ABSTRACT:
This study analyzed the effectiveness of the essential oil of Aloysia triphylla (EOA) as an anesthetic and stress reducing agent in the transport of fat snook (Centropomus parallelus ). Juveniles were exposed to different concentrations of EOA (seawater-adapted: 25-300μL L-1; freshwater-adapted: 200μL L-1) to identify the anesthetic induction and recovery times. In an additional experiment, seawater-adapted fat snook were transported in plastic bags with 10 or 20μL L-1 EOA for 6 or 24h. The increased concentration of EOA proportionally decreased the time required for anesthesia induction. Mortality was 30 and 70% in fat snook transported with 10 and 20µL L-1, respectively, 24h after transport. The addition of 20µL L-1 EOA significantly reduced plasma cortisol levels during transport compared to the control group, but increased blood glucose levels after 6 and 12h of transport. The EOA reduced total ammonia and dissolved oxygen levels at the end of transport compared to control fish. The EOA is recommended for sedation (25-50µL L-1) and anesthesia (100-300µL L-1) but not for transport of fat snook because it increased mortality.
Key words:
anesthesia; blood parameters; fat snook; natural product; transport
RESUMO:
Este estudo verificou a eficácia do óleo essencial deAloysia triphylla (OEA) como um anestésico e redutor de estresse no transporte de robalo-pevas (Centropomus parallelus ). Os juvenis foram expostos a diferentes concentrações de OEA (adaptados à água do mar: 25-300µL L-1; adaptados à água doce: 200µL L-1) para identificar os tempos de indução e recuperação da anestesia. Em um experimento adicional, robalos-peva adaptados à água do mar foram transportados em sacos plásticos com 10 ou 20µL L-1 OE por 6 ou 24h. O aumento da concentração do OEA diminuiu proporcionalmente o tempo necessário para a indução da anestesia. A mortalidade foi de 30 e 70% em robalo-pevas transportados com 10 e 20µL L-1, respectivamente, 24h após o transporte. A adição de 20µL L-1 OEA reduziu significativamente o cortisol plasmático durante o transporte, comparado ao grupo controle, mas aumentou os níveis de glicose sanguínea após 6 e 12h de transporte. O OEA reduziu os níveis de amônia total e de oxigênio dissolvido ao final do transporte em comparação com o grupo controle. O OEA é recomendado para sedação (25-50µL L-1) e anestesia (100-300µL L-1), mas não para o transporte de robalo-peva, porque aumentou a mortalidade.
Palavras-chave:
anestesia; parâmetros sanguíneos; robalo-peva; produto natural; transporte
INTRODUCTION:
Handling is an inevitable precursor to any transportation, and may cause mechanical abrasions and induce some degree of stress (BECKER et al., 2012BECKER, A.G. et al. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiology and Biochemistry, v.38, p.789-796, 2012. Available from: http://link.springer.com/article/10.1007/s10695-011-9562-4>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-011-9562-4.
http://link.springer.com/article/10.1007...
; PARODI et al., 2014PARODI, T.V. et al. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and grey strains of silver catfish, Rhamdia quelen. Fish Physiology and Biochemistry, v.40, p.323-334, 2014. Available from: http://link.springer.com/article/10.1007/s10695-013-9845-z>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-013-9845-z.
http://link.springer.com/article/10.1007...
). Stress during transportation directly causes a physiologic reaction in most animals and is known to increase cortisol in fish (BARTON, 2002BARTON, B.A. Stress in fishes: a diversity of responses with particular reference to change in circulating costicosteroids. Integrative and Comparative Biology, v.42, p.517-525, 2002. Available from: http://icb.oxfordjournals.org/content/42/3/517.full>. Accessed: Dec. 15, 2015. doi: 10.1093/icb/42.3.517.
http://icb.oxfordjournals.org/content/42...
). Use of anesthetics is increasingly common in modern aquaculture and has practical relevance in diverse husbandry manipulations in addition to potentially attenuate physiologic response to stress (CUNHA et al., 2010CUNHA, M.A. et al. Essential oil of Lippia alba: a new anesthetic for silver catfish, Rhamdia quelen. Aquaculture , v.306, p.403-406, 2010. Available from: http://www.sciencedirect.com/science/journal/00448486/306/1-4>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2010.06.014.
http://www.sciencedirect.com/science/jou...
