Abstract
Leishmaniases are a group of diseases of zoonotic importance caused by over 20 species of protozoa of the genus Leishmania, in which domestic dogs are considered to be the main reservoir for the disease. However, the involvement of other vertebrates as reservoirs for these parasites has also been investigated. Therefore, the objective of the present study was to carry out a systematic review with meta-analysis on occurrences of leishmaniasis in equids. The case reports described animals with cutaneous symptoms of leishmaniasis (papules, nodules, ulcers or crusts) that regressed spontaneously, located mainly on the head and limbs, from which three species of protozoa were identified in the lesions: Leishmania braziliensis, Leishmania infantum and Leishmania siamensis. In turn, the meta-analysis showed a combined prevalence of 25%, although with high heterogeneity among the studies, which was attributed to the use of different methods for diagnosing the disease. Leishmaniasis in equids is a benign disease but it should be included in the differential diagnosis of cutaneous diseases among these species. Seroepidemiological studies are important in investigating and monitoring suspected exposure of these hosts to the parasite, especially in endemic areas. However, there is also a need to standardize diagnostic methods.
Keywords:
Horse; diagnosis; Leishmania; meta-analysis; zoonosis
Resumo
As leishmanioses são um grupo de doenças de importância zoonótica causadas por mais de 20 espécies de protozoários do gênero Leishmania, sendo o cão doméstico considerado o principal reservatório da doença. No entanto, diversas pesquisas têm investigado o envolvimento de outros vertebrados como reservatórios do parasita. Portanto, o objetivo do presente estudo foi realizar uma revisão sistemática com meta-análise da ocorrência de leishmaniose em equídeos. Os relatos de caso descreviam animais com sintomas cutâneos de leishmaniose (pápulas, nódulos, úlceras, crostas) que regrediam espontaneamente, localizadas principalmente na cabeça e membros, sendo identificadas três espécies do protozoário nas lesões: Leishmania braziliensis, Leishmania infantum e Leishmania siamensis. Por sua vez, a meta-análise evidenciou uma prevalência combinada de 25%, porém com alta heterogeneidade entre os estudos, atribuída às diferenças nos métodos utilizados no diagnóstico da doença. A leishmaniose em equídeos é uma doença benigna, porém deve ser incluída no diagnóstico diferencial de doenças cutâneas nessas espécies. Os estudos soroepidemiológicos são importantes para investigar e monitorar a suspeita de exposição desses hospedeiros ao parasita, principalmente em áreas endêmicas, porém há necessidade de padronização dos métodos de diagnóstico.
Palavras-chave:
Equino; diagnóstico; Leishmania; meta-análise; zoonose
Introduction
According to the World Health Organization (WHO, 2017World Health Organization – WHO. Global leishmaniasis update, 2006–2015: a turning point in leishmaniasis surveillance. Weekly Epidemiological Record [online] 2017 [cited 2018 Nov 11]; 92(38): 557-572. Available from: http://apps.who.int/iris/bitstream/10665/258973/1/WER9238.pdf?ua=1
http://apps.who.int/iris/bitstream/10665...
), leishmaniases are a group of parasitic diseases caused by over 20 different species of Leishmania spp., protozoa that are transmitted mainly through the bite of sandflies. Four forms of the disease are known: visceral leishmaniasis (or kala-azar), post-kala-azar dermal leishmaniasis, cutaneous leishmaniasis and mucocutaneous leishmaniasis.
In humans, the most common clinical manifestation is the cutaneous form, which is considered endemic in 44% of the countries that notified cases of the disease in 2015 (WHO, 2017World Health Organization – WHO. Global leishmaniasis update, 2006–2015: a turning point in leishmaniasis surveillance. Weekly Epidemiological Record [online] 2017 [cited 2018 Nov 11]; 92(38): 557-572. Available from: http://apps.who.int/iris/bitstream/10665/258973/1/WER9238.pdf?ua=1
http://apps.who.int/iris/bitstream/10665...
); while the visceral form is the most severe, nearly always fatal if untreated, and is endemic in 38% of the countries that reported occurrence of the disease in that same year (WHO, 2017World Health Organization – WHO. Global leishmaniasis update, 2006–2015: a turning point in leishmaniasis surveillance. Weekly Epidemiological Record [online] 2017 [cited 2018 Nov 11]; 92(38): 557-572. Available from: http://apps.who.int/iris/bitstream/10665/258973/1/WER9238.pdf?ua=1
http://apps.who.int/iris/bitstream/10665...
). In addition to affecting humans, leishmaniasis also affects several domestic mammals. Infected dogs are the most important reservoirs for the parasite in urban areas, which makes them the main source of infection for people living in endemic areas (PACE, 2014Pace D. Leishmaniasis. J Infect 2014; 69(Suppl 1): S10-S18. http://dx.doi.org/10.1016/j.jinf.2014.07.016. PMid:25238669.
http://dx.doi.org/10.1016/j.jinf.2014.07...
).
However, several studies have sought to identify other vertebrates that can host and participate in the cycle of these protozoa (QUARESMA et al., 2011Quaresma PF, Rêgo FD, Botelho HA, Silva SR, Moura AJ Jr, Teixeira RG No, et al. Wild, synanthropic and domestic hosts of Leishmania in an endemic area of cutaneous leishmaniasis in Minas Gerais State, Brazil. Trans R Soc Trop Med Hyg 2011; 105(10): 579-585. http://dx.doi.org/10.1016/j.trstmh.2011.07.005. PMid:21890159.
http://dx.doi.org/10.1016/j.trstmh.2011....
; GAO et al., 2015Gao CH, Wang JY, Zhang S, Yang YT, Wang Y. Survey of wild and domestic mammals for infection with Leishmania infantum following an outbreak of desert zoonotic visceral leishmaniasis in jiashi, people’s Republic of China. PLoS One 2015; 10(7): e0132493. http://dx.doi.org/10.1371/journal.pone.0132493. PMid:26177101.
http://dx.doi.org/10.1371/journal.pone.0...
; KENUBIH et al., 2015Kenubih A, Dagnachew S, Almaw G, Abebe T, Takele Y, Hailu A, et al. Preliminary survey of domestic animal visceral leishmaniasis and risk factors in north-west Ethiopia. Trop Med Int Health 2015; 20(2): 205-210. http://dx.doi.org/10.1111/tmi.12418. PMid:25327874.
http://dx.doi.org/10.1111/tmi.12418...
; ROHOUSOVA et al., 2015Rohousova I, Talmi-Frank D, Kostalova T, Polanska N, Lestinova T, Kassahun A, et al. Exposure to Leishmania spp. and sand flies in domestic animals in northwestern Ethiopia. Parasit Vectors 2015; 8(1): 360. http://dx.doi.org/10.1186/s13071-015-0976-1. PMid:26152578.
http://dx.doi.org/10.1186/s13071-015-097...
). Among the investigations that have sought new possible reservoirs, there have been reports on equids showing clinical manifestations of cutaneous leishmaniasis (KOEHLER et al., 2002Koehler K, Stechele M, Hetzel U, Domingo M, Schönian G, Zahner H, et al. Cutaneous leishmaniosis in a horse in southern Germany caused by Leishmania infantum. Vet Parasitol 2002; 109(1-2): 9-17. http://dx.doi.org/10.1016/S0304-4017(02)00246-7. PMid:12383621.
http://dx.doi.org/10.1016/S0304-4017(02)...
; SOLANO-GALLEGO et al., 2003Solano-Gallego L, Fernández-Bellon H, Serra P, Gállego M, Ramis A, Fondevila D, et al. Cutaneous leishmaniosis in three horses in Spain. Equine Vet J 2003; 35(3): 320-323. http://dx.doi.org/10.2746/042516403776148336. PMid:12755438.
http://dx.doi.org/10.2746/04251640377614...
