Abstracts
PURPOSE: To evaluate the antioxidant effect of Propofol and N-Acetylcysteine (NAC) on intestinal ischemia/reperfusion (I/R) in rats by determining carbonyl protein level. METHODS: Forty Wistar rats were randomly assigned into the following groups: Control; Sham; I/R with Propofol; I/R with Propofol and NAC; I/R with Ketamine and Xylazine. The I/R groups underwent 60 minutes of ischemia and an equal period of reperfusion. Blood samples, collected by cardiac punction, were centrifuged for plasma obtainment. Protein carbonyl level in plasma samples was determined by immunoblotting. RESULTS: No significant difference in protein carbonyl level was found between Control and Sham groups (P>0.05). The highest reduction in protein carbonyl level (P<0.05) was obtained with the administration of Propofol and NAC (Group 4) in intestinal I/R procedure. CONCLUSION: The administration of Propofol and NAC showed the best antioxidant effect on oxidative stress in rats that underwent intestinal I/R procedure, suggesting a synergistic interaction.
Propofol; Acetylcysteine; Drug Synergism; Oxidative Stress; Ischemia; Reperfusion; Protein Carbonylation; Immunoblotting
OBJETIVO: Avaliar o efeito antioxidante do Propofol e N-Acetilcisteína (NAC) na isquemia/reperfusão (I/R) intestinal em ratos através da determinação do nível de proteína carbonilada. MÉTODOS: 40 ratos Wistar foram aleatoriamente distribuídos nos seguintes grupos: Controle; Sham; I/R com Propofol; I/R com Propofol e NAC; I/R com Ketamina e Xilazina. Os grupos I/R foram submetidos à isquemia durante 60 minutos e à reperfusão por igual período de tempo. Amostras de sangue, coletadas por punção cardíaca, foram centrifugadas para a obtenção de plasma. O nível de proteína carbonilada nas amostras de plasma foi determinado por imunoblotting. RESULTADOS: Nenhuma diferença significativa foi encontrada entre os grupos Controle e Sham (P>0.05). Uma redução marcante no nível de proteína carbonilada (P<0.05) foi obtida com a administração combinada de Propofol e NAC (Grupo 4) durante o procedimento de I/R intestinal, comparando-se com os demais grupos I/R testados. CONCLUSÃO: A administração de Propofol e NAC apresentou o melhor efeito antioxidante sobre o estresse oxidativo em ratos submetidos ao procedimento de I/R intestinal, sugerindo-se uma interação sinergística.
Propofol; Acetilcisteína; Sinergismo Farmacológico; Estresse Oxidativo; Isquemia; Reperfusão; Carbonilação Protéica; Immunoblotting
6 - ORIGINAL ARTICLE
ISCHEMIA-REPERFUSION
Propofol and N-Acetylcysteine attenuate oxidative stress induced by intestinal ischemia/reperfusion in rats. Protein carbonyl detection by immunoblotting1 Correspondence: Marcelo Aragão Insuellas de Azeredo Rua São Cristovão, 51 casa 01 / apto. 101 20940-001 Rio de Janeiro - RJ Brazil Phone: (55 21)2589-3160 charlinchaplin@uol.com.br parabolando@hotmail.com
Propofol e N-Acetilcisteína atenuam o estresse oxidativo induzido pela isquemia/reperfusão intestinal em ratos. Detecção de proteína carbonilada por immunoblotting
Marcelo Aragão Insuellas de AzeredoI; Luciana Aragão Insuellas de AzeredoII; Elis Cristina Araújo EleuthérioIII; Alberto SchanaiderIV
IMaster degree, Experimental Surgery, Faculty of Medicine, UFRJ, Rio de Janeiro, Brazil
IIPhD, Biochemistry Department, Institute of Chemistry, UFRJ, Rio de Janeiro, Brazil
IIIPhD, Full Professor, Biochemistry Department, Institute of Chemistry, UFRJ, Rio de Janeiro, Brazil
IVPhD, Full Professor, Department of Surgery, Faculty of Medicine, UFRJ, Rio de Janeiro, Brazil
Correspondence Correspondence: Marcelo Aragão Insuellas de Azeredo Rua São Cristovão, 51 casa 01 / apto. 101 20940-001 Rio de Janeiro - RJ Brazil Phone: (55 21)2589-3160 charlinchaplin@uol.com.br parabolando@hotmail.com
ABSTRACT
PURPOSE: To evaluate the antioxidant effect of Propofol and N-Acetylcysteine (NAC) on intestinal ischemia/reperfusion (I/R) in rats by determining carbonyl protein level.
