ABSTRACT:
The aim of this study was to verify the activity of some digestive enzymes in four fish species with different feeding habits. Knowledge of these enzymatic activities can help us to understand the species' digestive processes. The species chosen for this study wereCtenopharyngodon idella (herbivore),Rhamdia quelen (omnivore),Leporinus obtusidens (omnivore) andHoplias malabaricus (carnivore). The digestive tract of these species was divided into four portions to estimate enzymatic activity: stomach, anterior, mid and posterior intestine. Ctenopharyngodon idella had the highest amylase and maltase activities in all portions of the gut, followed byL. obtusidens . The highest trypsin activity was observed in all gut portions of H. malabaricus, followed by the mid intestine of L. obtusidens and the anterior intestine of C. idella . The highest chymotrypsin activity was found in all portions of C. idella followed by the anterior intestines ofR. quelen, L. obtusidens andH. malabaricus . In the stomach, acid protease activity was significantly lower in R. quelen andL. obtusidens compared toH. malabaricus. Ctenopharyngodon idella showed high activity of enzymes that hydrolyze carbohydrates, represented in this study by amylase and maltase andH. malabaricus showed higher protease activity and low amylase activity.
Key words:
freshwater fishes; feeding habits; enzymatic activity; amylase; alkaline protease; maltase; trypsin
RESUMO:
O objetivo deste estudo foi verificar a atividade de algumas enzimas digestivas em quatro espécies de peixes com hábitos alimentares diferentes. O conhecimento das atividades dessas enzimas pode nos ajudar a compreender os processos digestivos das espécies. As espécies escolhidas para este estudo foramCtenopharyngodon idella (herbívoro),Rhamdia quelen (onívoro),Leporinus obtusidens (onívoro) eHoplias malabaricus (carnívoro). O trato digestivo dessas espécies foi dividido em quatro partes para estimar a atividade enzimática: estômago, intestino anterior, médio e posterior.Ctenopharyngodon idella teve a maior atividade da amilase e maltase em todas as porções do intestino, seguido por L. obtusidens . A maior atividade da tripsina foi observada em todas as porções do intestino de H. malabaricus , seguido pelo intestino médio deL. obtusidens e o intestino anterior de C. idella . A maior atividade de quimotripsina foi encontrada em todas as partes do intestino do C. idella , seguida pelo intestino anterior deR. quelen, L. obtusidens eH. malabaricus . No estômago, a atividade de protease ácida foi significativamente menor emR. quelen eL. obtusidens comparado com H. malabaricus. Ctenopharyngodon idella mostrou alta atividade de enzimas que hidrolisam carboidratos, representadas neste estudo por amilase e maltase eH. malabaricus mostrou maior atividade de proteases e baixa atividade de amilase.
Palavras-chave:
peixes de água doce; hábitos alimentares; atividade enzimática; amilase; protease alcalina; maltase; tripsina
INTRODUCTION:
Fish usually exhibit high versatility in their feeding habits that is reflected in different anatomical and functional features. This allows fish to explore a wide range of food resources and maximize the use of available food in the environment (PERETTI & ADRIAN, 2008PERETTI, D.; ANDRIAN, I. F. Feeding and morphological analysis of the digestive tract of four species of fish (Astyanax altiparanae, Parauchenipterus galeatus, Serrasalmus marginatus and Hoplias aff. malabaricus) from the upper Paraná River floodplain, Brazil. Brazilian Journal of Biology, v.68, n.3, p.671-679, 2008. Avaliable from: < Avaliable from: http://www.scielo.br/scielo.php?pid= S1519-69842008000300027&script=sci_arttext
>. Accessed: Feb. 01, 2016. doi: 10.1590/S1519-698420080 00300027.
http://www.scielo.br/scielo.php?pid= S15...
). The digestive potential of fish is highly variable, changing with species, age, size, food and feeding history, stage of maturity and temperature (GARCÍA-CARREÑO et al., 2002GARCÍA-CARREÑO, F. L. et al. Digestive proteinases of Brycon orbignyanus (Characidae, Teleostei): characteristics and effects of protein quality. Comparative Biochemistry and Physiology - Part B, v.132, p.343-352, 2002. Available from: http://www.bashanfoundation. org/carreno/carrrenocharacidae.pdf>. Accessed: Feb. 01, 2016.
http://www.bashanfoundation. org/carreno...
). The analysis of digestive enzymes provides information on fish nutritional physiology and on their ability to take advantage of the different nutritional fractions of the feed (TENGJAROENKUL et al., 2000TENGJAROENKUL, B. et al. Distribution of intestinal enzyme activities along the intestinal tract of cultured Nile tilapia, Oreochromis niloticus L. Aquaculture , v.182, p.317-327, 2000. Available from: http://www.thaiscience.info/Article%20for%20ThaiScience/Article/2/Ts-2%20distrib.of%20intestinal%20enzyme%20act.along%20intestinal%20tract%20of%20cultured%20nile%20tilapia,%20oreochromis%20niloticus%20l..pdf>. Accessed: Feb. 01, 2016.
http://www.thaiscience.info/Article%20fo...
; ODEDEYI & FAGBENRO, 2010ODEDEYI, D. O.; FAGBENRO, O. A. Feeding habits and digestive enzymes in the gut of Mormyrus rume (Valenciennes 1846) (Osteichthtes, Mormyridae). Tropical Zoology, v.23, p.75-89, 2010. Avaliable from: < Avaliable from: https://www.researchgate.net/publication/287551841_Feeding_ habits_and_digestive_enzymes_in_the_gut_of_Mormyrus_rume_Valenciennes_1846_Osteichth yes_Mormyridae
>. Accessed: Feb. 01, 2016.
https://www.researchgate.net/publication...