; MÜLLER et al., 2009Müller N, Welle M, Lobsiger L, Stoffel MH, Boghenbor KK, Hilbe M, et al. Occurrence of Leishmania sp. in cutaneous lesions of horses in Central Europe. Vet Parasitol 2009; 166(3-4): 346-351. http://dx.doi.org/10.1016/j.vetpar.2009.09.001. PMid:19800739.
http://dx.doi.org/10.1016/j.vetpar.2009....
; SOARES et al., 2013Soares IR, Silva SO, Moreira FM, Prado LG, Fantini P, Maranhão RPA, et al. First evidence of autochthonous cases of Leishmania (Leishmania) infantum in horse (Equus caballus) in the Americas and mixed infection of Leishmania infantum and Leishmania (Viannia) braziliensis. Vet Parasitol 2013; 197(3-4): 665-669. http://dx.doi.org/10.1016/j.vetpar.2013.06.014. PMid:23845306.
http://dx.doi.org/10.1016/j.vetpar.2013....
). Furthermore, prevalence studies conducted in Europe (FERNÁNDEZ-BELLON et al., 2006Fernández-Bellon H, Solano-Gallego L, Bardagí M, Alberola J, Ramis A, Ferrer L. Immune response to Leishmania infantum in healthy horses in Spain. Vet Parasitol 2006; 135(2): 181-185. http://dx.doi.org/10.1016/j.vetpar.2005.09.007. PMid:16213661.
http://dx.doi.org/10.1016/j.vetpar.2005....
; LOPES et al., 2013Lopes AP, Sousa S, Dubey JP, Ribeiro AJ, Silvestre R, Cotovio M, et al. Prevalence of antibodies to Leishmania infantum and Toxoplasma gondii in horses from the north of Portugal. Parasit Vectors 2013; 6(1): 178. http://dx.doi.org/10.1186/1756-3305-6-178. PMid:23773870.
http://dx.doi.org/10.1186/1756-3305-6-17...
; SGORBINI et al., 2014Sgorbini M, Bonelli F, Pizzolli I, Tognetti R, Corazza M. Seroprevalence of Leishmania sp. infection in healthy horses housed in endemic areas in Tuscany. J Equine Vet Sci 2014; 34(4): 572-574. http://dx.doi.org/10.1016/j.jevs.2013.09.009.
http://dx.doi.org/10.1016/j.jevs.2013.09...
), Asia (GAO et al., 2015Gao CH, Wang JY, Zhang S, Yang YT, Wang Y. Survey of wild and domestic mammals for infection with Leishmania infantum following an outbreak of desert zoonotic visceral leishmaniasis in jiashi, people’s Republic of China. PLoS One 2015; 10(7): e0132493. http://dx.doi.org/10.1371/journal.pone.0132493. PMid:26177101.
http://dx.doi.org/10.1371/journal.pone.0...
; AHARONSON-RAZ et al., 2015Aharonson-Raz K, Baneth G, Lopes AP, Brancal H, Schallig H, Cardoso L, et al. Low seroprevalence of Leishmania infantum and Toxoplasma gondii in the horse population in Israel. Vector Borne Zoonotic Dis 2015; 15(12): 726-731. http://dx.doi.org/10.1089/vbz.2015.1826. PMid:26580094.
http://dx.doi.org/10.1089/vbz.2015.1826...
), Africa (MUKHTAR et al., 2000Mukhtar MM, Sharief AH, el Saffi SH, Harith AE, Higazzi TB, Adam AM, et al. Detection of antibodies to Leishmania donovani in animals in a kala-azar endemic region in eastern Sudan: a preliminary report. Trans R Soc Trop Med Hyg 2000; 94(1): 33-36. http://dx.doi.org/10.1016/S0035-9203(00)90429-2. PMid:10748894.
http://dx.doi.org/10.1016/S0035-9203(00)...
; KENUBIH et al., 2015Kenubih A, Dagnachew S, Almaw G, Abebe T, Takele Y, Hailu A, et al. Preliminary survey of domestic animal visceral leishmaniasis and risk factors in north-west Ethiopia. Trop Med Int Health 2015; 20(2): 205-210. http://dx.doi.org/10.1111/tmi.12418. PMid:25327874.
http://dx.doi.org/10.1111/tmi.12418...
; ROHOUSOVA et al., 2015Rohousova I, Talmi-Frank D, Kostalova T, Polanska N, Lestinova T, Kassahun A, et al. Exposure to Leishmania spp. and sand flies in domestic animals in northwestern Ethiopia. Parasit Vectors 2015; 8(1): 360. http://dx.doi.org/10.1186/s13071-015-0976-1. PMid:26152578.
http://dx.doi.org/10.1186/s13071-015-097...
) and South America (AGUILAR et al., 1989Aguilar CM, Rangel EF, Garcia L, Fernandez E, Momen H, Grimaldi G Fo, et al. Zoonotic cutaneous leishmaniasis due to Leishmania (Viannia) braziliensis associated with domestic animals in Venezuela and Brazil. Mem Inst Oswaldo Cruz 1989; 84(1): 19-28. http://dx.doi.org/10.1590/S0074-02761989000100005. PMid:2319948.
http://dx.doi.org/10.1590/S0074-02761989...
; FEITOSA et al., 2012Feitosa FLF, Leal J, Mendes LCN, Peiró JR, Perri SHV, Lima VMF, et al. Estudo soroepidemiológico de leishmaniose em equinos na região de Araçatuba-SP, Brasil, área endêmica para leishmaniose visceral. Braz J Vet Res Anim Sci 2012; 49(6): 500-502. http://dx.doi.org/10.11606/issn.1678-4456.v49i6p500-502.
http://dx.doi.org/10.11606/issn.1678-445...
; TRUPPEL et al., 2014Truppel JH, Otomura F, Teodoro U, Massafera R, Costa-Ribeiro MCV, Catarino CM, et al. Can equids be a reservoir of Leishmania braziliensis in endemic areas? PLoS One 2014; 9(4): e93731. http://dx.doi.org/10.1371/journal.pone.0093731. PMid:24721908.
http://dx.doi.org/10.1371/journal.pone.0...
; OLIVEIRA et al., 2017Oliveira PM, Garcia F, Evers F, Barbosa VM, Obando DCM, Nasciutti NR, et al. Seroepidemiology of Leishmania spp. in equids from Uberlândia, Minas Gerais, Brazil. Cienc Rural 2017; 47(5): e20160697. http://dx.doi.org/10.1590/0103-8478cr20160697.
http://dx.doi.org/10.1590/0103-8478cr201...
; BENASSI et al., 2018Benassi JC, Benvenga GU, Ferreira HL, Soares RM, Silva DT, Pereira VF, et al. Molecular and serological detection of Leishmania spp. in horses from an endemic area for canine visceral leishmaniasis in southeastern Brazil. Pesq Vet Bras 2018; 38(6): 1058-1063. http://dx.doi.org/10.1590/1678-5150-pvb-5214.
http://dx.doi.org/10.1590/1678-5150-pvb-...
) have demonstrated that horses (Equus caballus), donkeys (Equus asinus), mules (Equus asinus caballus) and ponies (E. caballus) are parasitized by different species of Leishmania, such as Leishmania braziliensis, Leishmania infantum and Leishmania siamensis, including cases of mixed infections.
The importance of research on leishmaniasis in equids lies in the fact that these species of domestic animal, just like dogs and cats, are in close contact with humans, which may be through use as a means of transportation or for work or leisure activities. Moreover, the low socioeconomic level of the population living in endemic areas for the disease could generate risks of infection across the zoonotic cycle, such that equids would play the role of either potential reservoirs or sources of food for sandflies in peridomestic areas.
Therefore, the objective of the present study was to carry out a qualitative and quantitative synthesis (with meta-analysis) based on a systematic review of the literature. Greater clarification regarding the main clinical aspects of leishmaniasis in equids and methods for diagnosing it was sought, with a view to enabling support for future studies on this subject.