METHODS: Forty Wistar rats were randomly assigned into the following groups: Control; Sham; I/R with Propofol; I/R with Propofol and NAC; I/R with Ketamine and Xylazine. The I/R groups underwent 60 minutes of ischemia and an equal period of reperfusion. Blood samples, collected by cardiac punction, were centrifuged for plasma obtainment. Protein carbonyl level in plasma samples was determined by immunoblotting.
RESULTS: No significant difference in protein carbonyl level was found between Control and Sham groups (P>0.05). The highest reduction in protein carbonyl level (P<0.05) was obtained with the administration of Propofol and NAC (Group 4) in intestinal I/R procedure.
CONCLUSION: The administration of Propofol and NAC showed the best antioxidant effect on oxidative stress in rats that underwent intestinal I/R procedure, suggesting a synergistic interaction.
Key words: Propofol. Acetylcysteine. Drug Synergism. Oxidative Stress. Ischemia. Reperfusion. Protein Carbonylation. Immunoblotting.
RESUMO
OBJETIVO: Avaliar o efeito antioxidante do Propofol e N-Acetilcisteína (NAC) na isquemia/reperfusão (I/R) intestinal em ratos através da determinação do nível de proteína carbonilada.
MÉTODOS: 40 ratos Wistar foram aleatoriamente distribuídos nos seguintes grupos: Controle; Sham; I/R com Propofol; I/R com Propofol e NAC; I/R com Ketamina e Xilazina. Os grupos I/R foram submetidos à isquemia durante 60 minutos e à reperfusão por igual período de tempo. Amostras de sangue, coletadas por punção cardíaca, foram centrifugadas para a obtenção de plasma. O nível de proteína carbonilada nas amostras de plasma foi determinado por imunoblotting.
RESULTADOS: Nenhuma diferença significativa foi encontrada entre os grupos Controle e Sham (P>0.05). Uma redução marcante no nível de proteína carbonilada (P<0.05) foi obtida com a administração combinada de Propofol e NAC (Grupo 4) durante o procedimento de I/R intestinal, comparando-se com os demais grupos I/R testados.
CONCLUSÃO: A administração de Propofol e NAC apresentou o melhor efeito antioxidante sobre o estresse oxidativo em ratos submetidos ao procedimento de I/R intestinal, sugerindo-se uma interação sinergística.
Descritores: Propofol. Acetilcisteína. Sinergismo Farmacológico. Estresse Oxidativo. Isquemia. Reperfusão. Carbonilação Protéica. Immunoblotting.
Introduction
Oxygen radical formation (ORF) occurs in most organ systems and has a pivotal role in various physiological and pathophysiological processes such as inflammation, aging, carcinogenesis, and others1. Reactive oxygen species have been increasingly implicated as mediators of intestinal ischemia-reperfusion (I/R)2.
Protein carbonylation has been shown an early event in oxidative stress, and nowadays is the most general and well-used biomarker of protein oxidation damage both in vivo and in vitro. Its measurement offers several advantages over the monitoring of lipid peroxidation; including the relative stability of oxidized proteins1. Besides, it could be a diagnostic tool to identify individuals at risk of developing organic functional impairments and diseases associated with oxidative injury induced by proteins. Immunoblotting is a specific and highly sensitive immunochemical assay for the determination of carbonyl level in a variety of biological samples3. However, only few studies linking ORF and protein carbonylation to intestinal disturbances are available2,4,5.