; LAZZARI et al., 2010LAZZARI, R. et al. Protein sources and digestive enzyme activities in jundiá (Rhamdia quelen). Scientia Agricola, v.67, n.3, p.259-266, 2010. Avaliable from: < Avaliable from: http://www.scielo.br/pdf/s/v67n 3/a02v67n3.pdf
>. Accessed: Feb. 01, 2016.
http://www.scielo.br/pdf/s/v67n 3/a02v67...
; 2015LAZZARI, R. et al. Utilização de resíduos de frutas em dietas para piava. Boletim do Instituto de Pesca, v.41, n.2, p. 227-237, 2015. Avaliable from: < Avaliable from: ftp://ftp.sp.gov.br/ftppesca/41_2_227-237.pdf
Accessed: Feb. 01, 2016.
ftp://ftp.sp.gov.br/ftppesca/41_2_227-23...
).
Ctenopharyngodon idella (grass carp) (Characiformes, Cyprinidae ), is an herbivorous fish species that feeds on aquatic plants (BILLARD & BERNI, 2004BILLARD, R.; BERNI, P. Trends in cyprinid polyculture. Cybium, v.28, p.255-261, 2004. Avaliable from: < Avaliable from: http://sfi.mnhn.fr/cybium/numeros/2004/283/11-Billard193.pdf
>. Accessed: Feb. 01, 2016.
http://sfi.mnhn.fr/cybium/numeros/2004/2...
). The omnivorous Leporinus obtusidens (piava) (Characiformes, Anostomidae ) feed on plants, insects and fishes (REYNALTE-TATAJE & ZANIBONI-FILHO, 2010REYNALTE-TATAJE, D.; ZANIBONI-FILHO, E. Cultivo de piapara, piauçu, piava e piau - gênero Leporinus. In: BALDISSEROTTO, B.; GOMES, L. C. (Eds.). Espécies nativas para piscicultura no Brasil . Santa Maria: UFSM , 2010. p.73-99.; LAZZARI et al., 2015LAZZARI, R. et al. Utilização de resíduos de frutas em dietas para piava. Boletim do Instituto de Pesca, v.41, n.2, p. 227-237, 2015. Avaliable from: < Avaliable from: ftp://ftp.sp.gov.br/ftppesca/41_2_227-237.pdf
Accessed: Feb. 01, 2016.
ftp://ftp.sp.gov.br/ftppesca/41_2_227-23...
). Rhamdia quelen (silver catfish) (Siluriformes, Heptapteridae ) is an omnivorous fish with a tendency towards ichthyophagy, depending on food availability in the environment (BALDISSEROTTO et al., 2013BALDISSEROTTO, B. et al. Jundiá (Rhamdia sp.). In: BALDISSEROTTO, B.; GOMES, L.C. (Eds.). Espécies nativas para piscicultura no Brasil. 2.ed. Santa Maria: UFSM, 2013. p.301-323.). Hoplias malabaricus (traira) (Characiformes, Erythrinidae ) is a carnivorous fish species that, in the adult stage, ingests intact preys (MENIN & MIMURA, 1991MENIN, E.; MIMURA, O. M. Anatomia funcional da cavidade bucofaringeana de Hoplias malabaricus (Bloch, 1794) (Characiformes, Erythrinidae). Revista Ceres, v.38, p.240-255, 1991. Avaliable from: < Avaliable from: http://www.ceres.ufv.br/ojs/index.php/ceres/article/download/2137/188
>. Accessed: Feb. 01, 2016.
http://www.ceres.ufv.br/ojs/index.php/ce...
).
Several studies of digestive enzymes in fish species with different feeding habits have been reported (KUZ´MINA & KUZ´MINA, 1990KUZʹMINA, V. V.; KUZʹMINA, Y. G. Level of total proteolytic activity in some species of fish from the Volga barin. Journal of Ichthyology, v.30, p.25-35, 1990.; CHAKRABARTI et al., 2006CHAKRABARTI, R. et al. Functional changes in digestive enzymes and characterization of proteases of silver carp (♂) and bighead carp (♀) hybrid, during early ontogeny. Aquaculture, v.253, p.694-702, 2006. Avaliable from: < Avaliable from: https://www.researchgate.net/profile/RajaRathore /publication/232380390Functionalchangesindigestiveenzymesandcharacterizationofproteasesofsilvercarp%28%29andbigheadcarp%28%29hybridduringearlyontogeny/links/0046351c18baa96e1400 0000.pdf
>. Accessed: Feb. 01, 2016. doi: 10.1016/j.aquaculture.2005.08.018.
https://www.researchgate.net/profile/Raj...
; LÓPEZ-VÁSQUEZ et al., 2009LÓPEZ-VÁSQUEZ, K. et al. Digestive enzymes of eight Amazonian teleosts with different feeding habits. Journal of Fish Biology, v.74, p.1620-1628, 2009. Available from: http://onlinelibrary.willey.com/doi/10.1111/j.1095-8649.2009.02196.x/epdf>. Acessed: Feb. 01, 2016.
http://onlinelibrary.willey.com/doi/10.1...
