Abstracts
Information related to the diet of one species always contributes to the knowledge of its bionomy and the functioning of the ecosystem in which the species lives. Therefore, understanding the feeding ecology of one fish population and its ontogenetic, spatial and temporal aspects help to understand the structure of fish assemblages and river communities. Knowledge of this structure is essential for habitat management and biodiversity conservation. The feeding ecology of Deuterodon langei Travassos, 1957 was studied through analyses of diet composition, sharing of resources, feeding strategy and contribution of food items to the width of its niche. The analysis included an assessment of ontogenetic, spatial and seasonal variations. The species was considered omnivorous with great plasticity caused by seasonal variation in food availability throughout the river basin, but mainly through ontogeny. The diet of smaller individuals revealed a predominance of insects and other arthropods, while the diet of larger specimens showed allochthonous plant items as the main components. The low intestinal quotient (IQ) values for the smaller individuals were accounted for their mainly insectivorous diet. The greater relative length of the intestine can account for the greater plasticity of the adult diet, enabling them to use diet items of larger size and more difficult digestion, such as plant items. Therefore, shifts in feeding ecology during growth are confirmed in this omnivorous tropical river fish, strengthening the concept that, when comparing food habits among species, fish size must be taken into account. This study also provides needed information regarding the feeding ecology of fishes intrinsically associated with the Atlantic rainforest, one of the most threatened and biodiverse ecosystems of the planet.
Diet; Characins; Ontogeny; Niche; Atlantic Forest
Informações relacionadas à alimentação de uma espécie contribuem para o conhecimento de sua bionomia e funcionamento do ecossistema no qual ela vive. Desta forma, entender a ecologia alimentar de uma população de peixes e seus aspectos ontogenéticos, temporais e espaciais auxiliam na compreensão da assembléia de peixes e de toda a comunidade de um rio. Reconhecer tal estrutura é fundamental para o manejo e conservação da biodiversidade do habitat. A ecologia alimentar de Deuterodon langei Travassos, 1957 foi analisada por meio da composição da dieta, partilha de recursos, estratégia alimentar e contribuição dos itens alimentares para o nicho alimentar. As análises incluem uma abordagem das variações ontogenéticas, espaciais e temporais. A espécie foi considerada onívora com uma grande plasticidade alimentar causada pela variabilidade sazonal na disponibilidade de recursos e ao longo da bacia hidrográfica, mas principalmente ao longo de sua ontogenia. A dieta dos indivíduos menores revelou uma predominância de insetos e outros artrópodes, enquanto nos indivíduos maiores ocorreu um predomínio de vegetais alóctones. Os baixos valores de Quociente Intestinal (IQ) para os indivíduos menores foram associados a uma dieta predominantemente insetívora. Os maiores comprimentos intestinais estiveram associados com uma maior plasticidade dos adultos, os quais são capazes de utilizar itens maiores e de difícil digestão, como itens de origem vegetal. Neste sentido, mudanças na ecologia alimentar durante o crescimento são confirmadas neste peixe onívoro de rios tropicais, ampliando o conceito de que, quando se compara os hábitos alimentares entre espécies, o tamanho dos peixes examinados deve ser levado em consideração. Este estudo contribui também com informações necessárias sobre a ecologia alimentar de peixes intrinsecamente associados com a Floresta Atlântica, um dos mais biodiversos e ameaçados ecossistemas do planeta.
Ontogenetic, spatial and temporal variations in the feeding ecology of Deuterodon langei Travassos, 1957 (Teleostei: Characidae) in a Neotropical stream from the Atlantic rainforest, southern Brazil
Jean R. S. Vitule; Marcelo R. Braga; José M. R. Aranha
Laboratório de Ecologia de Rios (EcoRios), Departamento de Zoologia, Setor de Ciências Biológicas, Universidade Federal do Paraná, Centro Politécnico, 81531-990 Curitiba, PR, Brazil. biovitule@gmail.com (JRSV)
ABSTRACT
Information related to the diet of one species always contributes to the knowledge of its bionomy and the functioning of the ecosystem in which the species lives. Therefore, understanding the feeding ecology of one fish population and its ontogenetic, spatial and temporal aspects help to understand the structure of fish assemblages and river communities. Knowledge of this structure is essential for habitat management and biodiversity conservation. The feeding ecology of Deuterodon langei Travassos, 1957 was studied through analyses of diet composition, sharing of resources, feeding strategy and contribution of food items to the width of its niche. The analysis included an assessment of ontogenetic, spatial and seasonal variations. The species was considered omnivorous with great plasticity caused by seasonal variation in food availability throughout the river basin, but mainly through ontogeny. The diet of smaller individuals revealed a predominance of insects and other arthropods, while the diet of larger specimens showed allochthonous plant items as the main components. The low intestinal quotient (IQ) values for the smaller individuals were accounted for their mainly insectivorous diet. The greater relative length of the intestine can account for the greater plasticity of the adult diet, enabling them to use diet items of larger size and more difficult digestion, such as plant items. Therefore, shifts in feeding ecology during growth are confirmed in this omnivorous tropical river fish, strengthening the concept that, when comparing food habits among species, fish size must be taken into account. This study also provides needed information regarding the feeding ecology of fishes intrinsically associated with the Atlantic rainforest, one of the most threatened and biodiverse ecosystems of the planet.
Key words: Diet, Characins, Ontogeny, Niche, Atlantic Forest.