Materials and Methods
Study design
The present study consisted of a systematic review of the literature, with synthesis and analysis of clinical findings and meta-analysis on the quantitative data available in articles from indexed journals, both from Brazil and from other countries. The study was conducted based on the methodological recommendations of PRISMA: the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (MOHER et al., 2009Moher D, Liberati A, Tetzlaff J, Altman DG, Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009; 6(7): e1000097. http://dx.doi.org/10.1371/journal.pmed.1000097. PMid:19621072.
http://dx.doi.org/10.1371/journal.pmed.1...
).
Article eligibility
Articles published in indexed journals were considered eligible if they consisted of case reports and cross-sectional studies describing the clinical and epidemiological characteristics (prevalence, species, sex and age) and diagnostic methods that are used to identify equids naturally infected with Leishmania spp. There were no restrictions regarding the year in which the study was developed or published, the language or the country where the study was conducted.
The types of publications included were complete articles, short communications and case reports that addressed issues within the following criteria: (I) information on the clinical presentation of leishmaniasis in equids; (II) prevalence of the disease in equid populations; or (III) diagnosis of leishmaniasis in equids. Reviews of the literature, research notes, editorials, experimental assays and other types of publications not within the inclusion criteria were excluded.
Information sources and search strategies
Considering the pre-established inclusion criteria, the process of identifying articles was developed using the PubMed, SciELO, ScienceDirect, Scopus and Web of Science databases. The following combination of search terms in English was used: {leishmania OR leishmaniosis} AND {equids OR equine OR horses OR donkeys OR mules}. The citations of studies thus identified, containing title and abstract, were saved in BibTex format and were exported to a bibliographic manager for subsequent selection. The searches were conducted between October 19 and 26, 2018.
Selection of studies and data extraction
A bibliographic manager tool was used to exclude duplicate records. After this stage, two researchers selected studies independently based on an analysis of titles and abstracts, followed by a full reading of the text. Through this full evaluation of the texts, other studies were excluded because they did not meet the eligibility criteria. Occurrences of divergences between the two researchers were resolved by reaching a consensus.
To make it easier to extract and analyze the data, the articles selected were divided into two groups: the first included all case reports of leishmaniasis in equids, for qualitative synthesis; while the second included cross-sectional studies, for quantitative synthesis and meta-analysis.
Data extraction was conducted independently by two researchers and the information was added to a previously developed electronic spreadsheet. The qualitative data extracted from the first group of articles comprised the authors, year of publication, country, clinical characteristics (type and location of lesions), etiological agent and identification of animals (species, sex and age). In turn, the quantitative data extracted from the second group of articles comprised the references (authors and year of publication), country where the study was conducted, sample size, number of positive animals, prevalence (%) and diagnostic methods.
Data analysis
The qualitative data were analyzed using descriptive statistics, by means of absolute and percentage distributions, to characterize the clinical aspects of leishmaniasis in equids and methods for diagnosing it. The primary outcome for the quantitative data was the crude prevalence of leishmaniasis in equids, with a 95% confidence interval (95% CI).
Heterogeneity was assessed using Cochran’s Q test and was quantified through the I2 test of Higgins and Thompson. The combined estimates and 95% confidence interval were calculated based on the random-effects model through the inverse of variance, using the DerSimonian-Laird method. Funnel plots were also analyzed visually and Egger’s test was applied, as alternatives for identifying possible biases. All analyses were conducted using the R statistical software (version 3.5.1), through the RStudio software interface (version 1.1.463).
Results
The initial search of databases and study selection are presented in Figure 1. Out of the total number of studies searched (n = 531), 29 met the eligibility criteria and were divided as follows: 11 studies described the clinical characteristics of leishmaniasis in equids, and thus were included in the qualitative synthesis; and another 18 were considered to be cross-sectional studies (prevalence studies), with sufficient data for quantitative synthesis and meta-analysis.
Flow chart of the search, selection and inclusion process for studies in the systematic review.
Qualitative synthesis of the clinical aspects of leishmaniasis in equids
The 11 studies included in the qualitative synthesis were conducted in seven different countries (Germany, Brazil, Spain, United States, Puerto Rico, Portugal and Switzerland) and reported occurrences of cutaneous leishmaniasis in a total of 22 equids (E. caballus), of which 15 were male (68.18%) and 7 were female (31.82%), with ages between 0 and 2 (n = 2), 2 and 5 (n = 8), 5 and 10 (n = 9) and over 10 years (n = 3).
The major clinical manifestations described in the studies were limited to the skin, and included ulcers (n = 10), nodules (n = 7), crusts (n = 2), papules (n = 1), areas of alopecia (n = 1), presence of exudate (n = 1) and pruritus (n = 1). Most lesions were described as multiple, although single lesions were also observed in some studies, distributed over different parts of the body. The protozoa identified in the studies were Leishmania braziliensis, Leishmania infantum, Leishmania siamensis and also mixed infection by L. braziliensis and L. infantum. Further details of the clinical factors (characteristics and locations of lesions), methods used for diagnosis and etiological agents are described in Table 1.
Characteristics of the studies included in the qualitative synthesis regarding leishmaniasis in equids.
Quantitative synthesis and meta-analysis on leishmaniasis prevalence in equids
The studies included in this stage were conducted in Brazil (n = 10), China (n = 1), Spain (n = 1), Ethiopia (n = 2), Greece (n = 1), Israel (n = 1), Italy (n = 1), Portugal (n = 1), Sudan (n = 1) and Venezuela (n = 2). In one of these studies (AGUILAR et al., 1989Aguilar CM, Rangel EF, Garcia L, Fernandez E, Momen H, Grimaldi G Fo, et al. Zoonotic cutaneous leishmaniasis due to Leishmania (Viannia) braziliensis associated with domestic animals in Venezuela and Brazil. Mem Inst Oswaldo Cruz 1989; 84(1): 19-28. http://dx.doi.org/10.1590/S0074-02761989000100005. PMid:2319948.
http://dx.doi.org/10.1590/S0074-02761989...
), data collected in both Brazil and Venezuela were published. In turn, in two other studies (TRUPPEL et al., 2014Truppel JH, Otomura F, Teodoro U, Massafera R, Costa-Ribeiro MCV, Catarino CM, et al. Can equids be a reservoir of Leishmania braziliensis in endemic areas? PLoS One 2014; 9(4): e93731. http://dx.doi.org/10.1371/journal.pone.0093731. PMid:24721908.
http://dx.doi.org/10.1371/journal.pone.0...
; BENASSI et al., 2018Benassi JC, Benvenga GU, Ferreira HL, Soares RM, Silva DT, Pereira VF, et al. Molecular and serological detection of Leishmania spp. in horses from an endemic area for canine visceral leishmaniasis in southeastern Brazil. Pesq Vet Bras 2018; 38(6): 1058-1063. http://dx.doi.org/10.1590/1678-5150-pvb-5214.
http://dx.doi.org/10.1590/1678-5150-pvb-...
), two different diagnostic techniques were used. We therefore considered the use of these different techniques to constitute different studies and, for this reason, the initial total of 18 studies identified was then counted as 21 in the quantitative analysis phase.