) and some showed that fish growth might be related to digestive enzyme capacity (CHAKRABARTI et al., 2006; FILIPPOV et al., 2013FILIPPOV, A. A. et al. Effects of organic pollutants on fish digestive enzymes: a review. Inland Water Biology, v.6, p.155-160, 2013. Available from: http://link.springer.com/article/10.1134%2FS199508291302003X>. Accessed: Feb. 01, 2016. doi: 10.1134/S199508291302003X.
http://link.springer.com/article/10.1134...
; LAZZARI et al., 2015LAZZARI, R. et al. Utilização de resíduos de frutas em dietas para piava. Boletim do Instituto de Pesca, v.41, n.2, p. 227-237, 2015. Avaliable from: < Avaliable from: ftp://ftp.sp.gov.br/ftppesca/41_2_227-237.pdf
Accessed: Feb. 01, 2016.
ftp://ftp.sp.gov.br/ftppesca/41_2_227-23...
). The aim of this study was to investigate protease and carbohydrase activities in four fish species raised in Brazil. The major propose is collected information concerning fish cultivated in Southern Brazil for improved in the future fish diets according to digestive enzymatic profile.
MATERIALS AND METHODS:
Ten individuals of each species, C. idella (20.46±3.9g and 12.95±1.7cm),R. quelen (66.20±10.5g and 18.75±1.5cm) andL. obtusidens (69.80±11.7g and 25.41±0.8cm) with approximate age were obtained from the fish culture sector at Universidade Federal de Santa Maria. Hoplias malabaricus (60.30±10.9g and 16.64±1.6cm) was obtained from regional pond producer. Both species were fasted for 12h before euthanasia by section of spinal cord. Body weight and length were taken subsequently.
The fish were kept in 250L tanks with proper charge density for each species, and water quality parameters were monitored regularly: dissolved oxygen (5.8±0.6mg L-1); temperature (23±0.4ºC, using an oxygen meter Y5512; YSI Inc., Yellow Springs, OH, USA); pH 7.5±0.3 (DMPH-2 pH meter, Digimed, São Paulo, SP, Brazil); total ammonia nitrogen levels (0.14±0.04mg L-1) (EATON et al., 2005EATON, A. D. et al. Standard methods for the examination of water and wastewater. 21. ed. Washington: American Public Health Association, 2005. 1193p.); un-ionized ammonia (NH3) levels 0.007±0.001mg L-1 (COLT, 2002COLT, J. List of spreadsheets prepared as a complement. In: WEDEMEYER, G. A. (Ed.). Fish hatchery management. 2. ed. Bethesda: American Fisheries Society Publications, 2002. 751p.); alkalinity (42±2.7mg L-1 CaCO3); nitrite (0.0033±0.003mg L-1) (BOYD & TUCKER, 1992BOYD, C. E.; TUCKER, C. S. Water quality and pond soil analyses for aquaculture. Alabama: Alabama Agricultural Experiment Station, Auburn University, 1992. 188p.) and; water hardness (26.0±1.4mg L-1 CaCO3) (EDTA titrimetric method).
The digestive tract was immediately removed after euthanasia and divided into four sections: stomach (except for the grass carp, which does not possess stomach), anterior (or pyloric ceca), mid and posterior intestines. Portions were placed into ice and then stored at -20ºC. Tissues were homogenized in buffer solution containing phosphate (10mM) and Tris (20mM), pH 7.0 using a Potter-Elvehijen homogenizer. The homogenates were centrifuged at 10.000g for 10min at 4ºC and the supernatant (crude extract) was used as an enzyme source for all assays.
The effect of different pH on the incubation medium was studied for protease alkaline and amylase activities. Total acid protease activity was measured in the stomach, using a non-specific substrate (casein 1.5%) according to HIDALGO et al. (1999HIDALGO, M. C. et al. Comparative study of digestive enzymes in fish with different nutritional habits. Proteolytic and amylase activities. Aquaculture , v.170, p.267-283, 1999. Available from: http://ac.els-cdn.com/S004484869800413X/1-s2.0-S004484869800413X-main.pdf?_tid=cc3f0 85e-7c72-11e3-8614-00000aab0f26&acdnat=1389632004_81ce406d86b8e1217d669d5a1545a 634> . Accessed: Jan. 13, 2014.
http://ac.els-cdn.com/S004484869800413X/...
).
Trypsin, chymotrypsin, amylase and maltase were determined in homogenates from the stomach and anterior, mid and posterior intestine. The experimental protocol was modified according to BERNFELD (1955BERNFELD, P. Amylases a and b: colorimetric assay methods. In: COLOWICK, S. P.; KAPLAN, N. O. Methods in Enzimology. New York: Academic, 1955. p.49-158.). The starch hydrolyzed by the enzyme and glucose levels were determined according to PARK & JOHNSON (1949PARK, J. T.; JOHNSON, M. J. A submicro determination of glucose. Journal of Biological Chemistry , v.181, p.149-151, 1949. Available from: www.jbc.org/content/181/1/149.long>. Accessed: Feb. 03, 2016.
www.jbc.org/content/181/1/149.long...
). The protein content of crude extracts was determined by the method of LOWRY et al. (1951LOWRY, O. H. et al. Protein measurement with folin phenol reagent. Journal of Biological Chemistry, v.193, p.265-275, 1951. Avaliable from: < Avaliable from: http://garfield.library.upenn.edu/classics1977/A1977DM02300001.pdf
>. Accessed: Feb. 01, 2016.
http://garfield.library.upenn.edu/classi...