Administration of exogenous free radicals scavengers had been tried to reduce oxidative stress induced by I/R, and antioxidants may be particularly useful4. Propofol and N-Acetylcysteine (NAC), when separately administrated, exert beneficial effects on reperfusion injury6,7. As far as we concerned, no published report linking the antioxidant effect of Propofol and NAC in intestinal I/R procedure was described hitherto. Within this context, the purpose of this study was to evaluate the effect of Propofol and NAC on oxidative stress in rats undergoing intestinal I/R by the determination of carbonyl protein level.
Methods
This study was approved by the Ethics Committee for Laboratory Animals of the Federal University of Rio de Janeiro. Male Wistar rats (n = 40), each weighing 200 to 250 g and around three months old, were fed a standard laboratory diet. After food fasting of 12 h but with free access to water, rats were randomly separated in five groups (n = 8) as follow: Group 1- Control; Group 2 - Sham laparotomy with Propofol (10 mg/kg) and Fentanyl Citrate (6-8 µg/kg)8 IV ; Group 3 - I/R with Propofol (10 mg/kg) and Fentanyl Citrate (6-8 µg/kg)8 IV; Group 4 - I/R with Propofol (10 mg/kg)8, NAC (150 mg/kg)9 and and Fentanyl Citrate (6-8 µg/kg)8 IV; Group 5 - I/R with a solution of Ketamine (50 mg/kg) and Xylazine (10 mg/kg)8 IP. Accordingly to Lumb and Jones criteria10, all anesthetics were administered as bolus to maintain a stable light plane of anesthesia during intestinal I/R process.
Intestinal ischemia was produced by clamping four ileal branches of the superior mesenteric artery during 60 minutes and followed by an equal period of reperfusion. The infusion of NAC was performed a minute before reperfusion procedure was initiated. After 24 hours of I/R process, blood samples were obtained by cardiac punction, centrifuged for plasma obtainment and immediately frozen at -80 ºC for protein carbonyl assay.
Immunoblotting, a sensitive solid-phase immunochemical assay3, was used for the determination of the protein carbonyl content in plasma samples. Prior to the carbonyl content analysis, samples were treated with streptomycin C for the removal of nucleic acids, because DNA contains potentially reactive carbonyl groups. Plasma samples were normalized to a concentration of 5 µg protein/mL and then adsorbed to polyvinylidene difluoride (PVDF) membranes under vacuum pressure.
Afterward, the samples were derived with 2,4-dinitrophenylhydrazine (DNPH) (100 mg/mL) in 2NHCl solution for exactly 5 min, leading to the formation of stable hydrazone products. Before immunoassay, membranes were soaked in TBS buffer solution for 5 min, and immediately immersed in 5 % fat free milk TBS solution during 2 h for the blockage of non-specific protein binding sites. After, membranes were washed three times (5 minutes) in TBS/Tween solution, and DNPH-derived plasma protein carbonyl groups were sequentially reacted with a rabbit anti-DNPH primary monoclonal antibody for 2 hours and with goat anti-rabbit immunoglobulin G peroxidase-linked secondary antibody for 1 hour. Continuous shaking was used during all incubation and washing steps. Protein blanks were prepared by adding 2NHCl instead of DNPH to the assay containing protein sample. Image-analysis of the blots was carried out by Quantity One software (Bio-Rad).
All experimental data are expressed as the mean value with 95 % confidence interval, standard deviation and standard error mean, where applicable. Kruskal-Wallis one way analysis11 of variance test was used for the statistical comparisons among the experimental groups.
Results
A darkening was observed in all spots from the I/R groups samples, comparing to Control and Sham groups (Figure 1).
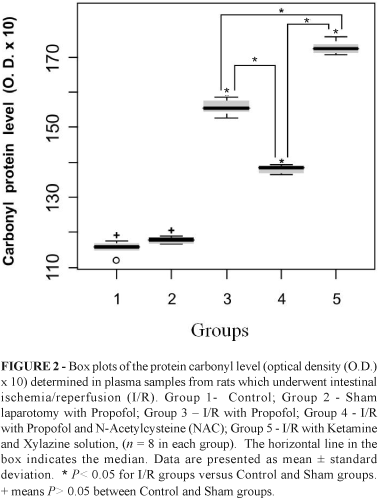
There was no significant difference in terms of protein carbonyl level (P > 0.05) between the Control and Sham groups (Figure 2).