; BECKER et al., 2012BECKER, A.G. et al. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiology and Biochemistry, v.38, p.789-796, 2012. Available from: http://link.springer.com/article/10.1007/s10695-011-9562-4>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-011-9562-4.
http://link.springer.com/article/10.1007...
; PARODI et al., 2014PARODI, T.V. et al. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and grey strains of silver catfish, Rhamdia quelen. Fish Physiology and Biochemistry, v.40, p.323-334, 2014. Available from: http://link.springer.com/article/10.1007/s10695-013-9845-z>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-013-9845-z.
http://link.springer.com/article/10.1007...
).
The essential oil of Aloysia triphylla (L'Herit) Britton (EOA) presented anesthetic efficacy in the freshwater silver catfish Rhamdia quelen (PARODI et al., 2014PARODI, T.V. et al. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and grey strains of silver catfish, Rhamdia quelen. Fish Physiology and Biochemistry, v.40, p.323-334, 2014. Available from: http://link.springer.com/article/10.1007/s10695-013-9845-z>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-013-9845-z.
http://link.springer.com/article/10.1007...
), but no studies in other fish species have been performed. The fat snook, Centropomus parallelus , is an inshore tropical euryhaline species with high flesh quality and market value, as well as significant aquaculture potential (BOUCHEREAU et al., 2000BOUCHEREAU, J.L. et al. Selection of candidate fish species for farming in the Bay of Guaratuba, Brazil. Brazilian Archives of Biology and Technology, v.43, p.15-25 2000. Available from: http://dx.doi.org/10.1590/S1516-89132000000100003>. Accessed: Dec. 15, 2015. doi: 10.1590/S1516-89132000000100003.
http://dx.doi.org/10.1590/S1516-89132000...
). Therefore the aim of this study was to investigate the anesthetic induction and recovery times of the EOA, its efficacy as a stress-reducing agent during the transport of fat snook and the influence of salinity on the effect of EOA.
MATERIALS AND METHODS:
Juvenile fat snook (2.7±0.6g; 6.4±0.5cm) were acquired from a local supplier in Vitória, ES, Brazil and kept for seven days prior to the experiment in continuously aerated 100L aquaria with controlled temperatures (23.3ºC), salinity (30.6±0.2ppt), dissolved oxygen levels (6.0±0.4mg L-1), total ammonia nitrogen (1.17±0.10mg L-1) and conductivity (49.5±0.1mS). Water was renewed every two days. Animals received normal feeding two days after arriving in the laboratory and were fed daily to satiation with commercial carnivorous fish feed with 54% crude protein.
The cultivation and collection ofA. triphylla (voucher specimen SMDB No.11169) and preparation of the EOA from fresh leaves were performed according to PARODI et al. (2014PARODI, T.V. et al. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and grey strains of silver catfish, Rhamdia quelen. Fish Physiology and Biochemistry, v.40, p.323-334, 2014. Available from: http://link.springer.com/article/10.1007/s10695-013-9845-z>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-013-9845-z.
http://link.springer.com/article/10.1007...
). Analyses by gas chromatography-mass spectrometry aimed to identify and quantify the components of the EOA and were carried out in a hyphenated system equipped with a mass selective detector (AGILENT 6890, 5973 series). Analyses confirmed that E-citral (42.30%) and Z-citral (29.92%) were the major components of this EOA.
To test the effect of EOA on anesthesia induction and recovery, juveniles were individually transferred to an aquaria that contained 1 L of seawater and EOA at 25, 50, 100, 200 or 300µL L-1, first diluted in ethanol (1:10) (n=10 for each concentration tested). Each juvenile was used only once, and anesthesia stages were defined as explained by PARODI et al. (2014PARODI, T.V. et al. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and grey strains of silver catfish, Rhamdia quelen. Fish Physiology and Biochemistry, v.40, p.323-334, 2014. Available from: http://link.springer.com/article/10.1007/s10695-013-9845-z>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-013-9845-z.
http://link.springer.com/article/10.1007...
). The maximum observation time was 30min. Control experiments were performed using aquaria that contained only ethanol at the concentration that was equivalent to the highest EOA dilution (300µL L-1). After induction, the juveniles were transferred to anesthetic-free aquaria to measure the anesthesia recovery time.