), using bovine serum albumin as a standard. For more details, see the technical of study LAZZARI et al. (2010LAZZARI, R. et al. Protein sources and digestive enzyme activities in jundiá (Rhamdia quelen). Scientia Agricola, v.67, n.3, p.259-266, 2010. Avaliable from: < Avaliable from: http://www.scielo.br/pdf/s/v67n 3/a02v67n3.pdf
>. Accessed: Feb. 01, 2016.
http://www.scielo.br/pdf/s/v67n 3/a02v67...
).
Differences between species were analyzed by one-way analysis of variance followed by the Duncan test (Statistica 5.0). Data were expressed as mean ± SEM, and differences were considered significant at a probability level of 95% (P<0.05).
RESULTS:
Trypsin, chymotrypsin and maltase activities were observed in the stomach of R. quelen , L. obtusidens , and H. malabaricus . The greatest trypsin activity was found in H. malabaricus following for L. obtusidens . The low values for trypsin were recorded to R. quelen . Chymotrypsin activity in the stomach was similar to R. quelen and H. malabaricus , and L. obtusidens showed lower activity as compared to other fish species. Maltase activity was highest in L. obtusidens comparing to H. malabaricus and R. quelen that showed similar maltase activity in stomach (Table 1).
Trypsin, chymotrypsin and maltase activities in the stomach of the studied species data on enzyme digestive activities (n=10) are expressed as U mg protein-1 where U=1μmol of substrate hydrolyzed min-1. Different superscript letters represent significant difference of digestive enzymes activity comparing different fish species (P<0.05).
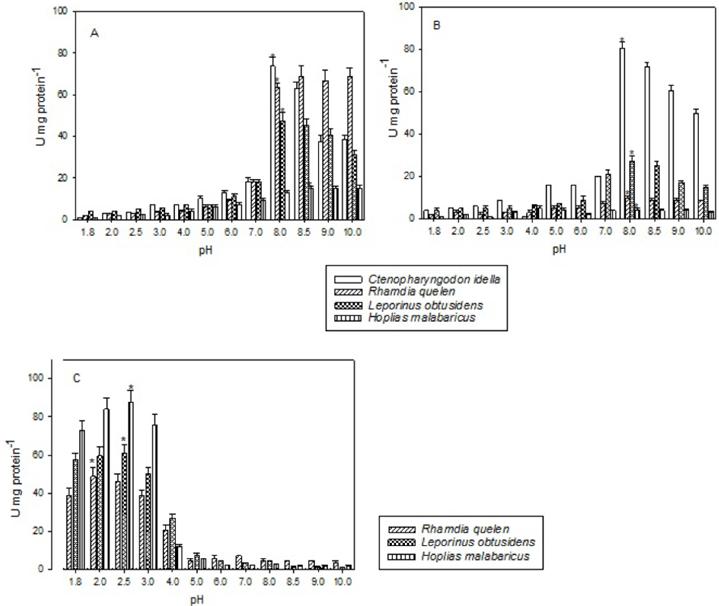
Ctenopharyngodon idella andL. obtusidens showed the highest amylase activity at pH 7.0 and H. malabaricus at pH 8.5 and pH 8.0 in the anterior and midintestine, respectively (Figure 1A and B). Rhamdia quelen showed highest activity at pH 7.0 in the middle intestine (Figure 1B). Higher amylase activity in the anterior and mid in C. idella (Figure 1A and B). In the stomach the highest amylase activity was observed at pH 7.0 for L. obtusidens and R. quelen and pH 8.0 for H. malabaricus (Figure 1C).
Amylase activity at different pHs in the anterior (A) and min-1 (B) intestine and stomach (C) of the studied species. Activity is expressed as U mg protein-1, where U=1 μmol glucose min mg protein-1. Data represent mean ± SEM (n=10). *Represents the pH at which the enzyme has the highest activity.
Alkaline protease presented the highest activities at pH 8.0 or 8.5 for all species, but R. quelen and H. malabaricus also showed high activities at pH 9.0 and 10.0. Compared to the other species, C. idella continued to show high activity of alkaline protease in the middle portion of the intestine (Figure 2A and B). The highest acid protease activity in the stomach was at pH 2.5 for H. malabaricus and L. obtusidens and pH 2.0 for R. quelen (Figure 2C). In C. idella the mid intestine showed the highest activity for amylase and maltase when compared to anterior and posterior segments. In R. quelen the highest activity of these enzymes was exhibited in the anterior intestine. In L. obtusidens the highest amylase activity was detected in the anterior intestine and maltase in the mid intestine, but H. malabaricus showed very low amylase activity in all intestine portions. On the other hand, H. malabaricus presented maltase activity in middle and posterior intestine portions similar to that obtained for R. quelen , but lower than L. obtusidens and C. idella. (Figure 3).
Alkaline protease activity at different pHs in the anterior (A) and mid (B) intestine and acid protease activity in the stomach (C) of the studied species. Activity is expressed as U mg protein-1, where U=1μg tyrosine min-1 mg protein-1. Data represent mean ±SEM (n=10). *Represents the pH at which the enzyme has the highest activity.
Amylase (A) and maltase (B) activities in anterior, middle and posterior intestine of the studied species. Activity is expressed as U mg protein-1, where U=1μmol glucose min-1 mg protein-1. Trypsin (C) and chymotrypsin (D) activity in anterior, middle and posterior intestine of the studied species. Activity is expressed as U mg protein-1, where U= g of substrate hydrolyzed (TAME or BTEE) min-1 mg protein-1. Data represent mean ± SEM (n=10). *Represents the portion of the intestine where the highest activity is observed for each enzyme.