In contrast, the level of protein carbonyl was significantly different (P< 0.05) among the I/R groups. The highest level of protein carbonyl level was found with Ketamine/Xylazine (Group 5). Propofol administration (Group 3) reduced significantly the protein carbonyl level (P< 0.05), but the highest reduction (P< 0.05) was obtained with the administration of Propofol and NAC (Group 4).
Discussion
The darkening spots observed in all I/R groups is probably correlated to the enhancement in the level of protein carbonyl in the plasma samples. Free radical-mediated oxidation of proteins results in the formation of carbonyl groups in quantities that reflect the intensity of the oxidative stress1,3. Oxidant stress is one of major factor contributing to intestinal I/R injury7. Immunoblotting assay provides valuable data on the correlation of carbonyl formation with oxidative stress comparing to the classical approach, which involves reactions with DNPH followed by a spectrophotometric quantification of the acid hydrazones3. The enhancement in protein carbonyl level observed in Group 5 (I/R with Ketamine and Xylazine) was in accordance with previous study performed in our laboratory5. Edema of intestinal mucosal villi and inflammatory infiltration of the lamina propria were also observed in the injury groups, and the crypt length was greater than in the normal intestine. Oxidation of plasma proteins has been also demonstrated in early experimental models of I/R injury of rat small bowel5. Besides, protein carbonylation has a potential to be a good biological marker not only to predict the severity of previous I/R injury, but also to help in the prognosis after surgical procedures in human diseases12.
The markedly reduction in protein carbonyl level obtained with the administration of Propofol and NAC suggest a synergistic interaction (Figure 2). Drug synergism occurs when the clinical effect of two or more drugs, given in combination, is greater than the sum of the effects of drugs administrated separately13. Some experts have suggested that more than one antioxidant is required for clinical effectiveness settings. According to Haramaki et al.14, the rationale is that antioxidants exist as a network wherein both lipid soluble (like tocopherals) and water soluble (ascorbate, glutathione, dihydrolipoic acid) molecules work for the removal of oxidant stress plus the regeneration of oxidant defenses. This theory reinforces the proposal of a combined scavenger therapeutic.
Propofol, characterized by a phenolic structure similar to a-tocopherol, provides an enhancement in antioxidant efficacies and erythrocytes protection against oxidative damage that have been demonstrated both in vitro and in vivo7,15,16,17. NAC, an attractive drug which has few side-effects, counteracts oxidative stress in many studies concerning I/R injury. Protective effects of NAC may lie within its ability to up regulate antioxidant systems6. The efficiency of NAC protective action depends on its dosage used; literature recommends 150 mg/kg as the most efficient one4. Saricaoglu et al.18 observed that oxidative stress, induced by I/R injury during arthroscopic knee surgery, was attenuated in the Group of patients which received NAC infusion and anesthesia with propofol, fentanyl and vecuronium bromide, maintained with desflurane. However, hitherto, there are no published reports linking the administration of Propofol and NAC in intestinal I/R procedure, as well as on the type of pharmacodynamic interaction that occurs between these two drugs.
Conclusion
Propofol has a beneficial interaction with NAC and hence potentially antioxidant effect on oxidative stress in rats that underwent intestinal I/R procedure. Meanwhile, further studies should be accomplished in order to better understand this scavenger synergism, before this treatment be established as a routine in clinical and surgical approaches.
Received: March 4, 2008
Review: May 5, 2008
Accepted: June 10, 2008
Conflict of interest: none
Financial source: none
1 Research performed at the Experimental Surgical Centre, Department of Surgery, Faculty of Medicine, Federal Post-Graduation Program, University of Rio de Janeiro (UFRJ), Brazil.
- 1. Dalle-Donne I, Rossi R, Giustarini D, Milzani A, Colombo R. Protein carbonyl groups as biomarkers of oxidative stress. Clin Chim Acta. 2003;329:23-38.
