sept 2016 -
Reviewed
22 Nov 2016