The highest trypsin activity was observed in all intestine portions of H. malabaricus . Ctenopharyngodon idella exhibited the highest trypsin activity in the anterior intestine, L. obtusidens in the mid intestine and R. quelen in the posterior intestine (Figure 3C). Ctenopharyngodon idella showed the highest chymotrypsin activity at all intestine portions. Rhamdia quelen and H. malabaricus showed high activity of this enzyme in the anterior portion but for L. obtusidens the highest activity was observed in the posterior intestine (Figure 3D).
DISCUSSION:
The knowledge of the feeding habits of different fish species associated with enzymes activities in the digestive tract is important to provide an appropriate diet for each species because digestive enzymes activity very reflects the changes in dietary (PERETTI & ANDRIAN, 2008PERETTI, D.; ANDRIAN, I. F. Feeding and morphological analysis of the digestive tract of four species of fish (Astyanax altiparanae, Parauchenipterus galeatus, Serrasalmus marginatus and Hoplias aff. malabaricus) from the upper Paraná River floodplain, Brazil. Brazilian Journal of Biology, v.68, n.3, p.671-679, 2008. Avaliable from: < Avaliable from: http://www.scielo.br/scielo.php?pid= S1519-69842008000300027&script=sci_arttext
>. Accessed: Feb. 01, 2016. doi: 10.1590/S1519-698420080 00300027.
http://www.scielo.br/scielo.php?pid= S15...
; LAZZARI et al., 2010LAZZARI, R. et al. Protein sources and digestive enzyme activities in jundiá (Rhamdia quelen). Scientia Agricola, v.67, n.3, p.259-266, 2010. Avaliable from: < Avaliable from: http://www.scielo.br/pdf/s/v67n 3/a02v67n3.pdf
>. Accessed: Feb. 01, 2016.
http://www.scielo.br/pdf/s/v67n 3/a02v67...
; 2015).
The carnivorous H. malabaricus showed lower amylase, maltase, alkaline protease and chymotrypsin activity compared to the other fish species studied. The main digestive enzymes of H. malabaricus are represented by acid protease in the stomach and trypsin in the intestine, but this species also showed some chymotrypsin and maltase activity in the stomach and all intestine portions. Other carnivorous species such as Oncorhynchus mykiss and Pseudoplatystoma corruscans also showed high protease activity in the stomach (HIDALGO et al., 1999HIDALGO, M. C. et al. Comparative study of digestive enzymes in fish with different nutritional habits. Proteolytic and amylase activities. Aquaculture , v.170, p.267-283, 1999. Available from: http://ac.els-cdn.com/S004484869800413X/1-s2.0-S004484869800413X-main.pdf?_tid=cc3f0 85e-7c72-11e3-8614-00000aab0f26&acdnat=1389632004_81ce406d86b8e1217d669d5a1545a 634> . Accessed: Jan. 13, 2014.
http://ac.els-cdn.com/S004484869800413X/...
; LUNDSTEDT et al., 2004LUNDSTEDT, L. M. et al. Digestive enzymes and metabolic profile of Pseudoplatystoma corruscans (Teleostei: Siluriformes) in response to diet composition. Comparative Biochemistry and Physiology - Part B , v.137, p.331-339, 2004. Available from: http://ac.els-cdn.com/S109649590303828/1-s2.0-S1096495903003828-main.pdf?tid=b8b1db7c-a4ec-11e6-91a2-00000aab0f01& acdnat=1478524910_3f0b1e79f3d7dca29dfb1f70145f627a>. Accessed: Feb. 01, 2016.
http://ac.els-cdn.com/S109649590303828/1...
). In the present study, amylase and maltase activities were found in H. malabaricus , likely because they would be required to digest the glycogen present in animal tissues. Another carnivorous fish, the sea bass (Lates calcarifer ), also presented amylase activity in the digestive tract (SABAPATHY & TÉO, 1992SABAPATHY, U.; TEO, L. H. A quantitative study of some digestive enzymes in the rabbitfish, Siganus canaliculatus and the Sea bass, Lates calcarifer. Journal of Fish Biology , v.42, p.595-602, 1992. Avaliable from: < Avaliable from: http://onlinelibrary.wiley.com/doi/10.1111/j.1095-8649.1993.tb003 62.x/pdf
>. Accessed: Feb. 01, 2016. doi: 10.1016/S0022-474X(00)00016-3.
http://onlinelibrary.wiley.com/doi/10.11...
).
The highest total alkaline protease activity measured in the intestine was found at pH 8.0 and 8.5 for all fish species studied. However, some activity was also detected at pH 9.0 and 10.0 for all species, mainly in the anterior intestine. On the other hand, GARCÍA-CARREÑO et al. (2002GARCÍA-CARREÑO, F. L. et al. Digestive proteinases of Brycon orbignyanus (Characidae, Teleostei): characteristics and effects of protein quality. Comparative Biochemistry and Physiology - Part B, v.132, p.343-352, 2002. Available from: http://www.bashanfoundation. org/carreno/carrrenocharacidae.pdf>. Accessed: Feb. 01, 2016.
http://www.bashanfoundation. org/carreno...