RESUMO
Informações relacionadas à alimentação de uma espécie contribuem para o conhecimento de sua bionomia e funcionamento do ecossistema no qual ela vive. Desta forma, entender a ecologia alimentar de uma população de peixes e seus aspectos ontogenéticos, temporais e espaciais auxiliam na compreensão da assembléia de peixes e de toda a comunidade de um rio. Reconhecer tal estrutura é fundamental para o manejo e conservação da biodiversidade do habitat. A ecologia alimentar de Deuterodon langei Travassos, 1957 foi analisada por meio da composição da dieta, partilha de recursos, estratégia alimentar e contribuição dos itens alimentares para o nicho alimentar. As análises incluem uma abordagem das variações ontogenéticas, espaciais e temporais. A espécie foi considerada onívora com uma grande plasticidade alimentar causada pela variabilidade sazonal na disponibilidade de recursos e ao longo da bacia hidrográfica, mas principalmente ao longo de sua ontogenia. A dieta dos indivíduos menores revelou uma predominância de insetos e outros artrópodes, enquanto nos indivíduos maiores ocorreu um predomínio de vegetais alóctones. Os baixos valores de Quociente Intestinal (IQ) para os indivíduos menores foram associados a uma dieta predominantemente insetívora. Os maiores comprimentos intestinais estiveram associados com uma maior plasticidade dos adultos, os quais são capazes de utilizar itens maiores e de difícil digestão, como itens de origem vegetal. Neste sentido, mudanças na ecologia alimentar durante o crescimento são confirmadas neste peixe onívoro de rios tropicais, ampliando o conceito de que, quando se compara os hábitos alimentares entre espécies, o tamanho dos peixes examinados deve ser levado em consideração. Este estudo contribui também com informações necessárias sobre a ecologia alimentar de peixes intrinsecamente associados com a Floresta Atlântica, um dos mais biodiversos e ameaçados ecossistemas do planeta.
Introduction
The Atlantic Forest is one of the richest and most threatened ecosystems of the planet, and is considered one of the main biodiversity hotspots for conservation priorities (Myers et al., 2000). Several rivers flow through this ecosystem, which contain many endemic fish species (Menezes, 1996). The "Serra do Mar" is a 3000-km mountain ridge covered by the Atlantic Forest which extends along the Brazilian coast. Its network of littoral streams has been poorly investigated (Mazzoni & Lobón-Cerviá, 2000). These streams are among the first environments to suffer from anthropic impacts, potentially threatening their fish fauna (Menezes, 1996; Faria & Marques, 1999; Vitule et al., 2006).
Fishes are major components of food webs in tropical rivers (Power, 1990; Winemiller & Jepsen, 1998). Tropical fishes have been demonstrated to be proportionally more diverse in species richness, ecological niches, and trophic specialization (herbivory, detritivory or omnivory) when compared to temperate river fishes (Winemiller & Jepsen, 1998). More than 20% of all known freshwater fish species of the world are found in the Neotropics (Nelson, 1994). The order Characiformes is one of the most diverse, presenting a wide variety of forms and biological or behavioral adaptations (Vazzoler & Menezes, 1992; Nelson, 1994). Within this order, the family Characidae has the largest number of known species and is widely distributed. The old subfamily Tetragonopterinae is considered the most successful characoid taxon, having invaded essentially all biotopes in the Neotropics (Géry, 1977; Nelson, 1994; Lowe-McConnell, 1999). The subfamily Tetragonopterinae does not exist anymore, and most genera within this subfamily are now considered incertae sedis in Characidae (Lima et al., 2003). Characins display a wide range of feeding modes with herbivores, insectivores, carnivores and omnivores, showing great plasticity associated with seasonal, spatial and ontogenetic variations (Sabino & Castro, 1990; Kramer & Bryant, 1995b; Aranha et al.,1998; Winemiller & Jepsen, 1998; Vitule & Aranha, 2002; Vilella et al.,2002; Fogaça et al.,2003; Barreto & Aranha, 2006)
Deuterodon Eigenmann, 1907 (Characiformes, Characidae) is a very important genus with a large distribution in the Atlantic Forest, and is always frequent and abundant within fish assemblages of littoral rivers, mainly in South and Southeastern Brazil. Despite that, there are few studies of its ecology: Sabino & Castro (1990), Aranha et al. (1998), Mazzoni & Petito (1999), Esteves & Lobón-Cerviá (2001), Vitule & Aranha (2002), Fogaça et al. (2003), Barreto & Aranha (2005), Barreto & Aranha (2006), Vitule et al. (2008). Deuterodon langei Travassos, 1957 is popularly called "lambari" and is one of seven currently recognized species of the genus, being endemic and very abundant in coastal streams of Paraná and north of Santa Catarina, southern Brazilian states (Aranha et al.,1998; Lucena & Lucena, 2002; Vitule & Aranha, 2002; Barreto & Aranha, 2005; Vitule et al., 2008).
Deuterodon langei is considered a resident of deep or shallow areas, lotic or lentic waters, and various types of substrates (Aranha et al.,1998; Fogaça et al.,2003; Barreto & Aranha, 2005). Studies on D. langei diet had recently been carried out in littoral streams of Paraná State. The fish has been shown to be omnivorous/herbivorous (Aranha et al.,1998; Barreto & Aranha, 2006) and insectivorous, with a predominance of aquatic insects in its diet (Fogaça et al.,2003). A trend toward omnivory with decrease in animal items and increase in plant items has been shown to occur during ontogeny (Vitule & Aranha, 2002).
All the previous work on D. langei had a local focus, using fish sampled from a single site, which is a major problem in understanding its biology in the whole draining basin, considering that some important differences can be found along the same river (Vannote et al.,1980; Allan, 1995; Mazzoni & Lobón-Cerviá, 2000). Therefore, biological characteristics such as feeding ecology interfere directly with fish population dynamics and constitute basic information. The aim of the present observational field study was to analyze the ontogenetic, spatial and temporal variations in the feeding ecology of D. langei in a Neotropical stream of the Atlantic Forest. Understanding the role that this important fish species plays in structuring fish assemblages and all the river community through their feeding ecology is essential for habitat management and biodiversity conservation (Lowe-McConnell, 1999), especially in such important but threatened and fragile Neotropical ecosystems.
Material and Methods
Study area. The study was conducted in rio Ribeirão basin (25°36'S; 48°37'W), east side of the Serra do Mar, sub-basin of Paranaguá bay, Paranaguá city, which is a portion of Atlantic rainforest in Paraná State, Brazil. The spring is located at Serra da Prata, 766 m above sea level. It is a short basin (< 40 km) that drains directly into the Paranaguá bay (Braga et al., 2008; Vitule et al., 2008).
This region has a tropical, sub-equatorial, super humid climate without dry season and frost. In general, annual rainfall rates are above 1000 mm, and average annual temperatures range between 17º C and 21º C (IAPAR, 1978; Maack, 1981). Rainfalls are distributed differently during the year; January and February were the rainiest months, and July was the driest month during the investigation period (Fig. 1).