As the fat snook is an euryhaline species that can survive in a wide salinity range, the anesthetic effect of the EOA was also verified in freshwater-adapted juveniles. In this experiment, salinity was gradually reduced (10ppt each day) and fish were then maintained for an additional seven days in freshwater prior to the experiment, as described by TONDOLO et al. (2013TONDOLO, J.S.M. et al. Anesthesia and transport of fat snook Centropomus parallelus with essential oil of Nectandra megapotamica (Spreng.) Mez. Neotropical Ichthyology, v.11, p.667-674, 2013. Available from: http://dx.doi.org/10.1590/S1679-62252013000300020>. Accessed: Apr. 29, 2016. doi: 10.1590/S1679-62252013000300020.
http://dx.doi.org/10.1590/S1679-62252013...
). The same procedure described for anesthetic induction and recovery of seawater-adapted fish was carried out in this group of animals at 200µL L-1 EOA (n=10). This concentration was chosen because it induced a fast anesthesia and recovery in the seawater-adapted fat snook.
To test the effect of EOA during transport, fat snook juveniles were placed in 45 plastic bags (volume 2L). Two juveniles were added per bag containing 0.5L of seawater with 0, 10 or 20µL L-1 EOA that had previously been diluted in ethanol (1:10), and they were transported for 6, 12 or 24h. There were five replicates for each combination of treatment and time. The concentrations tested in this experiment were slightly below the lowest concentration tested in the anesthesia experiment, which led to anesthesia stage 3a within 30 min, so the fat snook would only reach sedation during transportation. The control group (0µL L-1 EOA) was subjected to the same procedures but no anesthetic was added to the water. Bags were then inflated with oxygen, tied with rubber strings and packed in plastic boxes, as described by PARODI et al. (2014PARODI, T.V. et al. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and grey strains of silver catfish, Rhamdia quelen. Fish Physiology and Biochemistry, v.40, p.323-334, 2014. Available from: http://link.springer.com/article/10.1007/s10695-013-9845-z>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-013-9845-z.
http://link.springer.com/article/10.1007...
).
Water samples were collected before the plastic bags were closed and after transport for determination of dissolved oxygen (DO), temperature, conductivity and salinity with an oxygen meter (YSI, model Y5512 Yellow Springs, USA). Total ammonia nitrogen (TAN) levels were verified by the indophenol method according to APHA (1992APHA (AMERICAN PUBLIC HEALTH ASSOCIATION). Standard methods for examination of water and wastewater. 18.ed. Washington,1992. 541p.).
Blood samples (n=20 for each treatment, four fish per replicate) were collected from the caudal vein of each fish using heparinized 1mL syringes before and after transport (each fish was sampled only once) and the samples were kept on ice until the time of analysis, when samples were analyzed at room temperature as recommended by the commercial kits. Glucose concentration was measured with a portable digital glucometer (ACCU-CHECK(tm)) and lactate using a laboratory kit (Bioclin(tm)). Cortisol levels were measured using a competition ELISA kit (EIA, Kit 55050, Human(r)). This kit was previously validated for fish (SILVA et al., 2015SILVA, L.L et al. Effects of anesthesia with the essential oil of Ocimum gratissimum L. in parameters of fish stress. Revista Brasileira de Plantas Medicinais, v.17, p.215-223, 2015. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-05722015000200215>. Accessed: Apr. 29, 2016. doi: 10.1590/1983-084X/13_034.
http://www.scielo.br/scielo.php?script=s...
). The specificity of the test was evaluated by examining the extent of the parallelism between the standard curve (coefficient of variation = 13%) for human cortisol concentrations and the curve of a series of dilutions of the plasma samples in PBS (pH 7.4).
All data are expressed as mean ± S.E.M. Homogeneity of variances between treatments was tested with the Levene test following two-way ANOVA and Tukey's test. Comparisons between fresh- and seawater-adapted juveniles exposed to EOA 200µL L-1 were performed using the Student´s test. Analyses were performed using the software Statistica ver. 7.0 (StatSoft, Tulsa, OK). Evaluation of anesthetic activity was performed by regression analysis using the program Slide Write Plus (Advanced Graphics Software) version 4.0 (1996). The minimum significance level was set at P<0.05.