) verified that the optimum pH for intestinal enzymes of Brycon orbignyanus was 10.0. The results observed at high alkaline pHs (9.0 and 10.0) are probably due to other alkaline proteases as carboxipeptidase-like, elastase-like or collagenase-like activities (HIDALGO et al., 1999HIDALGO, M. C. et al. Comparative study of digestive enzymes in fish with different nutritional habits. Proteolytic and amylase activities. Aquaculture , v.170, p.267-283, 1999. Available from: http://ac.els-cdn.com/S004484869800413X/1-s2.0-S004484869800413X-main.pdf?_tid=cc3f0 85e-7c72-11e3-8614-00000aab0f26&acdnat=1389632004_81ce406d86b8e1217d669d5a1545a 634> . Accessed: Jan. 13, 2014.
http://ac.els-cdn.com/S004484869800413X/...
).
The herbivore C. idella presented the highest amylase and maltase activities within the fish species studied, in accordance with CHAKRABARTI et al. (2006CHAKRABARTI, R. et al. Functional changes in digestive enzymes and characterization of proteases of silver carp (♂) and bighead carp (♀) hybrid, during early ontogeny. Aquaculture, v.253, p.694-702, 2006. Avaliable from: < Avaliable from: https://www.researchgate.net/profile/RajaRathore /publication/232380390Functionalchangesindigestiveenzymesandcharacterizationofproteasesofsilvercarp%28%29andbigheadcarp%28%29hybridduringearlyontogeny/links/0046351c18baa96e1400 0000.pdf
>. Accessed: Feb. 01, 2016. doi: 10.1016/j.aquaculture.2005.08.018.
https://www.researchgate.net/profile/Raj...
), who found high amylase activity in herbivorous fish. However, C. idella also presented alkaline protease, trypsin and chymotrypsin activities in the intestine. In agreement with the present study, KUZʹMINA & KUZʹMINA (1990KUZʹMINA, V. V.; KUZʹMINA, Y. G. Level of total proteolytic activity in some species of fish from the Volga barin. Journal of Ichthyology, v.30, p.25-35, 1990.) and CHAKRABARTI et al. (2006)CHAKRABARTI, R. et al. Functional changes in digestive enzymes and characterization of proteases of silver carp (♂) and bighead carp (♀) hybrid, during early ontogeny. Aquaculture, v.253, p.694-702, 2006. Avaliable from: < Avaliable from: https://www.researchgate.net/profile/RajaRathore /publication/232380390Functionalchangesindigestiveenzymesandcharacterizationofproteasesofsilvercarp%28%29andbigheadcarp%28%29hybridduringearlyontogeny/links/0046351c18baa96e1400 0000.pdf
>. Accessed: Feb. 01, 2016. doi: 10.1016/j.aquaculture.2005.08.018.
https://www.researchgate.net/profile/Raj...
also found high protease activity in non-carnivorous fish. In addition, the herbivorous Oreochromis niloticus (Nile tilapia) demonstrated higher carbohydrase activity than protease activity when compared to carnivorous and omnivorous fish (TENGJAROENKUL et al., 2000TENGJAROENKUL, B. et al. Distribution of intestinal enzyme activities along the intestinal tract of cultured Nile tilapia, Oreochromis niloticus L. Aquaculture , v.182, p.317-327, 2000. Available from: http://www.thaiscience.info/Article%20for%20ThaiScience/Article/2/Ts-2%20distrib.of%20intestinal%20enzyme%20act.along%20intestinal%20tract%20of%20cultured%20nile%20tilapia,%20oreochromis%20niloticus%20l..pdf>. Accessed: Feb. 01, 2016.
http://www.thaiscience.info/Article%20fo...
). Different authors have reported a close relationship between herbivorous feeding habits and higher amylase activity (HIDALGO et al., 1999HIDALGO, M. C. et al. Comparative study of digestive enzymes in fish with different nutritional habits. Proteolytic and amylase activities. Aquaculture , v.170, p.267-283, 1999. Available from: http://ac.els-cdn.com/S004484869800413X/1-s2.0-S004484869800413X-main.pdf?_tid=cc3f0 85e-7c72-11e3-8614-00000aab0f26&acdnat=1389632004_81ce406d86b8e1217d669d5a1545a 634> . Accessed: Jan. 13, 2014.
http://ac.els-cdn.com/S004484869800413X/...
; ODEDEYI & FAGBENRO, 2010ODEDEYI, D. O.; FAGBENRO, O. A. Feeding habits and digestive enzymes in the gut of Mormyrus rume (Valenciennes 1846) (Osteichthtes, Mormyridae). Tropical Zoology, v.23, p.75-89, 2010. Avaliable from: < Avaliable from: https://www.researchgate.net/publication/287551841_Feeding_ habits_and_digestive_enzymes_in_the_gut_of_Mormyrus_rume_Valenciennes_1846_Osteichth yes_Mormyridae
>. Accessed: Feb. 01, 2016.
https://www.researchgate.net/publication...
).
In conclusion our study showed higher acid protease and trypsin activities in the carnivorous species studied, while higher amylase and maltase activities were found in the herbivorous species. Omnivorous species presented intermediate activity values. The results are within the expected range for each species and their feeding habits. The application of these feedings in the formulation of specific diets for each species using sources of low cost and good digestibility to fish
ACKNOWLEDGEMENTS
The authors thank Dr. Everton Behr for his technical help. V.L. Loro and B. Baldisserotto received research fellowships from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq - Brazil)
REFERENCES:
- BALDISSEROTTO, B. et al. Jundiá (Rhamdia sp.). In: BALDISSEROTTO, B.; GOMES, L.C. (Eds.). Espécies nativas para piscicultura no Brasil. 2.ed. Santa Maria: UFSM, 2013. p.301-323.