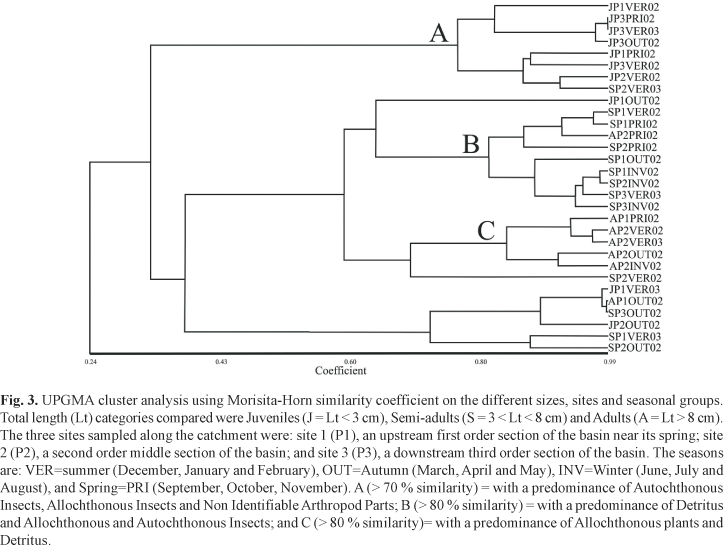
Three sample sites were established: site 1 (P1), an upstream first order section of the basin near its spring (25º35'17''S; 48º38'01''W); site 2 (P2), a second order middle section of the basin (25º36'02''S; 48º37'19''W), and site 3 (P3), a downstream third order section of the basin (25º35'21''S; 48º36'40''W) (Fig. 2). All sample sites were less than 30 m above sea level.
Like other coastal clear water streams in the Atlantic rainforest, rio Ribeirão is dependent on the high rainfall of the area; it displays characteristics of rain waters, neutral pH and low level of nutrients. In the rainy season (summer), the river is subject to flash floods, significantly increasing water volume and flow which occurs suddenly, causing great alterations in physical and biological parameters due to the water drag (Por, 1992).
In general, sample sections were 2 - 5 m wide, and the depths were between 10 cm deep in the shallower margin, to 1.80 m deep in central deeper portions. The bottom was mainly composed of sand, with some presence of clay, leaves, gravel, rocks, and tree trunks and branches. The vegetation of the river bank is composed partially of submerged shrubs, trees and grass which were usually covered with an algal layer. Aquatic macrophytes were rare in the area studied. Most of the sampling sites were very shaded, and there were some beach formations. The main characteristics of each of the three sampling sites are presented in Table 1.
Fish sampling and data analyses. Fish were sampled monthly from February of 2002 to February 2003. The samples were obtained using sieves, a small trawl net of 1.30 x 1.40 m with 2-mm mesh, and five gillnets of 1.50 x 5 m, three with 15-mm mesh and two with 20-mm mesh between consecutive knots. To gain a better knowledge of feeding tactics and strategies utilized by the species, some sporadic in situ observations were made using free diving techniques.
Samples were always taken during daytime, since personal underwater observations and information from the literature (Fogaça et al., 2003; Sabino & Castro, 1990; Barreto & Aranha, 2005) indicated that D. langei were active during daytime. Still, in the field, specimens were immersed in 10% formalin with the objective of stopping the digestive process, and after 48 h they were transferred to 70% alcohol for better conservation and to facilitate handling during dissection. In the laboratory, Lt (total length to 0.01 cm) was measured, and fish were eviscerated. Voucher specimens were deposited in the Museu de História Natural Capão da Imbuia, Curitiba, Brazil: MHNCI 11698.
After evisceration, the intestines were removed and measured (total length to 0.01 cm) to determine the intestinal quotient (IQ = intestine length / total body length) sensu Knöppel (1970) and Zavala-Camin (1996). The IQ values were statistically evaluated by the Kruskal-Wallis, nonparametric test to determine possible ontogenetic variations in putative response in feeding behavior. Stomach fullness was estimated and four categories were created: full (> 70% of the stomach filled up), almost full (50 to 70% of the stomach filled up), almost empty (< 50% of the stomach filled up) and empty. Only full stomachs were used in diet analysis. Stomach contents were observed under a stereomicroscope (here called macroscopic analyses) and light microscope (here called microscopic analyses). Items were identified to the lowest possible taxonomic level, with the aid of specialized bibliography. When necessary, specialists were consulted.
Based on previous studies (Vitule & Aranha, 2002) of ontogenetic analyses, the fish were grouped into three total length (Lt) categories: juveniles (Lt < 3 cm); semi-adults(3 < Lt < 8 cm) and adults(Lt > 8 cm). For temporal analyses, the samples were seasonally grouped, with December, January and February representing summer, March, April and May representing autumn, June, July and August representing winter, and September, October and November representing spring.
For analysis purposes, food resources were grouped into taxonomic and/or ecological categories: microscopic filamentous algae, protozoa, diatoms, unicellular chlorophytes, cyanophytes, allochthonous plants (leaf fragments, flowers, stems), autochthonous plants (macroscopic algae and bryophytes), fruits and seeds, non identifiable arthropod parts, autochthonous insects (semi-aquatic and aquatic insects), allochthonous insects (terrestrial insects), other allochthonous arthropods (arachnids, amphipods, and terrestrial arthropods), other autochthonous arthropods (crustaceans and others), detritus (non identifiable organic matter), remaining items (non representative, low frequency items and/or items that do not fit in any of the above categories such as fish scales and eggs).
For the diet analysis, the methods of Frequency of Occurrence (FO) and Percentage Composition (PC) (Hynes, 1950) were employed. The method of Points (Hynes, 1950; Hyslop, 1980) was adapted in a such way that the relative amount of the macroscopic item was estimated by the area it covered on a grid paper. The abundance of each item corresponded to a score proportional to its area in relation to the area covered by other items. The total area of food items was considered the total volume (100%). Calculations of the volumetric proportion of each food item were made according to the formula:
where Pix is the proportion by volume of item i in the gut of individual x and Nj is the number of individuals of the species j.
The feeding index (IAi) was used to ponder the results obtained by the methods of frequency of occurrence and the method of points (relative amounts or volume) of each item (Kawakami & Vazzoler, 1980):
where Fi is the frequency of occurrence of item i (%) and Vi is the relative volume of item i (% of total).