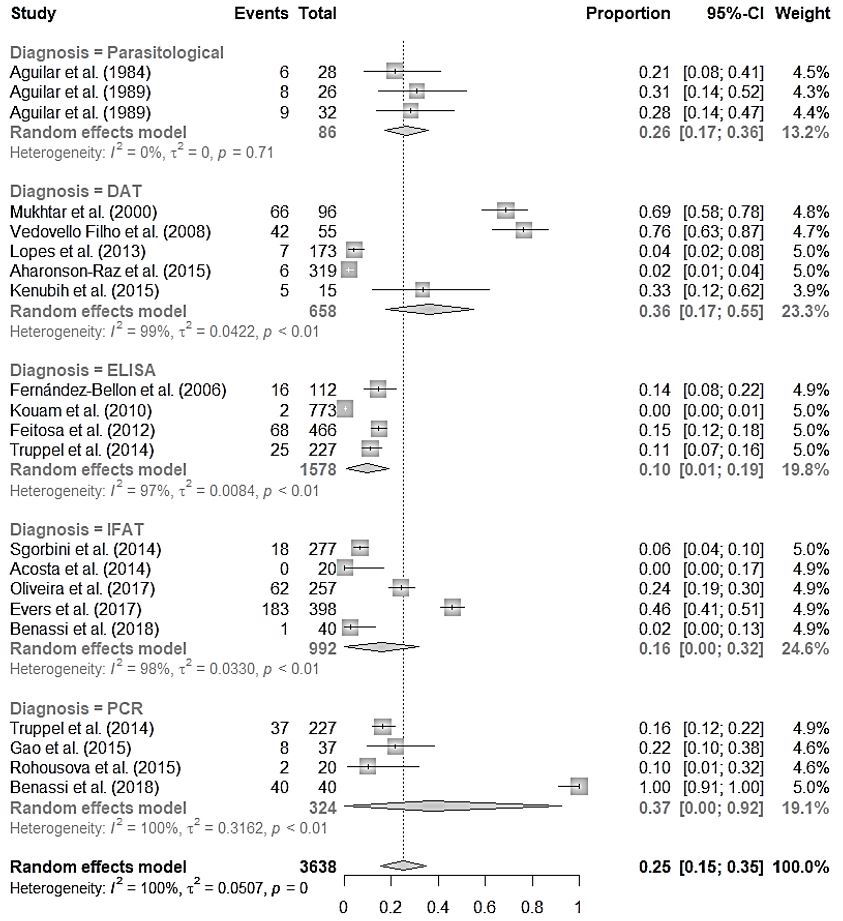
Table 2 summarizes the main characteristics of the studies included in the meta-analysis. In evaluating the prevalence results through Cochran’s Q test (in which p = 0) and the I2 statistic of Higgins and Thompson (in which I2 = 99.5%), high heterogeneity was observed among the studies. Therefore, a random-effects model was used, according to subgroup, to conduct the meta-analysis. This model yielded a combined prevalence of leishmaniasis in equids of 25% (CI: 15-35%), among the studies included (Figure 2). To assess the possible causes of heterogeneity, the studies were divided into subgroups according to the technique that had been used for the diagnosis: parasitological; enzyme-linked immunosorbent assay (ELISA); direct agglutination test (DAT); indirect fluorescence antibody test (IFAT); and polymerase chain reaction (PCR). These results are shown in Figure 2.
Quantitative synthesis regarding the main characteristics of the studies included in the meta-analysis.
Combination of 21 prevalence studies on leishmaniasis in equids, according to the diagnosis method used.
The visual analysis on the funnel plot (Figure 3) showed that there was asymmetrical distribution among the 21 studies, thus demonstrating the possibility of publication bias, which was confirmed through applying Egger’s test (p = 0.01).
Funnel plot presenting the asymmetrical distribution of studies on the prevalence of leishmaniasis in equids.
Discussion
The qualitative analysis on the case reports showed that all the equids were affected by the cutaneous form of leishmaniasis (Table 1), presenting lesions that began with papules and nodules and progressed to ulcers, with presence of crusts, alopecia, exudate and pruritus as a result of the evolving inflammatory process that was underway. There is a consensus among researchers that the common infection site of L. braziliensis is the skin, while L. infantum is responsible for visceral infection (KOEHLER et al., 2002Koehler K, Stechele M, Hetzel U, Domingo M, Schönian G, Zahner H, et al. Cutaneous leishmaniosis in a horse in southern Germany caused by Leishmania infantum. Vet Parasitol 2002; 109(1-2): 9-17. http://dx.doi.org/10.1016/S0304-4017(02)00246-7. PMid:12383621.
http://dx.doi.org/10.1016/S0304-4017(02)...
). However, the present review shows that this has not been observed among equids, given that all the reports cited in this review showed predominance of the cutaneous form of the disease, regardless of which species of Leishmania was identified.
When an etiological diagnosis of the disease was possible, the most frequent species found was L. infantum (n = 8), followed by L. siamensis (n = 5), L. braziliensis (n = 2) and mixed infection of L. infantum and L. braziliensis (n = 1). In the remaining cases (n = 6), the agent was not identified. Occurrences of species with higher pathogenicity towards humans (L. infantum and L. siamensis) infecting equids is a reason for greater concern, given that vectors use this mammal species as a source of food and could consequently ingest and become contaminated by the protozoa, although this type of transmission has not yet been demonstrated in equids in a general manner.
The lesions that were described in the studies surveyed here were located mainly on the head and limbs, although they were also observed on the neck, genital organs, abdomen and thorax, which are regions where there are few or even no hairs, which facilitates access by the vector mosquito to begin feeding and, consequently, to inoculate the parasite into the skin. Moreover, research on humans has shown that attractive volatile substances are exhaled from specific parts of the body, such as the ears, which attract greater numbers of insects to that region (REBOLLAR-TELLEZ et al., 1999Rebollar-Tellez EA, Hamilton JGC, Ward RD. Response of female Lutzomyia longipalpis to host odour kairomones from human skin. Physiol Entomol 1999; 24(3): 220-226. http://dx.doi.org/10.1046/j.1365-3032.1999.00133.x.
http://dx.doi.org/10.1046/j.1365-3032.19...
). Although no such reports relating to equids have been conducted, presence of volatile attractants forms a plausible argument for explaining the presence of lesions on certain body parts rather than on others, thus suggesting that this occurs not just because of absence of hair in these places.
Skin diseases are common among equids, and some present characteristics similar to those described for leishmaniasis, such as equine sarcoid, squamous cell carcinoma, pythiosis, habronemiasis (KOEHLER et al., 2002Koehler K, Stechele M, Hetzel U, Domingo M, Schönian G, Zahner H, et al. Cutaneous leishmaniosis in a horse in southern Germany caused by Leishmania infantum. Vet Parasitol 2002; 109(1-2): 9-17. http://dx.doi.org/10.1016/S0304-4017(02)00246-7. PMid:12383621.
http://dx.doi.org/10.1016/S0304-4017(02)...
; SOLANO-GALLEGO et al., 2003Solano-Gallego L, Fernández-Bellon H, Serra P, Gállego M, Ramis A, Fondevila D, et al. Cutaneous leishmaniosis in three horses in Spain. Equine Vet J 2003; 35(3): 320-323. http://dx.doi.org/10.2746/042516403776148336. PMid:12755438.
http://dx.doi.org/10.2746/04251640377614...
) or any other cutaneous disorder that causes either papular or nodular lesions and/or ulcers in equids (SOARES et al., 2013Soares IR, Silva SO, Moreira FM, Prado LG, Fantini P, Maranhão RPA, et al. First evidence of autochthonous cases of Leishmania (Leishmania) infantum in horse (Equus caballus) in the Americas and mixed infection of Leishmania infantum and Leishmania (Viannia) braziliensis. Vet Parasitol 2013; 197(3-4): 665-669. http://dx.doi.org/10.1016/j.vetpar.2013.06.014. PMid:23845306.
http://dx.doi.org/10.1016/j.vetpar.2013....
). Thus, leishmaniasis should be considered in making differential diagnoses of dermatopathies in equids, especially when the animals inhabit regions that are endemic for leishmaniasis and lesions occur in areas such as the genital organs, head, neck, ears and inguinal and axillary regions, and when there is no satisfactory response from administration of antimicrobial or antifungal therapies (RAMOS-VARA et al., 1996Ramos-Vara JA, Ortiz-Santiago B, Segalès J, Dunstan RW. Cutaneous leishmaniasis in two horses. Vet Pathol 1996; 33(6): 731-734. http://dx.doi.org/10.1177/030098589603300619. PMid:8952039.
http://dx.doi.org/10.1177/03009858960330...
).
Most lesions that were described in previous studies either regressed spontaneously without the need for treatment or did not recur after surgical removal (RAMOS-VARA et al., 1996Ramos-Vara JA, Ortiz-Santiago B, Segalès J, Dunstan RW. Cutaneous leishmaniasis in two horses. Vet Pathol 1996; 33(6): 731-734. http://dx.doi.org/10.1177/030098589603300619. PMid:8952039.
http://dx.doi.org/10.1177/03009858960330...