- BERNFELD, P. Amylases a and b: colorimetric assay methods. In: COLOWICK, S. P.; KAPLAN, N. O. Methods in Enzimology. New York: Academic, 1955. p.49-158.
- BILLARD, R.; BERNI, P. Trends in cyprinid polyculture. Cybium, v.28, p.255-261, 2004. Avaliable from: < Avaliable from: http://sfi.mnhn.fr/cybium/numeros/2004/283/11-Billard193.pdf >. Accessed: Feb. 01, 2016.
» http://sfi.mnhn.fr/cybium/numeros/2004/283/11-Billard193.pdf - BOYD, C. E.; TUCKER, C. S. Water quality and pond soil analyses for aquaculture. Alabama: Alabama Agricultural Experiment Station, Auburn University, 1992. 188p.
- CHAKRABARTI, R. et al. Functional changes in digestive enzymes and characterization of proteases of silver carp (♂) and bighead carp (♀) hybrid, during early ontogeny. Aquaculture, v.253, p.694-702, 2006. Avaliable from: < Avaliable from: https://www.researchgate.net/profile/RajaRathore /publication/232380390Functionalchangesindigestiveenzymesandcharacterizationofproteasesofsilvercarp%28%29andbigheadcarp%28%29hybridduringearlyontogeny/links/0046351c18baa96e1400 0000.pdf >. Accessed: Feb. 01, 2016. doi: 10.1016/j.aquaculture.2005.08.018.
» https://www.researchgate.net/profile/RajaRathore /publication/232380390Functionalchangesindigestiveenzymesandcharacterizationofproteasesofsilvercarp%28%29andbigheadcarp%28%29hybridduringearlyontogeny/links/0046351c18baa96e1400 0000.pdf - COLT, J. List of spreadsheets prepared as a complement. In: WEDEMEYER, G. A. (Ed.). Fish hatchery management. 2. ed. Bethesda: American Fisheries Society Publications, 2002. 751p.
- EATON, A. D. et al. Standard methods for the examination of water and wastewater. 21. ed. Washington: American Public Health Association, 2005. 1193p.
- FILIPPOV, A. A. et al. Effects of organic pollutants on fish digestive enzymes: a review. Inland Water Biology, v.6, p.155-160, 2013. Available from: http://link.springer.com/article/10.1134%2FS199508291302003X>. Accessed: Feb. 01, 2016. doi: 10.1134/S199508291302003X.
» https://doi.org/10.1134/S199508291302003X» http://link.springer.com/article/10.1134%2FS199508291302003X - GARCÍA-CARREÑO, F. L. et al. Digestive proteinases of Brycon orbignyanus (Characidae, Teleostei): characteristics and effects of protein quality. Comparative Biochemistry and Physiology - Part B, v.132, p.343-352, 2002. Available from: http://www.bashanfoundation. org/carreno/carrrenocharacidae.pdf>. Accessed: Feb. 01, 2016.
» http://www.bashanfoundation. org/carreno/carrrenocharacidae.pdf - HIDALGO, M. C. et al. Comparative study of digestive enzymes in fish with different nutritional habits. Proteolytic and amylase activities. Aquaculture , v.170, p.267-283, 1999. Available from: http://ac.els-cdn.com/S004484869800413X/1-s2.0-S004484869800413X-main.pdf?_tid=cc3f0 85e-7c72-11e3-8614-00000aab0f26&acdnat=1389632004_81ce406d86b8e1217d669d5a1545a 634> . Accessed: Jan. 13, 2014.
» http://ac.els-cdn.com/S004484869800413X/1-s2.0-S004484869800413X-main.pdf?_tid=cc3f0 85e-7c72-11e3-8614-00000aab0f26&acdnat=1389632004_81ce406d86b8e1217d669d5a1545a 634 - KUZʹMINA, V. V.; KUZʹMINA, Y. G. Level of total proteolytic activity in some species of fish from the Volga barin. Journal of Ichthyology, v.30, p.25-35, 1990.
- LAZZARI, R. et al. Protein sources and digestive enzyme activities in jundiá (Rhamdia quelen). Scientia Agricola, v.67, n.3, p.259-266, 2010. Avaliable from: < Avaliable from: http://www.scielo.br/pdf/s/v67n 3/a02v67n3.pdf >. Accessed: Feb. 01, 2016.
» http://www.scielo.br/pdf/s/v67n 3/a02v67n3.pdf - LAZZARI, R. et al. Utilização de resíduos de frutas em dietas para piava. Boletim do Instituto de Pesca, v.41, n.2, p. 227-237, 2015. Avaliable from: < Avaliable from: ftp://ftp.sp.gov.br/ftppesca/41_2_227-237.pdf Accessed: Feb. 01, 2016.
» ftp://ftp.sp.gov.br/ftppesca/41_2_227-237.pdf - LÓPEZ-VÁSQUEZ, K. et al. Digestive enzymes of eight Amazonian teleosts with different feeding habits. Journal of Fish Biology, v.74, p.1620-1628, 2009. Available from: http://onlinelibrary.willey.com/doi/10.1111/j.1095-8649.2009.02196.x/epdf>. Acessed: Feb. 01, 2016.
» http://onlinelibrary.willey.com/doi/10.1111/j.1095-8649.2009.02196.x/epdf - LOWRY, O. H. et al. Protein measurement with folin phenol reagent. Journal of Biological Chemistry, v.193, p.265-275, 1951. Avaliable from: < Avaliable from: http://garfield.library.upenn.edu/classics1977/A1977DM02300001.pdf >. Accessed: Feb. 01, 2016.