The macroscopic (observed under stereomicroscope, 25 x) and microscopic (observed under light microscope, 400 x) analyses were performed separately. For microscopic analyses, a relative abundance series of degrees was created, which consisted in the counting of 36 fields of 2 mm2 on a 24x24 mm coverslip, using a light microscope at 400 x. Each item was scored according to the number of fields where it was present: items that were present in more than 26 fields were considered very abundant (score = 4); items that appeared on 18 to 25 fields were considered abundant (score = 3); items that were present in 8 to 17 fields were considered little abundant (score = 2); items that were present in 1 to 7 fields were considered rare (score = 1); and finally, items with no presence were called absent (score = 0).
The analysis of macroscopic and microscopic data performed using different methods (Frequency of Occurrence, Percentage Composition and Feeding Index) produced rather similar results. Thus, only the feeding index IAi was used in subsequent statistical analyses.
The dietary data from index IAi were used to construct a similarity matrix, using the Morisita-Horn similarity coefficient, which was further processed in an agglomerative cluster analysis through the unweighted pair group method, UPGMA - Euclidean distance, using the NTsys v5.0 package. The differences in size, sites and seasonal groups were defined by Non-Metric Multidimensional Scaling (MDS) and one-way ANOSIM, using the PRIMER-E v5.0 package (Clarke & Gorley, 2001; Clarke & Warwick, 2001).
The statistical comparison of diets was made using the χ2 test (a = 0.05), "Comparison with more than two proportions" (Zar, 1999). The H0 hypothesis corresponded to the same item proportions for distinct samples (sample sites, length categories, and seasons). For samples where H0 was rejected, the test of "Comparison between two proportions" (Zar, 1999) was applied to detect significant variations in diet categories (P<0.05). Only significant variations were presented in the results.
Results
Ontogenetic and spatial foraging patterns in macroscopic analyses. The stomach contents of 260 fish were analyzed, where 235 were considered full, 19 almost full, 6 almost empty and no empty stomachs were found. The comparison of Intestinal Quotient (IQ) values demonstrated significant differences (P<0.0001) between the three length categories; Juveniles (J) showed an average of 0.72 for the IQ value (n=43); Semi-adults (S) showed an IQ of 0.89 (n=124); and Adults(A) had an average IQ of 1.06 (n=93).
Fifty-three macroscopic diet items were , and they were grouped into 10 ecological and/or taxonomic categories, as described in Table 2. The macroscopic diet of the juvenile size class demonstrated a predominance of autochthonous insects, allochthonous insects, and non identifiable arthropod parts. The semi-adults showed a predominance of detritus and allochthonous and autochthonous insects. For adults, there was a predominance of allochthonous plant parts and detritus (Table 2).
The diet analysis of the three length categories showed significant differences (P<0.05) (χ2: J x S = 48.87, J x A = 114.63, S x A = 51.87). The main differences when comparing diet between the three length categories were a greater proportion of autochthonous insects and non identifiable arthropod parts in the juvenile category, greater role of detritus and autochthonous plants in the semi-adults, and greater contribution of allochthonous plants, seeds and fruits in the adult category.
The diet analysis of the three length categories showed differences (P<0.05) along the sample sites (χ2: JP1 x JP2 = 45.83, JP1 x JP3 =47.66, JP2 x JP3= 38.72, SP1 x SP2 = 21.29, SP2 x SP3 = 42.34, AP1 x AP2 = 56.20, AP1 x AP3 = 86.76, AP2 x AP3 = 51.87) and revealed the following pattern. For the juvenile category, there was a greater inclusion of allochthonous and autochthonous insects and detritus at sample point 1, allochthonous and autochthonous insects at sample point 2, and allochthonous and autochthonous insects, and non identifiable arthropod parts at sample point 3. For the semi-adults, there was a predominance of detritus and allochthonous insects at sample point 1, detritus and autochthonous insects at sample point 2, and allochthonous insects and detritus at sample point 3. For the adults, there was a great contribution of allochthonous insects, allochthonous plants, seeds and fruits at sample point 1, allochthonous plant parts and detritus at sample point 2, and allochthonous insects, non identifiable arthropod parts and allochthonous plants at sample point 3 (Table 3).
Seasonal versus ontogenetic patterns in macroscopic analyses. A seasonal evaluation of the diet of the three length categories showed statistical (P<0.05) differences (χ2: JV2 x JO2 = 73.69, JV2 x JI2 = 61.816, JV2 x JP2 = 31.79, JV2 x JV3 = 49.46, JO2 x JI2 = 22.98, JO2 x JP2 = 38.57, JO2 x JV3 = 32.61, JI2 x JP2 = 21.89, JI2 x JV3 = 27.41, JP2 x JV3 = 31.91, SV2 x SO2 = 69.53, SV2 x SI2 = 50.53, SV2 x SP2 = 26.45, SV2 x SV3 = 84.28, SO2 x SI2 = 20.41, SO2 x SP2 = 32.43, SO2 x SV3 = 34.84, SI2 x SP2 = 32.64, SI2 x SV3 = 41.71, SP2 x SV3 = 42.51, AV2 x AO2 = 57.05, AV2 x AI2 = 30.75, AV2 x AP2 = 20.99, AV2 x AV3 = 41.54, AO2 x AP2 = 32.68, AO2 x AV3 = 28.95) and revealed the following pattern. Diet showed a greater involvement of autochthonous insects during the whole study period in the juvenile category. For the semi-adults, detritus predominated in summer and winter of 2002, and allochthonous insects prevailed in the autumn, while there was no predominance of any item category both in the spring of 2002 and in the summer of 2003. In the adult category, allochthonous plants predominated in the summer and spring of 2002, and in the summer of 2003; allochthonous plants and detritus predominated in the winter of 2002, and allochthonous insects and detritus prevailed in the autumn of 2002 (Table 3).
An analysis of similarity revealed that the major shifts in diet occurred due to ontogeny rather than seasonality. The three primordial groups in cluster group A with a predominance of autochthonous insects, allochthonous insects and non identifiable arthropod parts; B with a predominance of detritus, allochthonous and autochthonous insects; and C with a predominance of allochthonous plants and detritus were formed due to ontogenetic developmental state, thus showing that despite the importance of all other variations, ontogeny was the most important factor (Fig. 3). There was a gradual shift from insectivory to herbivory during development, as shown by MDS analysis (Figs. 4, 5 and 6) and one-way ANOSIM using the ontogeny factor (Table 4). This confirmed a predominance of autochthonous insects, allochthonous insects and non identifiable arthropod parts in juveniles; a predominance of detritus, allochthonous and autochthonous insects in semi-adults; and a predominance of allochthonous plants and detritus in adults, with diet depending on the ontogenetic developmental state .