RESULTS AND DISCUSSION:
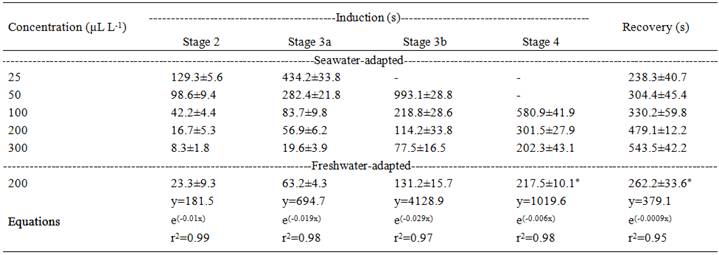
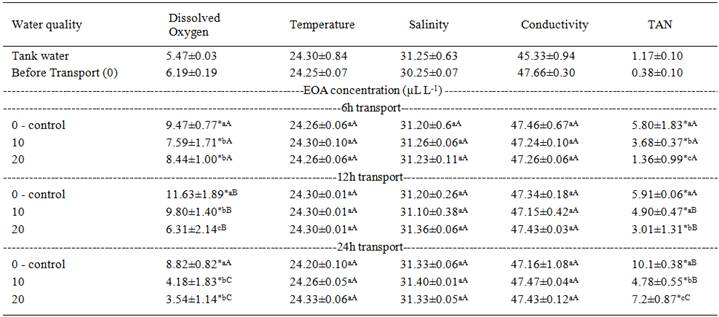
The sole application of ethanol did not produce an anesthetic effect even at a concentration equivalent to the greater dilution of the EOA, and no mortality resulted from anesthesia induction within the range tested for both groups (data not shown). The increasing concentration of EOA proportionally decreased the time required for sedation and anesthesia induction but proportionally increased the recovery time for seawater-adapted juveniles. When exposed to up to 50µL L-1 EOA, the fish did not reach stage 4 (deep anesthesia) within the 30min evaluation period. Freshwater-adapted juveniles exposed to 200µL L-1 EOA induced and recovered faster from deep anesthesia than seawater-adapted juveniles at the same EOA concentration (Table 1). Temperature, salinity and conductivity of the water after transport were not significantly different from before transport or between treatments at the end of transport. The water DO and TAN levels of the fat snook transported with EOA were significantly lower than the controls after 6, 12 and 24h of transport (except 10µL L-1 after 12h) (Table 2).
An anesthetic is effective when it is fast acting (about 3min), has a short recovery time (at most 10min), is easy to use, and presents a low risk to animals and humans with concentrations contained in a wide safety margin (PARK et al., 2009PARK, I-S. et al. Anesthetic effects of lidocaine-hydrochloride on water parameters in simulated transport experiment of juvenile winter flounder, Pleuronectes americanus. Aquaculture , v.294, p.76-79, 2009. Available from: http://www.sciencedirect.com/science/article/pii/S0044848609004608>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2009.05.011.
http://www.sciencedirect.com/science/art...
). The lowest concentration of EOA able to induce deep anesthesia in seawater-adapted fat snook was 100µL L-1, but it took around 10min to recover. The optimal EOA concentration for deep anesthesia with induction time and recovery from anesthesia within the limits proposed by PARK et al. (2009PARK, I-S. et al. Anesthetic effects of lidocaine-hydrochloride on water parameters in simulated transport experiment of juvenile winter flounder, Pleuronectes americanus. Aquaculture , v.294, p.76-79, 2009. Available from: http://www.sciencedirect.com/science/article/pii/S0044848609004608>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2009.05.011.
http://www.sciencedirect.com/science/art...
) was 300µL L-1 (induction to stage 4 in 3.3min and recovery in 9min). These results are similar to those observed for grey and albino strains of silver catfish: both anesthesia induction and recovery times with 300µL L-1 EOA took 3.6-6.7min and 8.3-9.5min, respectively (PARODI et al., 2014PARODI, T.V. et al. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and grey strains of silver catfish, Rhamdia quelen. Fish Physiology and Biochemistry, v.40, p.323-334, 2014. Available from: http://link.springer.com/article/10.1007/s10695-013-9845-z>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-013-9845-z.
http://link.springer.com/article/10.1007...
). The essential oil of Lippia alba in the 100-500µL L-1 concentration range induced deep anesthesia in silver catfish within 16.28-1.25min and recovery times were within 4.5-5.82min (CUNHA et al., 2010CUNHA, M.A. et al. Essential oil of Lippia alba: a new anesthetic for silver catfish, Rhamdia quelen. Aquaculture , v.306, p.403-406, 2010. Available from: http://www.sciencedirect.com/science/journal/00448486/306/1-4>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2010.06.014.
http://www.sciencedirect.com/science/jou...