; KOEHLER et al., 2002Koehler K, Stechele M, Hetzel U, Domingo M, Schönian G, Zahner H, et al. Cutaneous leishmaniosis in a horse in southern Germany caused by Leishmania infantum. Vet Parasitol 2002; 109(1-2): 9-17. http://dx.doi.org/10.1016/S0304-4017(02)00246-7. PMid:12383621.
http://dx.doi.org/10.1016/S0304-4017(02)...
; SOLANO-GALLEGO et al., 2003Solano-Gallego L, Fernández-Bellon H, Serra P, Gállego M, Ramis A, Fondevila D, et al. Cutaneous leishmaniosis in three horses in Spain. Equine Vet J 2003; 35(3): 320-323. http://dx.doi.org/10.2746/042516403776148336. PMid:12755438.
http://dx.doi.org/10.2746/04251640377614...
; ROLÃO et al., 2005Rolão N, Martins MJ, João A, Campino L. Equine infection with Leishmania in Portugal. Parasite 2005; 12(2): 183-186. http://dx.doi.org/10.1051/parasite/2005122183. PMid:15991833.
http://dx.doi.org/10.1051/parasite/20051...
; MÜLLER et al., 2009Müller N, Welle M, Lobsiger L, Stoffel MH, Boghenbor KK, Hilbe M, et al. Occurrence of Leishmania sp. in cutaneous lesions of horses in Central Europe. Vet Parasitol 2009; 166(3-4): 346-351. http://dx.doi.org/10.1016/j.vetpar.2009.09.001. PMid:19800739.
http://dx.doi.org/10.1016/j.vetpar.2009....
; GAMA et al., 2014Gama A, Elias J, Ribeiro AJ, Alegria N, Schallig HDFH, Silva F, et al. Cutaneous leishmaniosis in a horse from northern Portugal. Vet Parasitol 2014; 200(1-2): 189-192. http://dx.doi.org/10.1016/j.vetpar.2013.12.005. PMid:24388338.
http://dx.doi.org/10.1016/j.vetpar.2013....
). This emphasizes the idea that the immune response of equids against the parasite is effective (FERNÁNDEZ-BELLON et al., 2006Fernández-Bellon H, Solano-Gallego L, Bardagí M, Alberola J, Ramis A, Ferrer L. Immune response to Leishmania infantum in healthy horses in Spain. Vet Parasitol 2006; 135(2): 181-185. http://dx.doi.org/10.1016/j.vetpar.2005.09.007. PMid:16213661.
http://dx.doi.org/10.1016/j.vetpar.2005....
).
Use of medication to treat the disease was reported in three studies. In the first, Barbosa-Santos et al. (1994)Barbosa-Santos EG, Marzochi MC, Urtado W, Queirós F, Chicarino J, Pacheco RS. Leishmaniasis disseminated by Leishmania braziliensis in a mare (Equus cabalus) immunotherapy and chemotherapy assays. Mem Inst Oswaldo Cruz 1994; 89(2): 217-220. http://dx.doi.org/10.1590/S0074-02761994000200018. PMid:7885248.
http://dx.doi.org/10.1590/S0074-02761994...
initially used immunotherapy and observed an increase in antibody levels and worsening of lesions, which then led them to use conventional chemotherapy (pentavalent antimony), through which regression of lesions was achieved after a second application. In the second report, Ramos-Vara et al. (1996)Ramos-Vara JA, Ortiz-Santiago B, Segalès J, Dunstan RW. Cutaneous leishmaniasis in two horses. Vet Pathol 1996; 33(6): 731-734. http://dx.doi.org/10.1177/030098589603300619. PMid:8952039.
http://dx.doi.org/10.1177/03009858960330...
used sodium stibogluconate and reached complete cure for the lesions. Lastly, Solano-Gallego et al. (2003)Solano-Gallego L, Fernández-Bellon H, Serra P, Gállego M, Ramis A, Fondevila D, et al. Cutaneous leishmaniosis in three horses in Spain. Equine Vet J 2003; 35(3): 320-323. http://dx.doi.org/10.2746/042516403776148336. PMid:12755438.
http://dx.doi.org/10.2746/04251640377614...
used anti-inflammatory therapy with dexamethasone, but without any satisfactory response.
Regarding the quantitative synthesis, the meta-analysis indicated that the prevalence of leishmaniasis in equine populations was 25% (CI:15-35%), with distribution over four different continents (Africa, South America, Asia and Europe). However, the high heterogeneity of the studies analyzed gives rise to some reflection regarding the degree of reliability of this combined prevalence among the studies.
In seeking to elucidate this high heterogeneity, we were particularly interested in the number of different diagnostic techniques that have been used to investigate leishmaniasis in equids. Thus, one meta-analysis per subgroup was conducted, with separation according to the method used for the diagnosis: parasitological examinations, IFAT, ELISA, PCR and DAT.
The forest plot (Figure 2) brings the 21 studies included in the meta-analysis, where the points inside the boxes in the center of the graph represent the prevalence of each study individually and the horizontal line the confidence interval, and the diamonds demonstrate the combined prevalence among studies. Prevalence results and confidence intervals are shown in the fourth and fifth columns, respectively. The last column represents the weight with which each study participated in the aggregate result.
The heterogeneity was high both in the aggregate outcome of the 21 studies (I2 = 100%) and in the DAT (I2 = 99%), ELISA (I2 = 97%), IFAT (I2 = 98%) and PCR (I2 = 100%) subgroups. As shown in Figure 2, heterogeneity is clearly present between the studies that used different diagnostic techniques, and it is also present among studies that used the same technique. One exception were the three studies that used parasitological techniques, which did not show heterogeneity between each other (I2=0%). Parasitological examinations are considered to be the gold standard for diagnosis, and thus their results are more reliable.
Serological tests were the diagnostic techniques that presented greatest methodological differences when used to diagnose leishmaniasis in equids. Regarding DAT, for example, five studies used different antigen concentrations, serum dilutions and cutoff points (LOPES et al., 2013Lopes AP, Sousa S, Dubey JP, Ribeiro AJ, Silvestre R, Cotovio M, et al. Prevalence of antibodies to Leishmania infantum and Toxoplasma gondii in horses from the north of Portugal. Parasit Vectors 2013; 6(1): 178. http://dx.doi.org/10.1186/1756-3305-6-178. PMid:23773870.
http://dx.doi.org/10.1186/1756-3305-6-17...
; AHARONSON-RAZ et al., 2015Aharonson-Raz K, Baneth G, Lopes AP, Brancal H, Schallig H, Cardoso L, et al. Low seroprevalence of Leishmania infantum and Toxoplasma gondii in the horse population in Israel. Vector Borne Zoonotic Dis 2015; 15(12): 726-731. http://dx.doi.org/10.1089/vbz.2015.1826. PMid:26580094.
http://dx.doi.org/10.1089/vbz.2015.1826...
; KENUBIH et al., 2015Kenubih A, Dagnachew S, Almaw G, Abebe T, Takele Y, Hailu A, et al. Preliminary survey of domestic animal visceral leishmaniasis and risk factors in north-west Ethiopia. Trop Med Int Health 2015; 20(2): 205-210. http://dx.doi.org/10.1111/tmi.12418. PMid:25327874.
http://dx.doi.org/10.1111/tmi.12418...
), along with positive control serums from either humans or dogs (MUKHTAR et al., 2000Mukhtar MM, Sharief AH, el Saffi SH, Harith AE, Higazzi TB, Adam AM, et al. Detection of antibodies to Leishmania donovani in animals in a kala-azar endemic region in eastern Sudan: a preliminary report. Trans R Soc Trop Med Hyg 2000; 94(1): 33-36. http://dx.doi.org/10.1016/S0035-9203(00)90429-2. PMid:10748894.
http://dx.doi.org/10.1016/S0035-9203(00)...