Ontogenetic and spatial foraging patterns in microscopic analyses. Microscopic analyses of the diet revealed the presence of 20 items grouped in 11 categories, as described in Table 5. All length categories showed a predominance of diatoms (Bacylariophyta) and cyanophyte algae. The analysis of the diet of the length categories along the sample sites showed that in the juvenile category there was a predominance of diatoms, filamentous algae and unicellular chlorophytes at sample point 1; diatoms and cyanophyte algae at sample point 2 and 3. Semi-adults had greater amounts of diatoms and cyanophyte algae at the three sample sites. Adults showed a greater proportion of diatoms and cyanophyte algae at sample sites 1 and 3, and sample point 2 showed a homogeneous distribution of filamentous algae, diatoms, cyanophyte algae and unicellular chlorophyte algae (Table 5).
Seasonal analyses of the microscopic diet demonstrated that in the juvenile category there was a greater role of diatoms in the summer of 2002, autumn of 2002 and summer of 2003; cyanophyte algae predominated in the winter of 2002; and autochthonous micro-arthropods dominated in the spring of 2002. For the classes of semi-adults and adults, diatoms predominated during the whole study period, but an increase in the contribution by cyanophyte algae in the winter of 2002 for both categories (semi-adults and adults) was noticeable (Table 6).
The comparison of the microscopic items of the diet between the three length categories demonstrated significant differences (P<0.05) between the diets of juveniles and adults(χ2: J x A = 22.16). The cause of this difference was the greater proportion of autochthonous micro-arthropods in the juvenile category. An association between abundance and occurrence of detritus and/or macroscopic algae and microscopic algae was observed.
The analyses of the diet in the juvenile category at the three sample sites demonstrated significant differences (P<0.05) between all sample sites (χ2: JP1 x JP2 = 44.17, JP1 x JP3 = 49.56, JP2 x JP3 = 54.16). There was a greater contribution by filamentous algae at sample point 1, diatoms at sample point 2, and cyanophyte algae at sample point 3. Semi-adults showed differences (P<0.05) between sample points 1 and 3 (χ2: SP1 x SP3 = 20.02), caused by a greater proportion of filamentous algae at sample point 1. Adults displayed differences between sample sites 1, 2 and 3 (χ2: AP1 x AP2 = 25.65, AP2 x AP3 = 41.30). Sample sites 1 and 3 showed a greater contribution by diatoms, and sample point 2 was distinct showing a greater proportion of filamentous algae and non identifiable arthropod parts.
Seasonal versus ontogenetic patterns in microscopic analyses. A seasonal comparison of the microscopic diet of the Juvenile category revealed differences (P<0.05) between all seasons (χ2: JV2 x JO2 = 77.29, JV2 x JI2 = 111.11, JV2 x JP2 = 28.98, JV2 x JV3 = 24.76, JO2 x JI2 = 66.97, JO2 x JP2 = 119.94, JO2 x JV3 = 63.66, JI2 x JI2 = 148.38, JI2 x JV3 = 97.87, JP2 x JV3 = 26.78). There was a greater involvement of filamentous algae and unicellular chlorophytes in the autumn of 2002, cyanophyte algae in the winter of 2002, autochthonous micro-arthropods and nematodes in the spring of 2002, and diatoms in the summer of 2003. Semi-adults showed differences (P<0.05) between the winter of 2002 and the summer of 2003 (χ2: SI2 x SV3 = 23.57). There was a greater proportion of cyanophyte algae in the winter of 2002, and more abundant plant remains predominated in the summer of 2003. Adults showed differences (P<0.05) between summer and autumn of 2002 and summer and winter of 2002 (χ2: AV2 x AO2 = 17.13, AV2 x AI2 = 18.96). Non identifiable arthropod parts predominated in the summer of 2002, and cyanophyte algae prevailed in the 2002 winter.
Discussion
The richness of macroscopic and microscopic items found in this study indicates the opportunistic character of this species. This pattern is usual among fresh-water fish and supports the idea of Lowe-McConnell (1999) that most Neotropical fish have wide variable diets. The fact that almost all stomachs analyzed were full demonstrates an intense feeding activity during the sample period and is evidence of the efficiency of the methods employed. As the digestive process was paralyzed almost immediately, the amount of non identifiable digested material was reduced, thus increasing the reliability of the later analyses (Hynes, 1950; Amundsen et al.,1996). Underwater observations and literature data (e.g., Sabino & Castro, 1990) confirm a diurnal feeding activity for D. langei. It could be observed that individuals retreat to shelters during the night, probably to avoid nocturnal predators such as Hoplias malabaricus (Bloch, 1794), Rhamdia quelen (Quoy & Gaimard, 1824) and Synbranchus marmoratus (Bloch, 1795).
Most of the diet studies using species of the genus Deuterodon have been conducted employing only qualitative methods, although the importance of quantitative or semi-quantitative abundance estimates has always been emphasized by many authors (e.g., Hyslop, 1980; Esteves & Aranha, 1999). Considering that quantitative analyses of microscopic items are very difficult (e.g., Aranha, 1993), a semi-quantitative analysis of microscopic items was chosen, allowing greater confidence in the final results.
The Intestinal Quotient (IQ) can be a very useful method to evaluate inter-specific or ontogenetic variations that reflect the feeding behavior (Knöppel, 1970; Zavala-Camim, 1996). The variations found in the D. langei IQ values for the three length categories were marked in this work, differing from what Vitule & Aranha (2002) found for the same species in another study. That difference is probably the result of a smaller sample size and a greater number of length classes employed in the previous study. In D. iguape, the smaller IQ values of juveniles (2.8 to 4.1 cm standard length) have been related to a diet mainly composed of insects (Sabino & Castro, 1990). Similarly, the smaller IQ values found for juveniles of D. langei in this study may be associated with a mainly insectivorous diet in the rio Ribeirão basin. On the other hand, the greater relative length of the intestine can explain the greater plasticity of the adult diet, enabling them to use diet items of greater size and more difficult digestion such as plant items with thick cellulose cell walls (Knöppel, 1970; Sabino & Castro, 1990; Zavala-Camim, 1996; Amundsen et al., 2003). In fact, many tropical stream fishes show an ontogenetic increase in their gut length relative to body length; and those that consume relatively more plant items show disproportional ontogenetic growing, with high rates of increase in gut length (Kramer & Bryant, 1995b). In fishes from forest streams in Panama, the ontogenetic allometry in intestine length was related to increasing body size and absorptive capacity of the digestive tract (Kramer & Bryant, 1995a). In general, herbivorous fish ingest food resources with morphological and chemical defenses, encased in largely indigestible fibrous cell walls, which are thereby considered nutrient poor, and thus, ontogenetic increases in gut length are well known in these fishes. Along this line, many fishes appear to begin life as carnivores or omnivores and shift to a more herbivorous diet as they grow (e.g., Sabino & Castro, 1990; Zavala-Camim, 1996). Their gut lengths usually increase accordingly (Kramer & Bryant, 1995a,b; German & Horn, 2006).