» http://garfield.library.upenn.edu/classics1977/A1977DM02300001.pdf - LUNDSTEDT, L. M. et al. Digestive enzymes and metabolic profile of Pseudoplatystoma corruscans (Teleostei: Siluriformes) in response to diet composition. Comparative Biochemistry and Physiology - Part B , v.137, p.331-339, 2004. Available from: http://ac.els-cdn.com/S109649590303828/1-s2.0-S1096495903003828-main.pdf?tid=b8b1db7c-a4ec-11e6-91a2-00000aab0f01& acdnat=1478524910_3f0b1e79f3d7dca29dfb1f70145f627a>. Accessed: Feb. 01, 2016.
» http://ac.els-cdn.com/S109649590303828/1-s2.0-S1096495903003828-main.pdf?tid=b8b1db7c-a4ec-11e6-91a2-00000aab0f01& acdnat=1478524910_3f0b1e79f3d7dca29dfb1f70145f627a - MENIN, E.; MIMURA, O. M. Anatomia funcional da cavidade bucofaringeana de Hoplias malabaricus (Bloch, 1794) (Characiformes, Erythrinidae). Revista Ceres, v.38, p.240-255, 1991. Avaliable from: < Avaliable from: http://www.ceres.ufv.br/ojs/index.php/ceres/article/download/2137/188 >. Accessed: Feb. 01, 2016.
» http://www.ceres.ufv.br/ojs/index.php/ceres/article/download/2137/188 - ODEDEYI, D. O.; FAGBENRO, O. A. Feeding habits and digestive enzymes in the gut of Mormyrus rume (Valenciennes 1846) (Osteichthtes, Mormyridae). Tropical Zoology, v.23, p.75-89, 2010. Avaliable from: < Avaliable from: https://www.researchgate.net/publication/287551841_Feeding_ habits_and_digestive_enzymes_in_the_gut_of_Mormyrus_rume_Valenciennes_1846_Osteichth yes_Mormyridae >. Accessed: Feb. 01, 2016.
» https://www.researchgate.net/publication/287551841_Feeding_ habits_and_digestive_enzymes_in_the_gut_of_Mormyrus_rume_Valenciennes_1846_Osteichth yes_Mormyridae - PARK, J. T.; JOHNSON, M. J. A submicro determination of glucose. Journal of Biological Chemistry , v.181, p.149-151, 1949. Available from: www.jbc.org/content/181/1/149.long>. Accessed: Feb. 03, 2016.
» www.jbc.org/content/181/1/149.long - PERETTI, D.; ANDRIAN, I. F. Feeding and morphological analysis of the digestive tract of four species of fish (Astyanax altiparanae, Parauchenipterus galeatus, Serrasalmus marginatus and Hoplias aff. malabaricus) from the upper Paraná River floodplain, Brazil. Brazilian Journal of Biology, v.68, n.3, p.671-679, 2008. Avaliable from: < Avaliable from: http://www.scielo.br/scielo.php?pid= S1519-69842008000300027&script=sci_arttext >. Accessed: Feb. 01, 2016. doi: 10.1590/S1519-698420080 00300027.
» http://www.scielo.br/scielo.php?pid= S1519-69842008000300027&script=sci_arttext - REYNALTE-TATAJE, D.; ZANIBONI-FILHO, E. Cultivo de piapara, piauçu, piava e piau - gênero Leporinus. In: BALDISSEROTTO, B.; GOMES, L. C. (Eds.). Espécies nativas para piscicultura no Brasil . Santa Maria: UFSM , 2010. p.73-99.
- SABAPATHY, U.; TEO, L. H. A quantitative study of some digestive enzymes in the rabbitfish, Siganus canaliculatus and the Sea bass, Lates calcarifer. Journal of Fish Biology , v.42, p.595-602, 1992. Avaliable from: < Avaliable from: http://onlinelibrary.wiley.com/doi/10.1111/j.1095-8649.1993.tb003 62.x/pdf >. Accessed: Feb. 01, 2016. doi: 10.1016/S0022-474X(00)00016-3.
» https://doi.org/10.1016/S0022-474X(00)00016-3» http://onlinelibrary.wiley.com/doi/10.1111/j.1095-8649.1993.tb003 62.x/pdf - TENGJAROENKUL, B. et al. Distribution of intestinal enzyme activities along the intestinal tract of cultured Nile tilapia, Oreochromis niloticus L. Aquaculture , v.182, p.317-327, 2000. Available from: http://www.thaiscience.info/Article%20for%20ThaiScience/Article/2/Ts-2%20distrib.of%20intestinal%20enzyme%20act.along%20intestinal%20tract%20of%20cultured%20nile%20tilapia,%20oreochromis%20niloticus%20l..pdf>. Accessed: Feb. 01, 2016.
» http://www.thaiscience.info/Article%20for%20ThaiScience/Article/2/Ts-2%20distrib.of%20intestinal%20enzyme%20act.along%20intestinal%20tract%20of%20cultured%20nile%20tilapia,%20oreochromis%20niloticus%20l..pdf
-
1
CR-2016-0113.R1
Publication Dates
-
Publication in this collection
05 Jan 2017 -
Date of issue
2017
History
-
Received
04 Feb 2016 -
Accepted
30 May 2016 -
Reviewed
06 Dec 2016