; VEDOVELLO et al., 2008Vedovello D Fo, Jorge FA, Lonardoni MVC, Teodoro U, Silveira TGV. American Cutaneous Leishmaniasis in horses from endemic areas in the North-Central Mesoregion of Paraná State, Brazil. Zoonoses Public Health 2008; 55(3): 149-155. http://dx.doi.org/10.1111/j.1863-2378.2008.01106.x. PMid:18331518.
http://dx.doi.org/10.1111/j.1863-2378.20...
). In ELISA, there were also methodological differences, especially regarding the types of antigens and conjugates used in serum dilutions (FERNÁNDEZ-BELLON et al., 2006Fernández-Bellon H, Solano-Gallego L, Bardagí M, Alberola J, Ramis A, Ferrer L. Immune response to Leishmania infantum in healthy horses in Spain. Vet Parasitol 2006; 135(2): 181-185. http://dx.doi.org/10.1016/j.vetpar.2005.09.007. PMid:16213661.
http://dx.doi.org/10.1016/j.vetpar.2005....
; KOUAM et al., 2010Kouam MK, Diakou A, Kanzoura V, Papadopoulos E, Gajadhar AA, Theodoropoulos G. A seroepidemiological study of exposure to Toxoplasma, Leishmania, Echinococcus and Trichinella in equids in Greece and analysis of risk factors. Vet Parasitol 2010; 170(1-2): 170-175. http://dx.doi.org/10.1016/j.vetpar.2010.02.004. PMid:20197215.
http://dx.doi.org/10.1016/j.vetpar.2010....
; FEITOSA et al., 2012Feitosa FLF, Leal J, Mendes LCN, Peiró JR, Perri SHV, Lima VMF, et al. Estudo soroepidemiológico de leishmaniose em equinos na região de Araçatuba-SP, Brasil, área endêmica para leishmaniose visceral. Braz J Vet Res Anim Sci 2012; 49(6): 500-502. http://dx.doi.org/10.11606/issn.1678-4456.v49i6p500-502.
http://dx.doi.org/10.11606/issn.1678-445...
; TRUPPEL et al., 2014Truppel JH, Otomura F, Teodoro U, Massafera R, Costa-Ribeiro MCV, Catarino CM, et al. Can equids be a reservoir of Leishmania braziliensis in endemic areas? PLoS One 2014; 9(4): e93731. http://dx.doi.org/10.1371/journal.pone.0093731. PMid:24721908.
http://dx.doi.org/10.1371/journal.pone.0...
). Other serological studies used IFAT and also presented differences regarding the cutoff points and conjugates used (SGORBINI et al., 2014Sgorbini M, Bonelli F, Pizzolli I, Tognetti R, Corazza M. Seroprevalence of Leishmania sp. infection in healthy horses housed in endemic areas in Tuscany. J Equine Vet Sci 2014; 34(4): 572-574. http://dx.doi.org/10.1016/j.jevs.2013.09.009.
http://dx.doi.org/10.1016/j.jevs.2013.09...
; ACOSTA et al., 2014Acosta ICL, Costa AP, Gennari SM, Marcili A. Survey of Trypanosoma and Leishmania in wild and domestic animals in an atlantic rainforest fragment and surroundings in the state of Espírito Santo, Brazil. J Med Entomol 2014; 51(3): 686-693. http://dx.doi.org/10.1603/ME13177. PMid:24897863.
http://dx.doi.org/10.1603/ME13177...
; OLIVEIRA et al., 2017Oliveira PM, Garcia F, Evers F, Barbosa VM, Obando DCM, Nasciutti NR, et al. Seroepidemiology of Leishmania spp. in equids from Uberlândia, Minas Gerais, Brazil. Cienc Rural 2017; 47(5): e20160697. http://dx.doi.org/10.1590/0103-8478cr20160697.
http://dx.doi.org/10.1590/0103-8478cr201...
; EVERS et al., 2017Evers F, Ferreira FP, Navarro IT, Mitsuka-Breganó R, Pagliari S, Monica TC, et al. Presence of anti-Leishmania spp. antibodies in slaughter horses in Brazil. Semina: Ciênc Agrár 2017; 38(6): 3921-3926. http://dx.doi.org/10.5433/1679-0359.2017v38n6p3921.
http://dx.doi.org/10.5433/1679-0359.2017...
; BENASSI et al., 2018Benassi JC, Benvenga GU, Ferreira HL, Soares RM, Silva DT, Pereira VF, et al. Molecular and serological detection of Leishmania spp. in horses from an endemic area for canine visceral leishmaniasis in southeastern Brazil. Pesq Vet Bras 2018; 38(6): 1058-1063. http://dx.doi.org/10.1590/1678-5150-pvb-5214.
http://dx.doi.org/10.1590/1678-5150-pvb-...
).
Regarding molecular tests through PCR, the meta-analysis results also demonstrated high heterogeneity (I2 = 100%) among the four studies that used this technique (TRUPPEL et al., 2014Truppel JH, Otomura F, Teodoro U, Massafera R, Costa-Ribeiro MCV, Catarino CM, et al. Can equids be a reservoir of Leishmania braziliensis in endemic areas? PLoS One 2014; 9(4): e93731. http://dx.doi.org/10.1371/journal.pone.0093731. PMid:24721908.
http://dx.doi.org/10.1371/journal.pone.0...
; GAO et al., 2015Gao CH, Wang JY, Zhang S, Yang YT, Wang Y. Survey of wild and domestic mammals for infection with Leishmania infantum following an outbreak of desert zoonotic visceral leishmaniasis in jiashi, people’s Republic of China. PLoS One 2015; 10(7): e0132493. http://dx.doi.org/10.1371/journal.pone.0132493. PMid:26177101.
http://dx.doi.org/10.1371/journal.pone.0...
; ROHOUSOVA et al., 2015Rohousova I, Talmi-Frank D, Kostalova T, Polanska N, Lestinova T, Kassahun A, et al. Exposure to Leishmania spp. and sand flies in domestic animals in northwestern Ethiopia. Parasit Vectors 2015; 8(1): 360. http://dx.doi.org/10.1186/s13071-015-0976-1. PMid:26152578.
http://dx.doi.org/10.1186/s13071-015-097...
; BENASSI et al., 2018Benassi JC, Benvenga GU, Ferreira HL, Soares RM, Silva DT, Pereira VF, et al. Molecular and serological detection of Leishmania spp. in horses from an endemic area for canine visceral leishmaniasis in southeastern Brazil. Pesq Vet Bras 2018; 38(6): 1058-1063. http://dx.doi.org/10.1590/1678-5150-pvb-5214.
http://dx.doi.org/10.1590/1678-5150-pvb-...
). Although the authors of these studies used different primers to identify parasite DNA, we do not believe that this was the source of the heterogeneity that was observed in the meta-analysis of this subgroup. In this case, we believe it is coherent to attribute this heterogeneity to the variation in real prevalence of the disease in the studies indicated, given the high sensitivity and specificity of the PCR technique as a diagnostic method and the combined prevalence and confidence interval observed among the studies in which it was applied (37%; 0 – 92%).
Publication bias is often responsible for the heterogeneity between studies that is found through meta-analysis. In Figure 3 each point on the plot represents a study, with the prevalence arranged on the X axis and the standard error on the Y axis. In the absence of bias, the points are expected to have a symmetrical distribution under the dashed area of the triangle (inverted funnel), with the most accurate studies occupying the vertex (lowest standard error) and the least accurate distributed at the base of the triangle. It was possible to verify by observing the funnel plot (Figure 3) a marked asymmetry of the points (studies), indicating possible publication bias in the present meta-analysis, a fact also confirmed by the Egger’s test (p = 0.01). In relation to cross-sectional studies, it is especially common to find that researchers are not interested in publishing their studies when they do not find significant prevalence in their investigations. Moreover, the editors of journals even appear reluctant to publish these negative findings. This lack of negative findings generates bias that can interfere in the results from a meta-analysis (Pereira & Galvão, 2014Pereira MG, Galvão TF. Heterogeneidade e viés de publicação em revisões sistemáticas. Epidemiol Serv Saude 2014; 23(4): 775-778. http://dx.doi.org/10.5123/S1679-49742014000400021.
http://dx.doi.org/10.5123/S1679-49742014...