Besides this morphological relationship between gut length and diet, behavioral aspects can also be considered. Apparently, ontogeny and phylogeny more than diet influence fish gut dimensions, thus favoring genetic adaptation over phenotypic plasticity as the major force acting on the digestive system (Germam & Horn, 2006). The greater necessity of protein for juveniles relates to a shift from insectivore to omnivore during ontogenetic development. As fish grow they can explore new food sources contributing to a more omnivorous diet (Sabino & Castro, 1990; Vitule & Aranha, 2002, Barreto & Aranha, 2006). These results confirm that ontogenetic dietary shifts occur widely among omnivorous tropical stream fishes (Kramer & Bryant, 1995b). Trends in fish diet have also been detected in other tropical and temperate zones (Amundsen et al., 2003; Katunzi et al., 2006; Rezsu & Specziár, 2006; Alcaraz & García-Berthou, 2007). In this sense, shifts in the feeding ecology during growth strengthen the notion that species may not be an adequate unit when comparing food habits among species that occupy the same area during ontogeny (Stoner & Livingston, 1984; Livingston,1988).
The great abundance of allochthonous plant and animal items in the diet of D. langei reflects the importance of the conservation of river-margin vegetation and nearby forest. The presence of fruits and seeds in the diet reinforces the importance of vegetation to the fish diet (Goulding, 1980; Araujo-Lima et al.,1995). Vilella et al. (2002) suggested that related characins of the genus Astyanax can have an active role in dispersion of seeds, and such role cannot be dismissed for D. langei.
Diet variations throughout sample sites demonstrated a predominance of animal items downstream, and an increasing inclusion of other items, mainly detritus and plant items in the more upstream sample sites. These differences were not due to sample size of length classes in each sample point, because the differences persisted even when the analyses were performed separately for each length class. These differences can be related to availability of feeding resources. Tropical rivers and streams have large spatial heterogeneity that range from microhabitats to landscapes across a continuum of spatial scales. In fact, succession of organisms in rivers and streams can be the cause of spatial and seasonal variation of occurrence and abundance (Allan, 1995; Allan et al., 1997; Winemiller & Jepsen, 1998). Fish populations and community dynamics show many responses to physical habitat disturbance and seasonal changes in habitat quality and quantity (e.g., Harrell, 1978; Winemiller & Jepsen, 1998).
Tropical fishes show the most diverse morphological and physiological adaptations to dry-wet seasons. A notable example is that reproductive seasons coincide with rising water levels, allowing juveniles to feed in the wider rich resources of floodplains (Lowe-McConnell, 1999). There are other examples of fish adaptations to dry-wet seasons in feeding ecology (e.g., Winemiller & Jepsen, 1998; Katunzi et al., 2006). In D. langei, seasonal diet variation revealed a pattern where there was a greater participation of detritus during the dry season (autumn/winter), and this was true for all length classes. This pattern can be related to lower availability of other feeding resources as the environment retracts due to the lack of rain during that period. In tropical streams, omnivorous fish (e.g., Astyanax fasciatus) and flash floods have a strong role in structuring aquatic insects and benthic algal assemblages (Pringle & Hamazaki, 1997).
Considering microscopic items, the diet showed little variation between sample sites and seasons and diatoms were always predominant. The most representative variation was detected in the dry season (autumn/winter) when a greater presence of blue-green algae (cyanophytes) occurred in the diet, probably because it was more available in the dry period. In the dry period there are no flash floods or storms that may result in decreases in total biovolume of algae diatom biovolume and algal taxon biodiversity. In small tropical streams, storms with high discharge events in the absence of omnivorous fishes would play an important role in structuring benthic algal assemblages, resulting in extreme fluctuations in algal biomass. In this sense, omnivorous fishes show a key role in maintaining the stability of benthic algal assemblages and their resistance to flash flood or storms (Pringle & Hamazaki, 1997). Many successions of organisms in rivers and streams have seasonal causes (e.g. Allan, 1995; Allan et al.,1997; Winemiller & Jepsen, 1998). On the other hand, risk-sensitive foraging can be a general phenomenon that influences food web dynamics at spatial scales in tropical streams (Winemiller & Jepsen, 1998). Moreover, the presence of an association between abundance and occurrence of detritus and/or macroscopic algae and microscopic algae was observed, suggesting that the greater the proportion of detritus and/or macroscopic algae, the greater will be the proportion of microscopic algae. In this sense, the abundance of microscopic algae can be related to the presence of detritus and/or macroscopic algae in benthic food resources such as periphyton, which increase with gradual drying.
In general, D. langei is an omnivorous and opportunistic species with great diet flexibility. However, it is noted that small individuals show a trend toward insectivory and that as fish develop the feeding spectrum widens, mainly with the addition of plant items of more difficult digestibility and lower protein content. The differences observed in the Intestinal Quotient during ontogenetic development can be related to the increase in plant items in larger individuals. Despite the importance of other diet variations observed, the variation of diet through ontogenetic development was the most prominent aspect for the population dynamics of this species. Most diet variations found between sample sites and seasons were the result of fluctuations in availability of resources. Seasonal diet variation revealed patterns where there is a greater role of detritus and algae during the dry season (autumn/winter), and this held true for all length classes. That pattern can be related to lower availability of other feeding resources as the environment retracts due to the lack of rain during that period; however, more studies on the availability of feeding resources are essential to corroborate this hypothesis.