). Menthol at 100-200mg L-1 can provoke deep anesthesia in tambaqui (Colossoma macropomum ) after 1-2 min, and recovery lasts 5-12min (FAÇANHA & GOMES, 2005FAÇANHA, M.F.; GOMES, L.C. Efficacy of menthol as an anesthetic for tambaqui (Colossoma macropomum, Characiformes: Characidae). Acta Amazonica, v.35, p.71-75, 2005. Available from: http://dx.doi.org/10.1590/S0044-59672005000100011>. Accessed: Dec. 15, 2015. doi: 10.1590/S0044-59672005000100011.
http://dx.doi.org/10.1590/S0044-59672005...
). Therefore, the optimal anesthetic concentration of the EOA is within the concentration range of other plant-originated anesthetics.
The anesthesia induction and recovery times of freshwater-adapted fat snook at 200µL L-1 EOA were faster than seawater-adapted ones. This difference is not related to changes in metabolism due to salinity because this parameter does not affect metabolic demand and growth of fat snook (TSUZUKI et al., 2007TSUZUKI, M.Y. et al. Survival, growth and digestive enzyme activity of juveniles of the fat snook (Centropomus parallelus) reared at different salinities. Aquaculture , v.271, p.319-325, 2007. Available from: http://www.sciencedirect.com/science/article/pii/S0044848607004127>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2007.05.002.
http://www.sciencedirect.com/science/art...
). Increased induction times with increased salinity were also observed in Caspian salmon (Salmo trutta caspius ) exposed to clove oil (GHAZILOU et al., 2010).
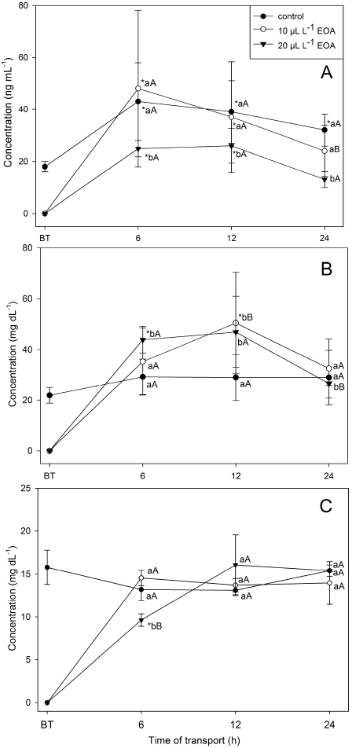
No mortality was observed in the control group after transport. Mortality after 24h of transport was 30 and 70% in fat snook transported with 10 and 20µL L-1 EOA, respectively. Plasma cortisol values increased after all times of transport compared to before transport, and the addition of 20µL L-1 EOA significantly reduced plasma cortisol levels (Figure 1A). Blood glucose and lactate levels were not significantly affected by transport in the control group, but fat snook transported for 6 and 12h with 20µL L-1 EOA (and 12h with 10µL L-1 EOA) presented significantly higher blood glucose levels than control fish at the same times. However, blood lactate levels after 6h, and blood glucose levels after 24h, were significantly lower in fat snook transported with 20µL L-1 EOA compared to control fish (Figure 1B, C).
Blood parameters before and after transport of Centropomus paralellus in plastic bags with essential oil of Aloysia triphylla (EOA) added to seawater. (A) cortisol, (B) glucose and (C) lactate. Control - only water, BT - before transport. Values represent mean ± SEM. Level of significance for all data: P<0.05. *Indicates a significant difference from the value before transport (BT). Different lowercase letters indicate significant differences between treatments at the same time of transport. Different capital letters indicate significant differences between times in the same treatment.
Fat snook mortality due to the use of 10 or 20µL L-1 EOA in the transport water cannot be explained by the increase of stress or impairment of water quality, because neither plasma cortisol nor dissolved oxygen and ammonia levels were related to mortality rates. The transport of cichlid Haplochromis obliquidens with 20µL L-1 clove oil for up to 48h did not induce mortality, but 30µL L-1 clove oil for 12 and 21h caused mortality (KAISER et al., 2006KAISER, H. et al. Testing clove oil as an anaesthetic for long-distance transport of live fish: the case of the Lake Victoria cichlid Haplochromis obliquidens. Jounal of Applied Ichtiology, v.22, p.510-514, 2006. Available from: http://onlinelibrary.wiley.com/doi/10.1111/j.1439-0426.2006.00786.x>. Accessed: Dec. 15, 2015. doi: 10.1111/j.1439-0426.2006.00786.x.
http://onlinelibrary.wiley.com/doi/10.11...