- 2. Thomson A, Hemphill D, Jeejeebhoy KN. Oxidative stress and antioxidants in intestinal disease. Digest Dis. 1998;16:152-8.
- 3. Robinson CE, Keshavarzian A, Pasco DS, Frommel TO, Winship DH, Holmes EW. Determination of protein carbonyl groups by immunoblotting. Anal Biochem 1999;266:48-57.
- 4. Byrka-Owczarek K, St?plewska-Mazur K, Krason M. The evaluation of the protective action of antioxidants on small intestine of rabbits experimentally injured by ischemia and reperfusion. J Pediatr Surg. 2004;39:1226-9.
- 5. Schanaider A, Menezes VJMT, Borchardt AC, Oliveira PL, Madis K. Oxidized tissue proteins after intestinal reperfusion injury in rats. Acta Cir Bras. 2005;20:434-6.
- 6. Hazinedaroglu SM, Dulger F, Kayaoglu HA, Pehlivan M, Serinsoz E, Canbolat O, Erverdi N. N-Acetylcysteine in intestinal reperfusion injury: an experimental study in rats. Anz J Surg. 2004;74:676-8.
- 7. Liu K, Rinne T, He W, Wang F, Xia Z. Propofol attenuates intestinal mucosa injury induced by intestinal ischemia-reperfusion in the rat. Can J Anesth. 2007;54:366-74.
- 8. Brookes ZLS, Brown NJ, Reilly CS. Response of the rat cremaster microcirculation to hemorrhage in vivo: differential effects of intravenous anesthetic agents. Shock. 2002;18:542-8.
- 9. Gu W, Weihrauch D, Tanaka K, Tessmer JP, Pagel PS, Kersten JR, Chilian WM, Warltier DC. Reactive oxygen species are critical mediators of coronary collateral development in a canine model. Am J Physiol Heart Circ Physiol. 2003;285:H1582-9.
- 10. Lumb WV, Jones EW. Veterinary anesthesia. Philadelphia: PA; 1973.
- 11. Kruskal WH, Wallis WA. Use of ranks in one-criterion variance analysis. J Am Stat Assoc. 1952;47(260):583-621.
- 12. Chevion M, Berenshtein E, Stadtman ER. Human studies related to protein oxidation: protein carbonyl content as a marker of damage. Free Radic Res. 2000;33(Suppl):S99-108.
- 13. Orser BA, Miller DR. Propofol-benzodiazepine interactions: insights from a "bench to bedside" approach. Can J Anesth. 2001;48:431-4.
- 14. Haramaki N, Stewart DB, Aggarwal S, Ikeda H, Reznick AZ, Packer L. Networking antioxidants in the isolated rat heart are selectively depleted by ischemia-reperfusion. Free Radical Bio Med. 1998;25:329-39.
- 15. Santos CHM, Gomes OM, Pontes JCDV, Miiji LNO, Higa EI. Uso do propofol (2,6 diisopropilfenol) como inibidor da lesão tecidual na isquemia e reperfusão mesentérica. Estudo experimental em ratos. Acta Cir Bras. 2003;18:347-54.
- 16. Tsuchiya M, Asada A, Kasahara E, Sato EF, Shindo M, Inoue M. Antioxidant protection of propofol and its recycling in erythrocyte membranes. Am J Respir Crit Care Med. 2002;65:54-60.
- 17. Volti GL, Murabito P, Attaguile G, Rodella LF, Astuto M, Di Giacomo C, Gullo A. Antioxidant properties of propofol when oxidative stress sleeps with patients. Excli J. 2006;5:25-32.
- 18. Saricaoglu F, Dal D, Salman AE, Atay ÖA, Doral MN, Salman MA, Kilinç K, Aypar Ü. Effect of low-dose N-acetyl-cysteine infusion on tourniquet-induced ischaemia-reperfusion injury in arthroscopic knee surgery. Acta Anaesthesiol Scand. 2005;49:847-51.
Publication Dates
-
Publication in this collection
03 Sept 2008 -
Date of issue
Oct 2008
History
-
Reviewed
05 May 2008 -
Received
04 Mar 2008 -
Accepted
10 June 2008