Ontogenetic shifts were the most prominent occurrence in the feeding ecology of the species. These shifts in feeding ecology during growth confirmed in an Atlantic Forest stream fish the concept that species may not be an adequate unit when comparing food habits without considering length. This study also contributed needed information regarding the feeding ecology of endemic tropical fishes intrinsically associated with one of the most threatened ecosystems of the planet.
Acknowledgments
We thank Carolina Arruda de Oliveira Freire, Viviane Prodocimo, José Francisco de Oliveira Neto and Flávia F. Gazola da Silva for revision of the text and useful suggestions regarding the manuscript. The Brazilian Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) provided financial support.
Literature Cited
Accepted May 2008
Published June 28, 2008
- Alcaraz C. & E. García-Berthou. 2007. Food of an endangered cyprinodont (Aphanius iberus): ontogenetic diet shift and prey electivity. Environmental Biology of Fishes, 78: 93-207.
- Allan, J. D. 1995. Stream Ecology: Structure and Function of Running Waters. London, Chapman & Hall, 388p.
- Allan, J. D., D. L. Erikson & J. Fay. 1997. The influence of catchment land use on stream integrity across multiple spatial scales. Freshwater Biology, 37: 149-161.
- Amundsen, P. A., H. M. Gabler & F. L. Staldvik. 1996. A new approach to graphical analysis of feeding strategy from stomach contents data - modification of the Costello (1990) method. Journal of Fish Biology, 48: 607-614.
- Amundsen, P., T. Bohn, O. A. Popova, F. J. Staldvik, Y. S. Reshetnikov, N. A. Kashulin & A. A Lukin. 2003. Ontogenetic niche shifts and resource partitioning in a subarctic piscivore fish guild. Hydrobiologia, 497: 109-119.
- Aranha, J. M. R. 1993. Método para análise quantitativa de algas e outros itens microscópicos de alimentação de peixes. Acta Biológica Paranaense, 22: 71-76.
- Aranha, J. M. R., D. F. Takeuti & T. Yoshimura. 1998. Habitat use and food partitioning of the fishes in a coastal stream of Atlantic Forest, Brazil. Revista de Biología Tropical, 46: 951-959.
- Araújo-Lima, C. A. R. M., A. A. Agostinho & N. M. Fabré. 1995. Trophic aspects of fish communities in Brazilian rivers and reservoirs. Pp. 105-136. In: Tundisi, J. G., C. E. M. Bicudo & T. Matsumura-Tundisi (Eds.). Limnology in Brazil. Rio de Janeiro ABC/SBL, 376p.
- Barreto, A. P. & J. M. R. Aranha. 2005. Assembléia de peixes de um riacho da Floresta Atlântica: composição e distribuição espacial (Guaraqueçaba, Paraná, Brasil). Acta Scientiarum Biological Science, 27: 153-160.
- Barreto, A. P. & J. M. R. Aranha. 2006. Alimentação de quatro espécies de Characiformes de um riacho da Floresta Atlântica, Guaraqueçaba, Paraná, Brasil. Revista Brasileira de Zoologia, 23: 779-788.
- Braga, M. R., J. R. S. Vitule & J. M. R. Aranha. 2008. Reproduction Period of Mimagoniates microlepis (Steindachner, 1876) (Characidae, Glandulocaudinae) from an Atlantic Forest Stream in Southern Brazil. Brazilian Archives of Biology and Technology, 51: 345-351.
- Clarke, K. R. & R. N. Gorley. 2001. PRIMER v5: user manual/tutorial. Plymouth: PRIMER-E, Plymouth, UK, 99p.
- Clarke K. R. & R. M Warwick. 2001. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation. 2nd edition: PRIMER-E, Plymouth, UK, 172p.
- Esteves, K. E. & J. M. R. Aranha. 1999. Ecologia trófica de peixes de riachos. Pp. 157-182. In: Caramaschi, E. P., R. Mazzoni, P. R. Peres-Neto. (Eds.). Ecologia de Peixes de Riachos. Série Oecologia Beasiliensis, Rio de Janeiro, PPGE-UFRJ, 353p.
- Esteves, K.E. & J. Lobón-Cerviá. 2001. Composition and trophic structure of a clear water Atlantic rainforest stream in southeastern Brazil. Environmental Biology of Fishes, 62: 429-440.
- Faria, A. P. & J. S. Marques. 1999. O desaparecimento dos pequenos rios brasileiros. Ciência Hoje, 148: 56-61.
- Fogaça, F. N. O., J. M. R. Aranha & M. de L. P. Esper. 2003. Ictiofauna do Rio do Quebra (Antonina, PR, Brasil): ocupação espacial e hábito alimentar. Interciencia, 28: 168-173.
- German, D. P. & M. H. Horn. 2006. Gut length and mass in herbivorous and carnivorous prickleback fishes (Teleostei: Stichaeidae): ontogenetic, dietary, and phylogenetic effects. Marine Biology, 148: 1123-1134.
- Géry, J. 1977. Characoids of the world. Neptune City, T.F.H. Publications, 672p.
- Goulding, M. 1980. The fishes and the forest. University of California Press, Berkeley, 280p.
- Harrell, H. L. 1978. Response of the Devil´s river (Texas) fish community to flooding. Copeia, 1978(1): 60-68.
- Hynes, H. B. N. 1950. The food of fresh-water stiklebacks (Gasterosteus aculeatus and Pygosteus pungitius), with a review of methods used in studies of the food fishes. Journal of Animal Ecology, 19: 36-58.
- Hyslop, E. J. 1980. Stomach contents analysis - a review of methods and their application. Journal of Fish Biology, 17: 411-429.
- IAPAR. 1978. Cartas climáticas básicas do Estado do Paraná. Instituto Agronômico do Paraná, Londrina, 41p.
- Katunzi, E. F. B., W. L. T. Van Densen, J. H. Wanink & F. Witte. 2006. Spatial and seasonal patterns in the feeding habits of juvenile Lates niloticus (L.), in the Mwanza Gulf of Lake Victoria, Hydrobiologia, 568: 121-133.
- Kawakami, E. & G. Vazzoler. 1980. Método gráfico e estimativa de índice alimentar aplicado no estudo de alimentação de peixes. Boletim do Instituto Oceanográfico São Paulo, 29: 205-207.