). Apparently, the concentration range tested in the present study for the EOA was not appropriate, and lower concentrations may reduce mortality.
Blood glucose levels of fat snook transported with EOA increased in the first 12h of transport while plasma cortisol decreased. Consequently, it is possible that the higher blood glucose levels were due to catecholamine release or an increased metabolism, because dissolved oxygen levels in the water of fat snook transported with EOA were lower than in control fish. The transport of three-spot gourami (Trichogaster trichopterus ) with 0.4mg L-1 metomidate for 12h also significantly increased plasma glucose levels (CROSBY et al., 2012CROSBY, T.C. et al. Effects of metomidate hydrochloride sedation on blood glucose and marketability of transported threespot gourami Trichogaster trichopterus. Journal of Aquatic Animal Health, v.24, p.73-80, 2012. Available from: http://afs.tandfonline.com/doi/full/10.1080/08997659.2012.675923>. Accessed: Dec. 15, 2015. doi:10.1080/08997659.2012.675923.
http://afs.tandfonline.com/doi/full/10.1...
). The EOA at 20µL L-1 was able to reduce plasma cortisol levels throughout transport compared to the control group. Cortisol is thought to be one of the mediators of the increase in plasma glucose levels observed during stressful events (BARTON et al., 2002BARTON, B.A. Stress in fishes: a diversity of responses with particular reference to change in circulating costicosteroids. Integrative and Comparative Biology, v.42, p.517-525, 2002. Available from: http://icb.oxfordjournals.org/content/42/3/517.full>. Accessed: Dec. 15, 2015. doi: 10.1093/icb/42.3.517.
http://icb.oxfordjournals.org/content/42...
).
The concentration of lactate may reflect the increased levels of physical activity as a response to the different stages of transport (ACERETE et al., 2004ACERETE, L. et al. Physiological responses in Eurasian perch (Perca fluviatilis, L.) subjected to stress by transport and handling. Aquaculture, v.237, p.167-178, 2004. Available from: http://www.sciencedirect.com/science/article/pii/S0044848604001802>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2004.03.018.
http://www.sciencedirect.com/science/art...
). Plastic bags used for transport reduced swimming area, but the fish have to fight against water movement at all times. The low plasma lactate concentrations seen in this study were possibly because, in spite of some swimming activity, fish were using aerobic support for their metabolism. That would explain the decreased dissolved oxygen levels and increased ammonia levels.
For the first 12h of fat snook transport (in most treatments), the DO levels were higher than at the beginning of transport because of the direct addition of pure oxygen in the plastic bags. The same results were observed in silver catfish transported with the essential oil of L. alba and eugenol (BECKER et al., 2012BECKER, A.G. et al. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiology and Biochemistry, v.38, p.789-796, 2012. Available from: http://link.springer.com/article/10.1007/s10695-011-9562-4>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-011-9562-4.
http://link.springer.com/article/10.1007...
). In the present study, the DO and TAN levels in the water of fat snook transported with EOA were lower than in the control group throughout transport. Clove oil (2, 5 or 10mg L-1) significantly reduced the un-ionized ammonia but failed to improve DO and pH at 12-24h in the transport water of Culter mongolicus . In contrast, the transport water of this species transported with MS 222 (10, 20 or 40mg L-1) in the first 12-24h had significantly higher DO and TAN than the control (LIN et al., 2012LIN, M. et al. Effects of two anesthetics on survival of juvenile Culter mongolicus during a simulated transport experiment. North American Journal ofAquaculture , v.74, p.541-546, 2012. Available from: http://afs.tandfonline.com/doi/full/10.1080/15222055.2012.700905#abstract>. Accessed: Dec. 15, 2015. doi:10.1080/15222055.2012.700905.
http://afs.tandfonline.com/doi/full/10.1...