). However, we believe that the difference in the methodologies that were used for diagnosing the disease in equids was the factor that most contributed towards the high heterogeneity observed.
Conclusion
The clinical disease caused by different species of Leishmania spp. is benign to equids, thus not requiring treatment in most cases, with the exception of basic care to avoid secondary contaminations, or cases in which the lesions reach larger proportions. However, leishmaniasis is clinically important, given that it can be mistaken for other common skin diseases in equids. Thus, leishmaniasis should be considered in the differential diagnosis for these diseases. Moreover, more detailed investigations are needed regarding the pathology of the disease in these hosts, particularly if development of visceral lesions occurs, and regarding the role of the immune response in determining whether clinical signs will appear.
Hence, seroepidemiological studies are important for investigating and monitoring suspected exposure of these hosts to the parasite, especially in endemic areas, given that natural equid-vector-human transmission has not yet been either demonstrated or refuted and could, therefore, represent a risk to vulnerable populations. However, the diagnostic techniques used for this purpose have not yet been standardized for equids and this may generate conflicting results and erroneous conclusions regarding prevalence of the disease in this host population, as observed in the present study.
References
- Acosta ICL, Costa AP, Gennari SM, Marcili A. Survey of Trypanosoma and Leishmania in wild and domestic animals in an atlantic rainforest fragment and surroundings in the state of Espírito Santo, Brazil. J Med Entomol 2014; 51(3): 686-693. http://dx.doi.org/10.1603/ME13177 PMid:24897863.
» http://dx.doi.org/10.1603/ME13177 - Aguilar CM, Fernández E, Fernández R, Deane LM. Study of an outbreak of cutaneous leishmaniasis in Venezuela. The role of domestic animals. Mem Inst Oswaldo Cruz 1984; 79(2): 181-195. http://dx.doi.org/10.1590/S0074-02761984000200005 PMid:6535915.
» http://dx.doi.org/10.1590/S0074-02761984000200005 - Aguilar CM, Rangel EF, Garcia L, Fernandez E, Momen H, Grimaldi G Fo, et al. Zoonotic cutaneous leishmaniasis due to Leishmania (Viannia) braziliensis associated with domestic animals in Venezuela and Brazil. Mem Inst Oswaldo Cruz 1989; 84(1): 19-28. http://dx.doi.org/10.1590/S0074-02761989000100005 PMid:2319948.
» http://dx.doi.org/10.1590/S0074-02761989000100005 - Aharonson-Raz K, Baneth G, Lopes AP, Brancal H, Schallig H, Cardoso L, et al. Low seroprevalence of Leishmania infantum and Toxoplasma gondii in the horse population in Israel. Vector Borne Zoonotic Dis 2015; 15(12): 726-731. http://dx.doi.org/10.1089/vbz.2015.1826 PMid:26580094.
» http://dx.doi.org/10.1089/vbz.2015.1826 - Barbosa-Santos EG, Marzochi MC, Urtado W, Queirós F, Chicarino J, Pacheco RS. Leishmaniasis disseminated by Leishmania braziliensis in a mare (Equus cabalus) immunotherapy and chemotherapy assays. Mem Inst Oswaldo Cruz 1994; 89(2): 217-220. http://dx.doi.org/10.1590/S0074-02761994000200018 PMid:7885248.
» http://dx.doi.org/10.1590/S0074-02761994000200018 - Benassi JC, Benvenga GU, Ferreira HL, Soares RM, Silva DT, Pereira VF, et al. Molecular and serological detection of Leishmania spp. in horses from an endemic area for canine visceral leishmaniasis in southeastern Brazil. Pesq Vet Bras 2018; 38(6): 1058-1063. http://dx.doi.org/10.1590/1678-5150-pvb-5214
» http://dx.doi.org/10.1590/1678-5150-pvb-5214 - Evers F, Ferreira FP, Navarro IT, Mitsuka-Breganó R, Pagliari S, Monica TC, et al. Presence of anti-Leishmania spp. antibodies in slaughter horses in Brazil. Semina: Ciênc Agrár 2017; 38(6): 3921-3926. http://dx.doi.org/10.5433/1679-0359.2017v38n6p3921
» http://dx.doi.org/10.5433/1679-0359.2017v38n6p3921 - Feitosa FLF, Leal J, Mendes LCN, Peiró JR, Perri SHV, Lima VMF, et al. Estudo soroepidemiológico de leishmaniose em equinos na região de Araçatuba-SP, Brasil, área endêmica para leishmaniose visceral. Braz J Vet Res Anim Sci 2012; 49(6): 500-502. http://dx.doi.org/10.11606/issn.1678-4456.v49i6p500-502
» http://dx.doi.org/10.11606/issn.1678-4456.v49i6p500-502 - Fernández-Bellon H, Solano-Gallego L, Bardagí M, Alberola J, Ramis A, Ferrer L. Immune response to Leishmania infantum in healthy horses in Spain. Vet Parasitol 2006; 135(2): 181-185. http://dx.doi.org/10.1016/j.vetpar.2005.09.007 PMid:16213661.
» http://dx.doi.org/10.1016/j.vetpar.2005.09.007 - Gama A, Elias J, Ribeiro AJ, Alegria N, Schallig HDFH, Silva F, et al. Cutaneous leishmaniosis in a horse from northern Portugal. Vet Parasitol 2014; 200(1-2): 189-192. http://dx.doi.org/10.1016/j.vetpar.2013.12.005 PMid:24388338.
» http://dx.doi.org/10.1016/j.vetpar.2013.12.005 - Gao CH, Wang JY, Zhang S, Yang YT, Wang Y. Survey of wild and domestic mammals for infection with Leishmania infantum following an outbreak of desert zoonotic visceral leishmaniasis in jiashi, people’s Republic of China. PLoS One 2015; 10(7): e0132493. http://dx.doi.org/10.1371/journal.pone.0132493 PMid:26177101.
» http://dx.doi.org/10.1371/journal.pone.0132493 - Kenubih A, Dagnachew S, Almaw G, Abebe T, Takele Y, Hailu A, et al. Preliminary survey of domestic animal visceral leishmaniasis and risk factors in north-west Ethiopia. Trop Med Int Health 2015; 20(2): 205-210. http://dx.doi.org/10.1111/tmi.12418 PMid:25327874.
» http://dx.doi.org/10.1111/tmi.12418 - Koehler K, Stechele M, Hetzel U, Domingo M, Schönian G, Zahner H, et al. Cutaneous leishmaniosis in a horse in southern Germany caused by Leishmania infantum. Vet Parasitol 2002; 109(1-2): 9-17. http://dx.doi.org/10.1016/S0304-4017(02)00246-7 PMid:12383621.
» http://dx.doi.org/10.1016/S0304-4017(02)00246-7 - Kouam MK, Diakou A, Kanzoura V, Papadopoulos E, Gajadhar AA, Theodoropoulos G. A seroepidemiological study of exposure to Toxoplasma, Leishmania, Echinococcus and Trichinella in equids in Greece and analysis of risk factors. Vet Parasitol 2010; 170(1-2): 170-175. http://dx.doi.org/10.1016/j.vetpar.2010.02.004 PMid:20197215.
» http://dx.doi.org/10.1016/j.vetpar.2010.02.004 - Lopes AP, Sousa S, Dubey JP, Ribeiro AJ, Silvestre R, Cotovio M, et al. Prevalence of antibodies to Leishmania infantum and Toxoplasma gondii in horses from the north of Portugal. Parasit Vectors 2013; 6(1): 178. http://dx.doi.org/10.1186/1756-3305-6-178 PMid:23773870.
» http://dx.doi.org/10.1186/1756-3305-6-178 - Moher D, Liberati A, Tetzlaff J, Altman DG, Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009; 6(7): e1000097. http://dx.doi.org/10.1371/journal.pmed.1000097 PMid:19621072.