- Knöppel, H. A. 1970. Food of central Amazonian fishes: contribution to the nutrient-ecology of Amazonian rain forest streams. Amazoniana, 2: 257-352.
- Kramer, D. L. & M. J. Bryant. 1995a. Intestine length in the fishes of a tropical stream: 1. Ontogenetic allometry. Environmental Biology of Fishes, 42: 115-127.
- Kramer, D. L. & M. J. Bryant. 1995b. Intestine length in the fishes of a tropical stream: 2. Relationships to diet: the long and short of a convoluted issue. Environmental Biology of Fishes, 42: 129-141.
- Lima, F. C. T., L. R. Malabarba, P. A. Buckup, J. F. Pezzi da Silva, R. P. Vari, A. Harold, R. Benine, O. T. Oyakawa, C. S. Pavanelli, N. A. Menezes, C. A. S. Lucena, M. C. S. L. Malabarba, Z. M. S. Lucena, R. E. Reis, F. Langeani, L. Cassati & V. A. Bertaco. 2003. Genera Incertae Sedis in Characidae. Pp. 106-168. In: R. E. Reis, S. O. Kullander & C. J. Ferraris, Jr. (Eds.). Checklist of the Freshwater Fishes of South and Central America. Porto Alegre, EDIPUCRS, 729p.
- Livingston, R. J. 1988. Inadequacy of species-level designations for ecological studies of coastal migratory fishes. Environmental Biology of Fishes, 22: 225-234.
- Lowe-McConnell, R. H. 1999. Estudos ecológicos de comunidades de peixes tropicais. São Paulo, EDUSP, 534p.
- Lucena, C. A. S. & Z. M. S. Lucena. 2002. Redefinição do gênero Deuterodon Eigenmann, 1907 (Ostariophysi: Characiformes: Characidae). Comunicações do Museu de Ciências e Tecnología da PUCRS, 15: 113-135.
- Maack, R. 1981. Geografia física do Estado do Paraná. Rio de Janeiro, J. Olympio, 450p.
- Mazzoni, R. & J. Lobón-Cerviá. 2000. Longitudinal structure, density and production rates of a neotropical stream fish assemblage: the river Ubatiba in the Serra do Mar, southeast Brazil. Ecography, 23: 588-602.
- Mazzoni, R. & J. Petito. 1999. Reproductive biology of a Tetragonopterinae (Osteichthyes, Characidae) of the Ubatiba fluvial system, Maricá-RJ. Brazilian Archives of Biology and Technology, 42: 455-461.
- Menezes, N. A. 1996. Padrões de distribuição da biodiversidade da mata atlântica do sul e sudeste brasileiro: peixes de água doce. In: Workshop "Padrões de biodiversidade da mata atlântica do sudeste e sul do Brasil". Base de dados tropical, Campinas: http://www.bdt.org.br/workshop/mata.atlantica Cited 23 jan 2006
- Myers, N., R. A. Mittermeier, C. G. Mittermeier, G. A. Fonseca & J. Kent. 2000. Biodiversity hotspots for conservation priorities. Nature, 403: 853-858.
- Nelson, J. S. 1994. Fishes of the world. New York, John Wiley & Sons Inc., 600p.
- Por, F. D. 1992. Sooretama, the Atlantic rain forest of Brazil. The Hague, SPB Academic Publishing, 130p.
- Power, M. E. 1990. Effects of fish in river food webs. Science, 250: 811-814.
- Pringle, C. M. & T. Hamazaki. 1997. Effects of fishes on algal response to storms in a tropical stream. Ecology, 78: 2432-2442.
- Rezsu, E. & A. Specziár. 2006. Ontogenetic diet profiles and size-dependent diet partitioning of ruffe Gymnocephalus cernuus, perch Perca fluviatilis and pumpkinseed Lepomis gibbosus in Lake Balaton. Ecology of Freshwater Fish, 15: 339-349.
- Sabino, J & R. C. Castro. 1990. Alimentação, período de atividade e distribuição espacial dos peixes de um riacho de floresta atlântica (sudeste do Brasil). Revista Brasileira de Biologia, 50: 23-36.
- Stoner, A. W. & R. J. Livingston. 1984. Ontogenetic patterns in diet and feeding morphology in sympatric sparid fishes from seagrass meadows. Copeia, 1984(1): 174-187.
- Vannote, R. L., G. W. Minshal, K. W. Cummins, K. W. Sedell & C. E. Cushing. 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences, 37: 130-137.
- Vazzoler, A. E. A. de M. & N. A. Menezes. 1992. Síntese de conhecimento sobre o comportamento reproductivo dos Characiformes da América do Sul (Teleostei: Characiformes). Revista Brasileira de Biologia, 52: 627-540.
- Vilella, F. S., F. G. Becker & S. M. Hartz. 2002. Diet of Astyanax species (Teleostei, Characidae) in an Atlantic Forest River in Southern Brazil. Brazilian Archives of Biology and Tecnology, 45: 223-232.
- Vitule, J. R. S. & J. M. R. Aranha. 2002. Ecologia alimentar do lambari, Deuterodon langei Travassos, 1957 (Characidae, Tetragonopterinae), de diferentes tamanhos em um riacho da Floresta Atlântica, Paraná (Brasil). Acta Biológica Paranaense, 31: 137-150.
- Vitule, J. R. S., S. C. Umbria & J. M. R. Aranha. 2006. Introduction of the African catfish Clarias gariepinus (Burchell, 1822) into Southern Brazil. Biological Invasions, 8: 677 - 681.
- Vitule, J. R. S., M. R. Braga & J. M. R. Aranha. (in press). Population Structure and Reproduction of Deuterodon langei Travassos, 1957 (Teleostei, Characidae) in a Neotropical Stream of Atlantic Forest, Southern Brazil. Brazilian Archives of Biology and Technology, 2008.
- Winemiller, K. O. & D. B. Jepsen. 1998. Effects of seasonality and fish movement on tropical river food webs. Journal of Fish Biology, 53: 267-296.
- Zar, J. H. 1999. Biostatistical analysis. New Jersey, Prentice Hall, 663p.
- Zavala-Camim, L. A. 1996. Introdução aos estudos sobre alimentação natural em peixes. Maringá, EDUEM, 129p.
Publication Dates
-
Publication in this collection
28 July 2008 -
Date of issue
2008
History
-
Accepted
28 June 2008 -
Received
May 2008