). PARK et al. (2009PARK, I-S. et al. Anesthetic effects of lidocaine-hydrochloride on water parameters in simulated transport experiment of juvenile winter flounder, Pleuronectes americanus. Aquaculture , v.294, p.76-79, 2009. Available from: http://www.sciencedirect.com/science/article/pii/S0044848609004608>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2009.05.011.
http://www.sciencedirect.com/science/art...
) suggested that lidocaine hydrochloride at 5, 10 or 20mg L-1 decreased the metabolic activity of flounder (Pleuronectes americanus ), because this substance reduced ammonia excretion (by about 27.4-30.5%) and oxygen consumption (by about 82.7-86%) compared with a control group after 5h transport time. These studies reported that the overall reduction in ammonia excretion could be directly related to a decrease in the metabolic rate produced by sedation, as observed in fat snook transported with EOA, but as DO was also lower, this hypothesis is not fully supported.
CONCLUSION:
The EOA was effective in inducing slight sedation in the fat snook at 25-50µL L-1 and deep anesthesia at 100-300µL L-1. The concentration recommended for rapid deep anesthesia is 200µL L-1. Our results suggested that the use of the EOA in the concentrations tested is not advisable for the transport of fat snook due to the mortality provoked.
ACKNOWLEDGEMENTS
This research was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), process 470964/2009-0 and Ministério da Pesca e Aquicultura, Ministério da Ciência e Tecnologia/FINEP 01.02.0130.00. The experiments were conducted in accordance with the approval of the Ethics Committee on Animal Use of the Universidade Federal de Santa Maria (UFSM) - protocol n. 074/2014.
REFERENCES:
- ACERETE, L. et al. Physiological responses in Eurasian perch (Perca fluviatilis, L.) subjected to stress by transport and handling. Aquaculture, v.237, p.167-178, 2004. Available from: http://www.sciencedirect.com/science/article/pii/S0044848604001802>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2004.03.018.
» https://doi.org/10.1016/j.aquaculture.2004.03.018» http://www.sciencedirect.com/science/article/pii/S0044848604001802 - APHA (AMERICAN PUBLIC HEALTH ASSOCIATION). Standard methods for examination of water and wastewater. 18.ed. Washington,1992. 541p.
- BARTON, B.A. Stress in fishes: a diversity of responses with particular reference to change in circulating costicosteroids. Integrative and Comparative Biology, v.42, p.517-525, 2002. Available from: http://icb.oxfordjournals.org/content/42/3/517.full>. Accessed: Dec. 15, 2015. doi: 10.1093/icb/42.3.517.
» https://doi.org/10.1093/icb/42.3.517.» http://icb.oxfordjournals.org/content/42/3/517.full - BECKER, A.G. et al. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiology and Biochemistry, v.38, p.789-796, 2012. Available from: http://link.springer.com/article/10.1007/s10695-011-9562-4>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-011-9562-4.
» https://doi.org/10.1007/s10695-011-9562-4.» http://link.springer.com/article/10.1007/s10695-011-9562-4 - BOUCHEREAU, J.L. et al. Selection of candidate fish species for farming in the Bay of Guaratuba, Brazil. Brazilian Archives of Biology and Technology, v.43, p.15-25 2000. Available from: http://dx.doi.org/10.1590/S1516-89132000000100003>. Accessed: Dec. 15, 2015. doi: 10.1590/S1516-89132000000100003.
» https://doi.org/10.1590/S1516-89132000000100003.» http://dx.doi.org/10.1590/S1516-89132000000100003 - CROSBY, T.C. et al. Effects of metomidate hydrochloride sedation on blood glucose and marketability of transported threespot gourami Trichogaster trichopterus. Journal of Aquatic Animal Health, v.24, p.73-80, 2012. Available from: http://afs.tandfonline.com/doi/full/10.1080/08997659.2012.675923>. Accessed: Dec. 15, 2015. doi:10.1080/08997659.2012.675923.
» https://doi.org/10.1080/08997659.2012.675923» http://afs.tandfonline.com/doi/full/10.1080/08997659.2012.675923 - CUNHA, M.A. et al. Essential oil of Lippia alba: a new anesthetic for silver catfish, Rhamdia quelen. Aquaculture , v.306, p.403-406, 2010. Available from: http://www.sciencedirect.com/science/journal/00448486/306/1-4>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2010.06.014.