» http://dx.doi.org/10.1371/journal.pmed.1000097 - Mukhtar MM, Sharief AH, el Saffi SH, Harith AE, Higazzi TB, Adam AM, et al. Detection of antibodies to Leishmania donovani in animals in a kala-azar endemic region in eastern Sudan: a preliminary report. Trans R Soc Trop Med Hyg 2000; 94(1): 33-36. http://dx.doi.org/10.1016/S0035-9203(00)90429-2 PMid:10748894.
» http://dx.doi.org/10.1016/S0035-9203(00)90429-2 - Müller N, Welle M, Lobsiger L, Stoffel MH, Boghenbor KK, Hilbe M, et al. Occurrence of Leishmania sp. in cutaneous lesions of horses in Central Europe. Vet Parasitol 2009; 166(3-4): 346-351. http://dx.doi.org/10.1016/j.vetpar.2009.09.001 PMid:19800739.
» http://dx.doi.org/10.1016/j.vetpar.2009.09.001 - Oliveira PM, Garcia F, Evers F, Barbosa VM, Obando DCM, Nasciutti NR, et al. Seroepidemiology of Leishmania spp. in equids from Uberlândia, Minas Gerais, Brazil. Cienc Rural 2017; 47(5): e20160697. http://dx.doi.org/10.1590/0103-8478cr20160697
» http://dx.doi.org/10.1590/0103-8478cr20160697 - Pace D. Leishmaniasis. J Infect 2014; 69(Suppl 1): S10-S18. http://dx.doi.org/10.1016/j.jinf.2014.07.016 PMid:25238669.
» http://dx.doi.org/10.1016/j.jinf.2014.07.016 - Pereira MG, Galvão TF. Heterogeneidade e viés de publicação em revisões sistemáticas. Epidemiol Serv Saude 2014; 23(4): 775-778. http://dx.doi.org/10.5123/S1679-49742014000400021
» http://dx.doi.org/10.5123/S1679-49742014000400021 - Quaresma PF, Rêgo FD, Botelho HA, Silva SR, Moura AJ Jr, Teixeira RG No, et al. Wild, synanthropic and domestic hosts of Leishmania in an endemic area of cutaneous leishmaniasis in Minas Gerais State, Brazil. Trans R Soc Trop Med Hyg 2011; 105(10): 579-585. http://dx.doi.org/10.1016/j.trstmh.2011.07.005 PMid:21890159.
» http://dx.doi.org/10.1016/j.trstmh.2011.07.005 - Ramos-Vara JA, Ortiz-Santiago B, Segalès J, Dunstan RW. Cutaneous leishmaniasis in two horses. Vet Pathol 1996; 33(6): 731-734. http://dx.doi.org/10.1177/030098589603300619 PMid:8952039.
» http://dx.doi.org/10.1177/030098589603300619 - Rebollar-Tellez EA, Hamilton JGC, Ward RD. Response of female Lutzomyia longipalpis to host odour kairomones from human skin. Physiol Entomol 1999; 24(3): 220-226. http://dx.doi.org/10.1046/j.1365-3032.1999.00133.x
» http://dx.doi.org/10.1046/j.1365-3032.1999.00133.x - Reuss SM, Dunbar MD, Mays MBC, Owen JL, Mallicote MF, Archer LL, et al. Autochthonous Leishmania siamensis in Horse, Florida, USA. Emerg Infect Dis 2012; 18(9): 1545-1547. http://dx.doi.org/10.3201/eid1809.120184 PMid:22932732.
» http://dx.doi.org/10.3201/eid1809.120184 - Rohousova I, Talmi-Frank D, Kostalova T, Polanska N, Lestinova T, Kassahun A, et al. Exposure to Leishmania spp. and sand flies in domestic animals in northwestern Ethiopia. Parasit Vectors 2015; 8(1): 360. http://dx.doi.org/10.1186/s13071-015-0976-1 PMid:26152578.
» http://dx.doi.org/10.1186/s13071-015-0976-1 - Rolão N, Martins MJ, João A, Campino L. Equine infection with Leishmania in Portugal. Parasite 2005; 12(2): 183-186. http://dx.doi.org/10.1051/parasite/2005122183 PMid:15991833.
» http://dx.doi.org/10.1051/parasite/2005122183 - Sgorbini M, Bonelli F, Pizzolli I, Tognetti R, Corazza M. Seroprevalence of Leishmania sp. infection in healthy horses housed in endemic areas in Tuscany. J Equine Vet Sci 2014; 34(4): 572-574. http://dx.doi.org/10.1016/j.jevs.2013.09.009
» http://dx.doi.org/10.1016/j.jevs.2013.09.009 - Soares IR, Silva SO, Moreira FM, Prado LG, Fantini P, Maranhão RPA, et al. First evidence of autochthonous cases of Leishmania (Leishmania) infantum in horse (Equus caballus) in the Americas and mixed infection of Leishmania infantum and Leishmania (Viannia) braziliensis. Vet Parasitol 2013; 197(3-4): 665-669. http://dx.doi.org/10.1016/j.vetpar.2013.06.014 PMid:23845306.
» http://dx.doi.org/10.1016/j.vetpar.2013.06.014 - Solano-Gallego L, Fernández-Bellon H, Serra P, Gállego M, Ramis A, Fondevila D, et al. Cutaneous leishmaniosis in three horses in Spain. Equine Vet J 2003; 35(3): 320-323. http://dx.doi.org/10.2746/042516403776148336 PMid:12755438.
» http://dx.doi.org/10.2746/042516403776148336 - Truppel JH, Otomura F, Teodoro U, Massafera R, Costa-Ribeiro MCV, Catarino CM, et al. Can equids be a reservoir of Leishmania braziliensis in endemic areas? PLoS One 2014; 9(4): e93731. http://dx.doi.org/10.1371/journal.pone.0093731 PMid:24721908.
» http://dx.doi.org/10.1371/journal.pone.0093731 - Vedovello D Fo, Jorge FA, Lonardoni MVC, Teodoro U, Silveira TGV. American Cutaneous Leishmaniasis in horses from endemic areas in the North-Central Mesoregion of Paraná State, Brazil. Zoonoses Public Health 2008; 55(3): 149-155. http://dx.doi.org/10.1111/j.1863-2378.2008.01106.x PMid:18331518.
» http://dx.doi.org/10.1111/j.1863-2378.2008.01106.x - World Health Organization – WHO. Global leishmaniasis update, 2006–2015: a turning point in leishmaniasis surveillance. Weekly Epidemiological Record [online] 2017 [cited 2018 Nov 11]; 92(38): 557-572. Available from: http://apps.who.int/iris/bitstream/10665/258973/1/WER9238.pdf?ua=1
» http://apps.who.int/iris/bitstream/10665/258973/1/WER9238.pdf?ua=1 - Yoshida ELA, Correa FMA, Marques SA, Stolf HO, Dillon NL, Momen H, et al. Human, canine and equine (Equus caballus) leishmaniasis due to Leishmania braziliensis (= L. braziliensis brazilienzis) in the south-west region of São Paulo state, Brazil. Mem Inst Oswaldo Cruz 1990; 85(1): 133-134. http://dx.doi.org/10.1590/S0074-02761990000100026 PMid:2215227.
» http://dx.doi.org/10.1590/S0074-02761990000100026 - Yoshida ELA, Marques SA, Stolf HO, Barsotti LA, Bueno MMF, Sogayar R. Infecção natural de Equus caballus por Leishmania sp - São Paulo, Brasil (Breve comunicação científica). Rev Inst Med Trop São Paulo 1988; 30(2): 79-80. http://dx.doi.org/10.1590/S0036-46651988000200004 PMid:3201063.
» http://dx.doi.org/10.1590/S0036-46651988000200004
Publication Dates
-
Publication in this collection
03 Oct 2019 -
Date of issue
Oct-Dec 2019
History
-
Received
07 May 2019 -
Accepted
08 Aug 2019