» https://doi.org/10.1016/j.aquaculture.2010.06.014» http://www.sciencedirect.com/science/journal/00448486/306/1-4 - FAÇANHA, M.F.; GOMES, L.C. Efficacy of menthol as an anesthetic for tambaqui (Colossoma macropomum, Characiformes: Characidae). Acta Amazonica, v.35, p.71-75, 2005. Available from: http://dx.doi.org/10.1590/S0044-59672005000100011>. Accessed: Dec. 15, 2015. doi: 10.1590/S0044-59672005000100011.
» https://doi.org/10.1590/S0044-59672005000100011.» http://dx.doi.org/10.1590/S0044-59672005000100011 - KAISER, H. et al. Testing clove oil as an anaesthetic for long-distance transport of live fish: the case of the Lake Victoria cichlid Haplochromis obliquidens. Jounal of Applied Ichtiology, v.22, p.510-514, 2006. Available from: http://onlinelibrary.wiley.com/doi/10.1111/j.1439-0426.2006.00786.x>. Accessed: Dec. 15, 2015. doi: 10.1111/j.1439-0426.2006.00786.x.
» https://doi.org/10.1111/j.1439-0426.2006.00786.x.» http://onlinelibrary.wiley.com/doi/10.1111/j.1439-0426.2006.00786.x - LIN, M. et al. Effects of two anesthetics on survival of juvenile Culter mongolicus during a simulated transport experiment. North American Journal ofAquaculture , v.74, p.541-546, 2012. Available from: http://afs.tandfonline.com/doi/full/10.1080/15222055.2012.700905#abstract>. Accessed: Dec. 15, 2015. doi:10.1080/15222055.2012.700905.
» https://doi.org/10.1080/15222055.2012.700905» http://afs.tandfonline.com/doi/full/10.1080/15222055.2012.700905#abstract - PARODI, T.V. et al. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and grey strains of silver catfish, Rhamdia quelen. Fish Physiology and Biochemistry, v.40, p.323-334, 2014. Available from: http://link.springer.com/article/10.1007/s10695-013-9845-z>. Accessed: Dec. 15, 2015. doi: 10.1007/s10695-013-9845-z.
» https://doi.org/10.1007/s10695-013-9845-z.» http://link.springer.com/article/10.1007/s10695-013-9845-z - PARK, I-S. et al. Anesthetic effects of lidocaine-hydrochloride on water parameters in simulated transport experiment of juvenile winter flounder, Pleuronectes americanus. Aquaculture , v.294, p.76-79, 2009. Available from: http://www.sciencedirect.com/science/article/pii/S0044848609004608>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2009.05.011.
» https://doi.org/10.1016/j.aquaculture.2009.05.011» http://www.sciencedirect.com/science/article/pii/S0044848609004608 - SILVA, L.L et al. Effects of anesthesia with the essential oil of Ocimum gratissimum L. in parameters of fish stress. Revista Brasileira de Plantas Medicinais, v.17, p.215-223, 2015. Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-05722015000200215>. Accessed: Apr. 29, 2016. doi: 10.1590/1983-084X/13_034.
» https://doi.org/10.1590/1983-084X/13_034.» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-05722015000200215 - TONDOLO, J.S.M. et al. Anesthesia and transport of fat snook Centropomus parallelus with essential oil of Nectandra megapotamica (Spreng.) Mez. Neotropical Ichthyology, v.11, p.667-674, 2013. Available from: http://dx.doi.org/10.1590/S1679-62252013000300020>. Accessed: Apr. 29, 2016. doi: 10.1590/S1679-62252013000300020.
» https://doi.org/10.1590/S1679-62252013000300020.» http://dx.doi.org/10.1590/S1679-62252013000300020 - TSUZUKI, M.Y. et al. Survival, growth and digestive enzyme activity of juveniles of the fat snook (Centropomus parallelus) reared at different salinities. Aquaculture , v.271, p.319-325, 2007. Available from: http://www.sciencedirect.com/science/article/pii/S0044848607004127>. Accessed: Dec. 15, 2015. doi:10.1016/j.aquaculture.2007.05.002.
» https://doi.org/10.1016/j.aquaculture.2007.05.002» http://www.sciencedirect.com/science/article/pii/S0044848607004127
-
1
CR-2016-0039.R1
Publication Dates
-
Publication in this collection
03 Oct 2016 -
Date of issue
Dec 2016
History
-
Received
15 Jan 2016 -
Accepted
29 June 2016 -
Reviewed
18 Sept 2016